Introduction
Peptide Oligonucleotide Conjugate Synthesis Methods have become increasingly important as peptide–oligonucleotide conjugates (POCs) gain attention in nucleic acid therapeutics, targeted delivery systems, and molecular diagnostics. These conjugates improve the stability, cellular uptake, and biological performance of nucleic acid–based drugs.
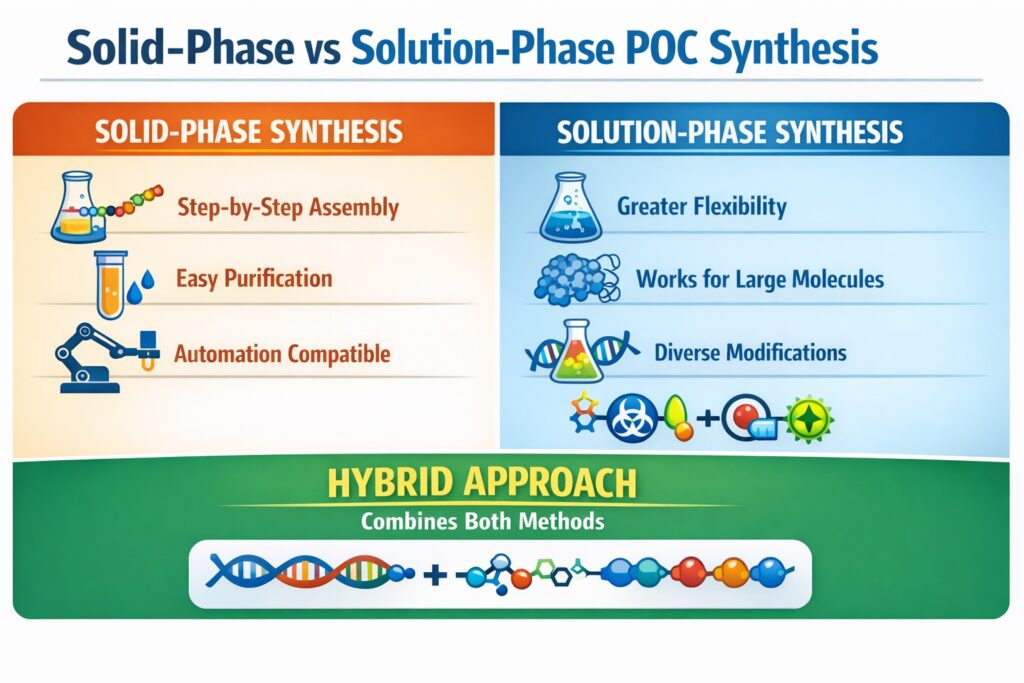
A key step in POC production is choosing between solid-phase synthesis and solution-phase synthesis. Each method offers different advantages depending on molecular complexity, chemical compatibility, and production scale. Solid-phase synthesis enables controlled, stepwise assembly on a solid support, while solution-phase synthesis links pre-synthesized fragments in solution.
Understanding the strengths and limitations of these two Peptide Oligonucleotide Conjugate Synthesis Methods helps researchers select the most efficient approach. This article compares both strategies and explains how they are used in modern nucleic acid therapeutic development.
Explore specialized support for your research: Learn more about Custom Synthesis for Drug Discovery
Share via:
Summary (Quick Insights)
- Peptide Oligonucleotide Conjugate Synthesis Methods primarily rely on two core strategies: solid-phase synthesis and solution-phase conjugation.
- Solid-phase synthesis provides high automation, precise sequence control, and simplified purification for complex peptide–oligonucleotide architectures.
- Solution-phase synthesis offers flexibility for large biomolecules and post-synthetic modification but often requires additional purification steps.
- Solid-phase methods are preferred for high-throughput and reproducible production, especially in therapeutic oligonucleotide development.
- Solution-phase approaches remain valuable for late-stage functionalization and large-scale conjugation strategies.
- Hybrid strategies combining both approaches are increasingly used to optimize yield, scalability, and structural complexity in modern Peptide Oligonucleotide Conjugate Synthesis Methods.
Solid-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Solid-phase synthesis allows scientists to construct peptide–oligonucleotide conjugates step by step on a solid support material. During the process, the growing molecule remains attached to an insoluble resin while chemical reactions occur. Because of this setup, excess reagents and byproducts can be removed easily through simple washing steps.
This method is widely used because it works well with automated peptide and oligonucleotide synthesizers. Automated systems perform repeated coupling and deprotection cycles with minimal manual work. As a result, laboratories can produce complex molecular sequences with excellent consistency.
Another advantage of solid-phase synthesis is improved process control. Each step of the synthesis can be monitored carefully before the next reaction begins. This is particularly helpful when synthesizing long oligonucleotide sequences or multi-component peptide conjugates.
The solid support also reduces product loss during purification. Since the molecule remains attached to the resin during most of the process, impurities can be removed without complicated separation techniques. This makes the workflow more efficient for both research laboratories and small-scale industrial production.
Accelerate your project with expert synthesis: View our Peptide Oligonucleotide Conjugates (POCs) Synthesis Services
How Solid-Phase POC Synthesis Works in Peptide Oligonucleotide Conjugate Synthesis Methods
In solid-phase Peptide Oligonucleotide Conjugate Synthesis Methods, the molecule is anchored to an insoluble support such as:
- Controlled pore glass (CPG)
- Polystyrene-based resins
- PEG-based solid supports
These materials provide a stable platform where chemical reactions can occur while the product remains attached to the support. The physical properties of the resin, such as pore size and chemical stability, influence synthesis efficiency. Selecting the correct support is therefore very important for successful synthesis.
The synthesis usually follows several repeated cycles:
- Attachment of the first fragment to the solid support
- Sequential nucleotide or peptide coupling
- Protection and deprotection cycles
- Final cleavage and purification
Each cycle adds a new molecular unit to the growing chain. Protecting groups are used to prevent unwanted side reactions during the coupling steps. After the full sequence is completed, the final molecule is released from the support and purified.
Understand the structural foundations: Discover the different Types of Peptide Oligonucleotide Conjugates
Two main variants of this approach exist:
- Sequential synthesis strategy
- Post-synthetic conjugation on solid support
These strategies differ in how the peptide and oligonucleotide components are assembled. The final choice depends on sequence complexity, chemical compatibility, and the desired structure of the conjugate.
Sequential Solid-Phase Assembly
Sequential assembly builds both peptide and oligonucleotide segments during one continuous synthesis process. The entire conjugate is assembled step by step on the same solid support. This integrated strategy reduces the need for separate synthesis procedures.
However, this approach requires compatible protecting groups and reaction conditions for both molecules. Peptides and oligonucleotides often require different chemical environments, which makes synthesis planning more challenging.
Key characteristics include:
- Phosphoramidite chemistry for oligonucleotides
- Fmoc or Boc chemistry for peptides
- Orthogonal protecting groups
Orthogonal protecting groups allow selective removal of specific groups without affecting others. This ensures that peptide and nucleotide synthesis steps can proceed independently during the same workflow.
Advantages include:
- Precise site-specific conjugation
- Minimal intermediate purification
- Compatibility with automated synthesis platforms
These features make sequential solid-phase synthesis attractive for laboratories focused on reproducibility. However, differences in solvent systems, temperature stability, and protecting group chemistry can still create technical challenges.
Dive deeper into the chemical bonds: Read about Peptide Oligonucleotide Conjugate Linker Chemistry
Solid-Phase Post-Synthetic Conjugation
Another strategy involves synthesizing peptide and oligonucleotide fragments separately and then coupling them while the oligonucleotide remains attached to the solid support. This method combines fragment synthesis with the purification advantages of solid-phase workflows.
Preparing fragments independently allows researchers to optimize reaction conditions for each component. Once both fragments are ready, they can be connected through a specific chemical reaction. The solid support still enables easy removal of excess reagents through washing.
Typical chemistries include:
- Amide bond formation
- Click chemistry (CuAAC)
- Thiol-maleimide coupling
- Disulfide formation
These reactions are widely used in bioconjugation chemistry because they are selective and efficient. They allow peptide and oligonucleotide molecules to be connected without disturbing other parts of the structure.
Purification becomes simpler because unreacted reagents can be washed away while the conjugate remains attached to the resin.
Optimize your synthesis and validation workflow: Explore POC Synthesis and Characterization services
Advantages of Solid-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Solid-phase Peptide Oligonucleotide Conjugate Synthesis Methods offer several benefits for both academic research and industrial production. These methods provide strong control over reaction conditions while simplifying purification steps.
1. Simplified Purification
Since the product remains attached to a resin during synthesis, impurities can be removed by washing the support. This eliminates the need for repeated chromatographic purification steps during intermediate stages.
Benefits include:
- Reduced chromatographic purification
- Lower process complexity
- Higher final purity
This is especially useful when synthesizing long or chemically sensitive sequences.
2. Automation Compatibility
Solid-phase synthesis works well with automated instruments used in peptide and oligonucleotide production. Automation reduces the chance of human error and ensures consistent reaction conditions.
This provides:
- High reproducibility
- Accurate stoichiometry control
- Scalable production workflows
Automated synthesizers can perform hundreds of reaction cycles with minimal supervision.
3. High Coupling Efficiency
Sequential addition of activated monomers often achieves coupling efficiencies above 98–99% per cycle. High efficiency is critical when building long sequences because small losses accumulate during multiple cycles.
Consistent reaction efficiency helps maintain the correct molecular sequence and reduces incomplete products.
4. Site-Specific Conjugation
Solid-phase synthesis enables accurate conjugation at defined locations such as:
- 3′ or 5′ termini
- Internal nucleotide positions
- Peptide N- or C-terminus
This precise control is important when designing conjugates for targeted therapeutic applications.
Ensure high-quality results for your conjugates: Learn about QC Testing for Peptide Oligonucleotide Conjugates
Limitations of Solid-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Although solid-phase synthesis has many advantages, it also presents certain technical challenges.
Chemical Compatibility Constraints
Peptide and oligonucleotide synthesis often require different chemical conditions:
Component — Typical Conditions
Peptide synthesis — Basic deprotection (piperidine)
Oligonucleotide synthesis — Acid-labile protecting groups
Balancing these conditions in a single synthesis workflow can be difficult. Researchers may need to modify protecting groups or adjust reaction steps to maintain compatibility.
Resin Loading Limitations
Solid supports can impose physical constraints on:
- Reaction diffusion
- Steric accessibility
- Scale-up capacity
Large biomolecules may experience steric hindrance while attached to the resin surface. Limited reagent diffusion inside resin pores may also affect reaction efficiency.
Cost Considerations
High-quality resins, reagents, and automated synthesizers can increase production costs. Laboratories must invest in specialized equipment and high-purity chemicals to achieve consistent results.
However, these costs are often justified by the improved reproducibility and efficiency of the method.
Solution-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Solution-phase Peptide Oligonucleotide Conjugate Synthesis Methods involve connecting separately synthesized peptide and oligonucleotide fragments in solution using chemical ligation reactions. Unlike solid-phase synthesis, both molecular components are produced independently before they are combined.
This approach allows each fragment to be synthesized under its optimal chemical conditions. It also avoids compatibility issues between peptide and oligonucleotide synthesis chemistries.
Solution-phase synthesis is especially useful for large biomolecules or complex structures that are difficult to assemble on solid supports.
Researchers also use this method for post-synthesis modifications. Functional groups such as targeting ligands or fluorescent probes can be attached after the main synthesis process is completed.
Partner with a specialized research organization: Discover our Peptide Oligonucleotide Conjugates CRO capabilities
Typical Workflow in Solution-Phase POC Synthesis
The process generally follows three steps:
- Independent synthesis of peptide and oligonucleotide fragments
- Introduction of reactive functional groups
- Conjugation via selective coupling chemistry
Each component is purified and verified before the final conjugation reaction takes place.
Common conjugation reactions include:
- Amide coupling
- Thiol–maleimide reactions
- Copper-catalyzed azide–alkyne cycloaddition (click chemistry)
- Disulfide bond formation
These reactions are widely used because they provide strong selectivity and efficient coupling.
Advantages of Solution-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Greater Chemical Flexibility
Solution-phase Peptide Oligonucleotide Conjugate Synthesis Methods allow greater chemical diversity because researchers are not limited by the compatibility of a single synthesis platform.
Scientists can introduce:
- Non-natural amino acids
- Modified nucleotides
- Targeting ligands
- Fluorescent probes
These modifications can improve biological performance and targeting capability.
Compatibility with Large Biomolecules
Solution-phase conjugation works well for:
- Long oligonucleotide sequences
- Complex peptide structures
- Multivalent constructs
Large molecules have greater mobility in solution, which improves reaction efficiency.
Easier Optimization
Each molecular fragment can be optimized separately before conjugation. Researchers can adjust synthesis conditions, purification methods, and reaction parameters independently.
This modular approach often improves the final yield and purity.
Bridge the gap from research to clinical application: See our CMC Services for Peptide Oligonucleotide Conjugates
Limitations of Solution-Phase POC Synthesis
Complex Purification
Solution-phase reactions produce mixtures that require purification after conjugation.
Common methods include:
- HPLC
- Ion-exchange chromatography
- Size-exclusion chromatography
These techniques help separate the desired conjugate from byproducts and unreacted fragments.
Lower Overall Yield
Fragment ligation reactions can sometimes be less efficient than stepwise solid-phase coupling. Because two large molecules must react in solution, steric effects may reduce reaction efficiency.
Multi-Step Workflow
Solution-phase synthesis introduces additional steps such as:
- Functional group installation
- Activation reactions
- Purification of intermediates
These steps increase the overall complexity of the synthesis process.
Solid-Phase vs Solution-Phase Peptide Oligonucleotide Conjugate Synthesis Methods
Below is a comparative overview of the two approaches.
| Feature | Solid-Phase Synthesis | Solution-Phase Synthesis |
|---|---|---|
| Reaction control | High | Moderate |
| Automation | Excellent | Limited |
| Purification | Simplified | Complex |
| Scalability | Moderate | High |
| Chemical flexibility | Moderate | High |
| Compatibility with large biomolecules | Limited | Excellent |
| Yield for long sequences | High | Variable |
Key insight:
Most modern Peptide Oligonucleotide Conjugate Synthesis Methods combine solid-phase synthesis for initial assembly with solution-phase chemistry for final conjugation steps.
Hybrid Strategies in Modern Peptide Oligonucleotide Conjugate Synthesis Methods
Many advanced synthesis workflows combine both solid-phase and solution-phase techniques. These hybrid strategies allow researchers to use the strengths of each method while minimizing their limitations.
Typical hybrid workflow:
- Solid-phase oligonucleotide synthesis
- Solid-phase peptide synthesis
- Solution-phase conjugation via click chemistry
By producing the components separately and linking them later, scientists can maintain high reaction efficiency and flexibility in molecular design.
Hybrid strategies offer:
- Better reaction efficiency
- Greater structural flexibility
- Scalable manufacturing
These methods are widely used in antisense therapeutics, siRNA delivery systems, and targeted nucleic acid medicines.
Choosing the Right Peptide Oligonucleotide Conjugate Synthesis Method
Selecting the correct Peptide Oligonucleotide Conjugate Synthesis Method depends on several factors including molecular size, chemical compatibility, production scale, and required modifications.
Consider Solid-Phase When
- Precise conjugation sites are required
- High-throughput synthesis is needed
- Automation is available
- Peptide length is relatively short
Consider Solution-Phase When
- Large peptides or proteins are involved
- Complex modifications are needed
- Large-scale production is required
In many research environments, a hybrid strategy offers the best balance of precision, flexibility, and scalability.
Conclusion
Solid-phase and solution-phase approaches represent the two main strategies used in Peptide Oligonucleotide Conjugate Synthesis Methods. Each technique provides unique benefits and limitations depending on the application and molecular complexity.
Solid-phase synthesis offers excellent sequence control, automation compatibility, and simplified purification. These advantages make it ideal for building well-defined molecular sequences with high precision.
Solution-phase synthesis provides greater flexibility and scalability, particularly for large biomolecules or complex molecular architectures. Researchers can modify fragments independently before final conjugation.
As peptide–oligonucleotide therapeutics continue to grow in gene therapy, targeted drug delivery, and diagnostics, hybrid strategies that combine both approaches are becoming increasingly important.
Organizations specializing in advanced Peptide Oligonucleotide Conjugate Synthesis Methods help researchers design optimized workflows for complex biomolecular projects. Their expertise supports efficient and reliable production of high-quality conjugates.
Master the functional performance of your molecules: Learn about Peptide Oligonucleotide Conjugates Mechanism of Action
Contact Us
If you are working on peptide–oligonucleotide conjugates and need support with custom Peptide Oligonucleotide Conjugate Synthesis Methods, our scientific team can help. Our specialists provide technical guidance, synthesis optimization, and analytical support for a wide range of biomolecular research projects.
Contact our experts: Contact us
FAQs: Solid-Phase vs Solution-Phase POC Synthesis
Hybrid strategies combine solid-phase and solution-phase techniques within the same workflow. Researchers often build molecular fragments separately and then join them using efficient conjugation reactions. This approach improves flexibility, yield, and scalability in modern Peptide Oligonucleotide Conjugate Synthesis Methods.
Several chemical reactions are used to connect peptide and oligonucleotide fragments. Common examples include amide bond formation, click chemistry (CuAAC), thiol–maleimide reactions, and disulfide linkages. These reactions are preferred because they are selective, efficient, and compatible with sensitive biomolecules.
Solution-phase synthesis generally provides better scalability for large molecules and industrial applications. Reactions in solution allow larger batch sizes and easier handling of complex biomolecular structures. However, additional purification steps are usually required to obtain a highly pure final product.
One major challenge is managing the different chemical requirements of peptides and oligonucleotides. Each molecule may require different protecting groups and reaction conditions. Researchers must carefully design the synthesis workflow to avoid unwanted side reactions and maintain efficiency.
After conjugation in solution, purification is needed to separate the final product from unreacted fragments and byproducts. Techniques such as HPLC, ion-exchange chromatography, and size-exclusion chromatography are commonly used. These analytical methods help ensure that the final conjugate meets the required purity standards.
Click chemistry is a widely used reaction in Peptide Oligonucleotide Conjugate Synthesis Methods because it is fast, selective, and reliable. It allows peptide and oligonucleotide fragments to be connected under mild reaction conditions. This helps preserve the structural integrity of sensitive biomolecules.
Solution-phase synthesis usually allows more flexibility when designing complex molecular structures. Researchers can introduce special chemical groups, targeting ligands, or fluorescent markers after fragment synthesis. This makes the approach useful for advanced therapeutic and diagnostic applications.
Reference:
- Klabenkova, K., Fokina, A., & Stetsenko, D. (2021). Chemistry of peptide-oligonucleotide conjugates: A review. Molecules, 26(17), 5420. https://doi.org/10.3390/molecules26175420
- Venkatesan, N., & Kim, B. H. (2006). Peptide conjugates of oligonucleotides: Synthesis and applications. Chemical Reviews, 106(9), 3712–3761. https://doi.org/10.1021/cr0502448
- Naganuma, M., Tsuji, G., Amiya, M., Hirai, R., Higuchi, Y., Hata, N., Nozawa, S., Watanabe, D., Nakajima, T., & Demizu, Y. (2025). High-resolution HPLC for separating peptide–oligonucleotide conjugates. ACS Omega, 10(20), 20578–20584. https://doi.org/10.1021/acsomega.5c01308
- Tung, C. H., & Stein, S. (2000). Preparation and applications of peptide–oligonucleotide conjugates. Bioconjugate Chemistry, 11(5), 605–618. https://doi.org/10.1021/bc0000334



