Introduction
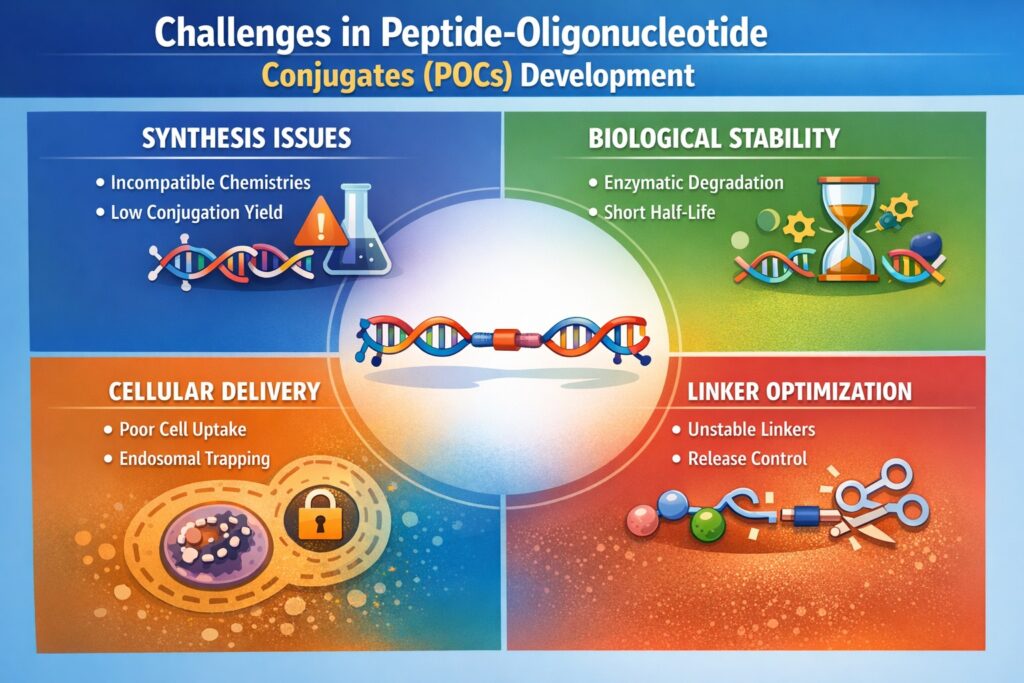
The Challenges in Peptide Oligonucleotide Conjugates development arise mainly from the complexity of combining peptides with nucleic acids into one functional therapeutic molecule. Both components must maintain their biological activity while remaining stable during synthesis, storage, and circulation in the body. Achieving this balance requires careful coordination of chemistry, molecular biology, and pharmaceutical engineering.
Peptide-oligonucleotide conjugates (POCs) offer strong potential for targeted drug delivery and gene regulation. Peptides can help guide therapeutics to specific tissues or improve cellular entry, while oligonucleotides can modulate gene expression or silence harmful RNA. When these molecules are successfully combined, they can enhance therapeutic precision and improve treatment outcomes.
Interest in POCs has grown rapidly with advances in RNA therapeutics, antisense technologies, and precision medicine. Researchers are exploring these conjugates for treating genetic diseases, cancer, and rare disorders. However, despite their promise, several technical barriers remain, making it important to understand the key Challenges in Peptide Oligonucleotide Conjugates development that affect synthesis, delivery, stability, and manufacturing.
Explore specialized support for your research: Learn more about Custom Synthesis for Drug Discovery
Share via:
Summary of Key Insights
- Peptide-oligonucleotide conjugates (POCs) development faces complex synthetic compatibility issues because peptide and oligonucleotide chemistries require different reaction conditions and protecting strategies.
- Stability in biological environments remains a major barrier, as nucleases, proteases, and serum components can degrade conjugates or alter their function.
- Efficient intracellular delivery and endosomal escape are among the most significant challenges in peptide-oligonucleotide conjugates development.
- Manufacturing scalability and analytical characterization require advanced techniques due to heterogeneous conjugation and structural complexity.
- Linker chemistry and conjugation strategy selection directly influence pharmacokinetics, biodistribution, and therapeutic efficacy.
- Regulatory and CMC challenges arise because POCs combine properties of both biologics and nucleic-acid therapeutics.
Challenges in Peptide Oligonucleotide Conjugates Development: Synthetic Compatibility
One of the most important Challenges in Peptide Oligonucleotide Conjugates development is the difference between peptide synthesis and oligonucleotide synthesis chemistry. These two molecules are produced using entirely different chemical platforms, which makes their integration technically difficult. When the two components are joined together, reaction conditions must be carefully selected so that neither structure is damaged.
Peptides are generally synthesized using solid-phase peptide synthesis (SPPS), which often involves acidic deprotection steps. In contrast, oligonucleotides are typically synthesized using phosphoramidite chemistry, which requires basic conditions for deprotection. These opposing chemical environments can create compatibility issues during conjugation.
For example, the strong bases used during oligonucleotide synthesis may damage sensitive peptide sequences. Likewise, acidic conditions used in peptide synthesis can affect nucleotide stability. Because of these differences, scientists must design orthogonal reactions that allow both components to remain intact during the assembly process.
Even small variations in reaction conditions can significantly affect yield and purity. As a result, extensive optimization is often required during early development stages to ensure successful conjugate formation.
Key synthetic challenges
- Protecting group incompatibility between peptide and nucleotide chemistries
- Degradation of sensitive peptide sequences during oligonucleotide deprotection
- Limited orthogonal conjugation strategies
- Reduced yields during multi-step synthesis
Common synthesis strategies
| Strategy | Description | Key Challenge |
|---|---|---|
| Stepwise solid-phase synthesis | Peptide and oligonucleotide assembled sequentially | Harsh conditions can degrade peptide |
| Post-synthetic conjugation | Peptide and oligonucleotide synthesized separately then linked | Requires efficient coupling chemistry |
| Click chemistry conjugation | Bioorthogonal reactions like azide-alkyne cycloaddition | Requires functional group incorporation |
According to Lu et al. (2010) and Venkatesan & Kim (2006), choosing the right conjugation strategy is essential because the stability and functionality of the final POC depend heavily on reaction compatibility and linker design.
Optimize your synthesis workflow: View our Peptide Oligonucleotide Conjugate Synthesis Methods
Challenges in Peptide Oligonucleotide Conjugates Development: Stability in Biological Systems
Maintaining stability inside biological environments is another critical aspect of the Challenges in Peptide Oligonucleotide Conjugates development. After administration, the conjugate must survive long enough in the bloodstream and tissues to reach its therapeutic target. However, many biological enzymes can quickly degrade peptide or nucleic-acid components.
Oligonucleotides are highly susceptible to degradation by nucleases, while peptides can be broken down by proteolytic enzymes. When these two molecules are linked together, the resulting conjugate may face multiple degradation pathways at the same time. This can significantly shorten the therapeutic lifetime of the molecule.
Major stability concerns
- Enzymatic degradation by nucleases and proteases
- Hydrolysis of linker chemistry
- Loss of structural integrity during circulation
- Reduced half-life in plasma
Research suggests that chemical modifications can greatly improve stability. Examples include phosphorothioate backbones, 2′-O-methyl nucleotide modifications, and peptide cyclization (Anwar et al., 2023). These modifications help protect the molecule from enzymatic attack and extend its circulation time.
Stability optimization strategies
- Backbone modifications in oligonucleotides
- Use of stable amide or triazole linkers
- Peptide cyclization or unnatural amino acids
- PEGylation or lipid conjugation
While these strategies improve stability, they also add complexity to the manufacturing and analytical process. Therefore, careful optimization is necessary to balance stability with therapeutic performance.
Understand the structural variations for stability: Discover the different Types of Peptide Oligonucleotide Conjugates
Challenges in Peptide Oligonucleotide Conjugates Development: Cellular Delivery
Efficient intracellular delivery remains one of the biggest Challenges in Peptide Oligonucleotide Conjugates development. Oligonucleotides are large, negatively charged molecules that cannot easily cross the lipid membrane of cells. Without assistance, they show very limited cellular uptake.
Even when the conjugates enter cells through endocytosis, they often become trapped inside endosomes. If the molecule cannot escape from these vesicles, it may never reach the cytoplasm or nucleus where its therapeutic target is located.
Key delivery barriers
- Poor membrane permeability
- Endosomal trapping after cellular uptake
- Non-specific tissue distribution
- Rapid renal clearance
To address this issue, researchers frequently use cell-penetrating peptides (CPPs). These peptides can improve cellular uptake and transport molecules across membranes. However, their use requires careful optimization.
Potential limitations include:
- Cytotoxicity at higher concentrations
- Non-specific uptake in off-target tissues
- Possible immunogenic responses
According to Juliano et al. (2012) and Hammond et al. (2021), improving endosomal escape efficiency is one of the most active research areas in oligonucleotide therapeutics. Scientists are exploring pH-responsive peptides, membrane-disruptive motifs, and nanoparticle carriers to improve delivery.
Master the functional performance of your molecules: Learn about Peptide Oligonucleotide Conjugates Mechanism of Action
Challenges in Peptide Oligonucleotide Conjugates Development: Linker Design
Linker chemistry plays a central role in addressing the Challenges in Peptide Oligonucleotide Conjugates development. The linker connects the peptide and oligonucleotide components and determines how the conjugate behaves in circulation and inside target cells.
An effective linker must provide both stability and controlled release. It should remain intact during synthesis and circulation but allow the oligonucleotide to be released when the conjugate reaches the target cell.
Key properties of an ideal linker
- Chemical stability during synthesis
- Stability during blood circulation
- Controlled release inside target cells
Common linker types
| Linker Type | Advantages | Limitations |
|---|---|---|
| Amide linkers | High stability | Limited cleavability |
| Disulfide linkers | Redox-responsive release | Premature cleavage possible |
| Triazole linkers | Highly stable click chemistry product | Non-cleavable |
| Ester linkers | Cleavable in biological environments | Lower stability |
Improper linker selection may lead to early drug release, loss of targeting capability, or reduced therapeutic activity. Because of this, linker optimization usually requires extensive experimental testing.
Dive deeper into the chemical bonds: Read about Peptide Oligonucleotide Conjugate Linker Chemistry
Challenges in Peptide Oligonucleotide Conjugates Development: Analytical Characterization
Analytical characterization is another critical component of addressing the Challenges in Peptide Oligonucleotide Conjugates development. These molecules are structurally complex and can exist as mixtures of closely related species. Accurate characterization is therefore essential for confirming structure, purity, and consistency.
Unlike small molecules, POCs require multi-dimensional analytical techniques. Scientists must verify both the peptide sequence and the oligonucleotide structure within the same conjugate molecule.
Major analytical challenges
- Confirming conjugation efficiency
- Detecting incomplete or side-product conjugates
- Measuring linker integrity
- Determining purity and structural identity
Common analytical methods
- LC-MS and high-resolution mass spectrometry
- Ion-pair reversed-phase chromatography
- Capillary electrophoresis
- Hybrid peptide-oligonucleotide sequencing techniques
Advanced mass spectrometry approaches are particularly valuable because they allow detailed structural verification and detection of subtle molecular variations.
Validate your conjugate’s integrity: Explore POC Synthesis and Characterization services
Challenges in Peptide Oligonucleotide Conjugates Development: Manufacturing and Scale-Up
Scaling up production is another major part of the Challenges in Peptide Oligonucleotide Conjugates development. Manufacturing requires combining two independent synthetic workflows—peptide synthesis and oligonucleotide synthesis—before performing the final conjugation step.
This multi-stage process introduces several operational and cost challenges.
Key manufacturing difficulties
- Low overall yields during conjugation
- Complex purification workflows
- Batch-to-batch variability
- High cost of goods
Large-scale production typically involves separate synthesis of each component followed by controlled conjugation and purification. Maintaining consistent quality across batches requires strict monitoring of every step in the process.
Purification is also difficult because:
- Product and impurities often have similar physicochemical properties
- Multiple charged species may be present
- Conjugation by-products can complicate separation
These factors increase both production cost and technical complexity.
Partner with a specialized research organization: Discover our Peptide Oligonucleotide Conjugates CRO capabilities
Challenges in Peptide Oligonucleotide Conjugates Development: Regulatory and CMC Considerations
Regulatory evaluation is another aspect of the Challenges in Peptide Oligonucleotide Conjugates development. Because these molecules combine features of biologics, peptides, and nucleic acid therapeutics, regulatory authorities may assess them under multiple frameworks.
Developers must provide extensive documentation to demonstrate safety, quality, and reproducibility.
Regulatory agencies typically require data on
- Conjugation chemistry
- Stability profiles
- Pharmacokinetics and biodistribution
- Immunogenicity risk
CMC documentation must include
- Detailed conjugation processes
- Impurity profiling
- Analytical validation
- Stability studies
Since peptide-oligonucleotide conjugates are still an emerging therapeutic class, regulatory expectations continue to evolve. Early planning of CMC strategies is therefore essential to avoid delays during clinical development.
Bridge the gap to clinical application: See our CMC Services for Peptide Oligonucleotide Conjugates
Conclusion
The Challenges in Peptide Oligonucleotide Conjugates development arise from the complexity of combining peptide chemistry with nucleic-acid therapeutics into a single drug candidate. These hybrid molecules must maintain structural stability, biological activity, and effective delivery while also meeting strict manufacturing and regulatory standards.
Key development barriers include
- Synthetic compatibility between peptide and oligonucleotide chemistries
- Stability and degradation in biological systems
- Efficient cellular delivery and endosomal escape
- Linker optimization
- Analytical characterization and quality control
- Manufacturing scale-up and regulatory requirements
Despite these obstacles, rapid advances in bioorthogonal chemistry, peptide engineering, and nucleic acid modification are improving the feasibility of POC therapeutics. As research progresses, these technologies may enable a new generation of highly targeted nucleic-acid medicines.
Organizations working in this field can benefit from specialized analytical expertise and early CMC planning to reduce development risks.
If your team is exploring peptide-oligonucleotide conjugates development and requires specialized analytical or CMC support, connecting with experts early in the process can significantly reduce development risks.
Ensure high-quality results for your conjugates: Learn about QC Testing for Peptide Oligonucleotide Conjugates
Contact our team to discuss your project: Contact us
Frequently Asked Questions (FAQs)
The main Challenges in Peptide Oligonucleotide Conjugates development include synthesis compatibility, biological stability, efficient cellular delivery, linker design, analytical characterization, and manufacturing scalability. Each of these factors can affect therapeutic performance and product quality. Addressing them requires coordinated work across chemistry, biology, and pharmaceutical development.
Peptide and oligonucleotide synthesis use very different chemical conditions. Some of the acids or bases used in one process may damage the other molecule. Scientists therefore need orthogonal reaction strategies that allow both structures to remain stable during conjugation.
Linkers determine how the peptide and oligonucleotide remain connected and how the drug behaves in the body. They influence stability, drug release, pharmacokinetics, and targeting ability. Choosing the right linker is critical for achieving the desired therapeutic performance.
Oligonucleotides have poor membrane permeability and often become trapped inside endosomes after entering the cell. This prevents them from reaching their target RNA or DNA. Delivery systems must therefore improve both cellular uptake and endosomal escape.
Cell-penetrating peptides help transport oligonucleotides into cells and improve intracellular delivery. However, they must be carefully optimized to avoid toxicity or unwanted uptake in non-target tissues. Proper design can greatly enhance therapeutic effectiveness.
Stability can be improved through backbone modifications, optimized linker chemistry, peptide cyclization, and other chemical modifications. These strategies help protect the molecule from enzymatic degradation. Improved stability often leads to longer circulation time and better therapeutic results.
Manufacturing involves separate synthesis workflows for peptides and oligonucleotides followed by complex conjugation and purification steps. Each stage requires strict quality control and advanced analytical verification. This multi-step process increases both time and production costs.
Reference:
- Venkatesan, N., & Kim, B. H. (2006). Peptide conjugates of oligonucleotides: Synthesis and applications. Chemical Reviews, 106(9), 3712–3761. https://doi.org/10.1021/cr0502448
- Naganuma, M., Tsuji, G., Amiya, M., Hirai, R., Higuchi, Y., Hata, N., Nozawa, S., Watanabe, D., Nakajima, T., & Demizu, Y. (2025). High-resolution HPLC for separating peptide–oligonucleotide conjugates. ACS Omega, 10(20), 20578–20584. https://doi.org/10.1021/acsomega.5c01308
- Tung, C. H., & Stein, S. (2000). Preparation and applications of peptide–oligonucleotide conjugates. Bioconjugate Chemistry, 11(5), 605–618. https://doi.org/10.1021/bc0000334



