
Introduction:
Peptide therapeutics are increasingly significant in modern medicine due to their specificity, potency, and safety profiles. For organizations looking to bring these innovative therapies to market, partnering with a peptide drug development CDMO is essential. A Contract Development and Manufacturing Organization (CDMO) like ResolveMass Laboratories Inc. offers comprehensive expertise from discovery to commercial-scale manufacturing, ensuring the highest quality standards and regulatory compliance.
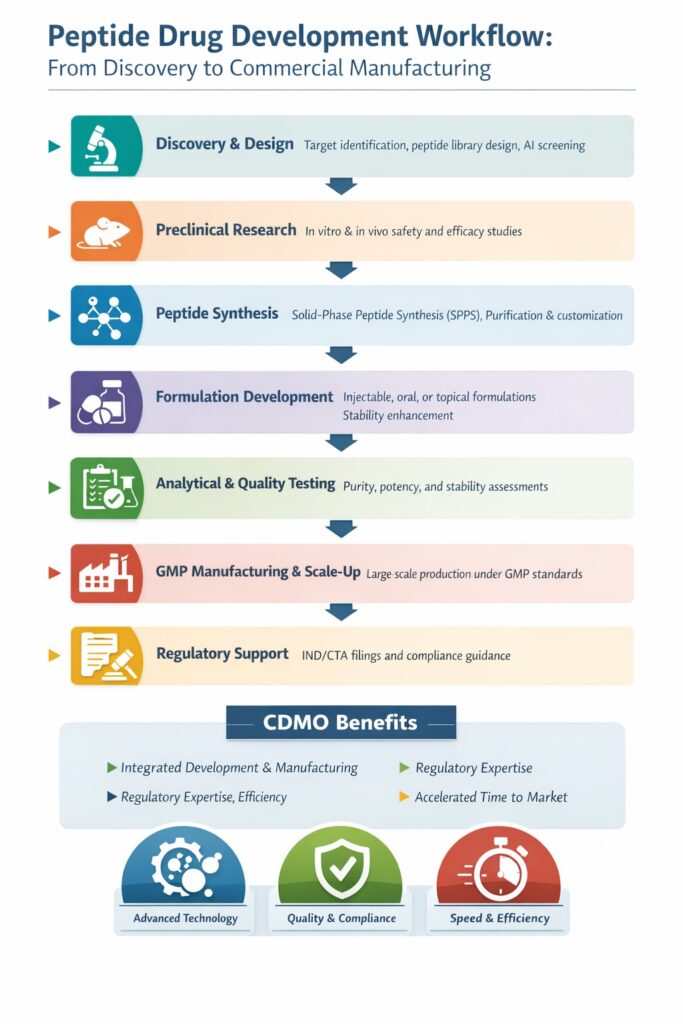
This article provides an in-depth overview of the peptide drug development workflow, helping biotech companies, pharmaceutical firms, and researchers understand the stepwise process involved in developing peptide therapeutics.
Share via:
Summary:
- Peptide drug development CDMO provides end-to-end services from early discovery to commercial manufacturing.
- The process involves discovery, preclinical research, peptide synthesis, formulation, analytical testing, and GMP manufacturing.
- CDMOs like ResolveMass Laboratories Inc. ensure quality, compliance, and scalability for peptide therapeutics.
- Choosing the right CDMO accelerates timelines, reduces costs, and ensures regulatory compliance.
- Advanced technologies and AI-driven processes improve efficiency in peptide design, synthesis, and quality control.
Watch the Full Peptide Drug Development Workflow
1. Discovery and Design of Peptides
How are peptide drug candidates discovered and designed?
Peptide drug discovery starts with identifying key proteins or pathways involved in a specific disease. Using advanced computational tools and AI-assisted screening, researchers can design peptides with high specificity, stability, and therapeutic potential—making peptide drug development CDMO services essential for efficient early-stage development.
Key Steps in Discovery:
- Target Identification: Understand disease mechanisms to select optimal peptide targets.
- Peptide Library Design: Generate diverse peptide sequences for experimental testing.
- In Silico Screening: Use AI-based models to predict peptide binding affinity, stability, and efficacy.
- Optimization: Apply modifications like cyclization or PEGylation to improve bioavailability and reduce immunogenicity.
Table: Discovery Phase Highlights
| Step | Purpose | Tools / Techniques |
|---|---|---|
| Target Identification | Identify disease-related proteins | Bioinformatics, Genomics |
| Peptide Library Design | Generate candidate peptides | Combinatorial chemistry, AI prediction |
| In Silico Screening | Predict efficacy & stability | Molecular modeling, AI algorithms |
| Optimization | Enhance therapeutic potential | Peptide modifications, cyclization |
ResolveMass Laboratories Inc. leverages proven computational modeling and laboratory expertise to ensure peptide candidates are designed with optimal drug-like properties, paving the way for successful preclinical development. For additional support in discovery-stage research, see Outsourced Chemistry in Drug Discovery.
2. Preclinical Research
What preclinical studies are required before moving to human trials?
Preclinical research evaluates the safety, efficacy, and pharmacokinetics of peptide candidates using in vitro (cell-based) and in vivo (animal) models. This stage is critical to ensure that the peptide is both effective and safe before initiating human clinical trials, making the role of a peptide drug development CDMO indispensable for thorough and compliant preclinical testing.
Key Focus Areas:
- Toxicology Studies: Assess safety at various dosage levels to prevent adverse effects.
- Pharmacokinetics (PK) and Pharmacodynamics (PD): Determine how the peptide is absorbed, distributed, metabolized, and how long it remains active in the body.
- Stability Testing: Evaluate peptide integrity under physiological conditions to ensure consistent therapeutic performance.
Why Partnering with a CDMO Matters:
- Access to standardized and validated protocols
- Regulatory-compliant preclinical testing
- Reduced risk of delays in drug development timelines
By collaborating with a peptide drug development CDMO like ResolveMass Laboratories Inc., biotech companies benefit from expertise, advanced facilities, and reliable preclinical evaluation, laying a strong foundation for successful clinical development.
For preclinical bioanalysis strategies, see Bioanalytical Strategy for Drug Development.
3. Peptide Synthesis
How are peptides synthesized for research and development?
Peptides are primarily synthesized using Solid-Phase Peptide Synthesis (SPPS), which enables precise assembly of amino acids into specific sequences. Partnering with a peptide CDMO ensures efficient, reproducible, and high-quality peptide production for both research and commercial purposes.
Important Aspects of Peptide Synthesis:
- Scalability: SPPS supports both small-scale synthesis for laboratory research and large-scale production for commercial needs.
- Purity: Advanced purification methods such as HPLC are used to achieve >95% peptide purity, ensuring therapeutic consistency.
- Customization: Chemical modifications like cyclization, PEGylation, or labeling can enhance stability, bioavailability, and efficacy.
Why Choose a CDMO:
- Access to state-of-the-art synthesis platforms
- Expertise from experienced peptide chemists
- Consistent, high-quality production aligned with regulatory standards
For peptide analytical and mass spectrometry services, see:
- Mass Spectrometry Experts in Drug Development
- Peptide Testing Service
- Peptide Characterization in Drug Development
ResolveMass Laboratories Inc. combines cutting-edge peptide synthesis technologies with expert personnel, delivering peptides that meet stringent quality requirements and accelerate the transition from discovery to clinical development.
4. Formulation Development
How are peptide drugs formulated for delivery?
Formulation development transforms raw peptides into stable, effective, and patient-friendly drug products. Collaborating with a peptide drug development CDMO ensures that peptides are optimized for safety, efficacy, and administration route.
Key Considerations in Peptide Formulation:
- Route of Administration: Formulations can be designed for injectable, oral, or topical delivery depending on therapeutic needs.
- Stabilizers: Special excipients and stabilizers prevent peptide degradation during storage and administration.
- Dosage Form Optimization: Peptides can be formulated as liquid solutions, lyophilized powders, or encapsulated products to enhance stability and convenience.
Benefits of Professional Peptide Formulation:
- Enhances shelf-life and long-term stability
- Improves bioavailability for better therapeutic effect
- Reduces immunogenic responses and adverse reactions
By leveraging the expertise of a peptide drug development CDMO like ResolveMass Laboratories Inc., companies can achieve precisely tailored formulations that maximize therapeutic potential and patient compliance.
5. Analytical Testing and Quality Control
How is quality ensured in peptide drug development?
Analytical testing validates peptide identity, purity, potency, and stability—critical for regulatory approval. Partnering with a peptide CDMO ensures robust analytical protocols.
Critical Tests Include:
- HPLC and Mass Spectrometry: Confirm peptide sequence and purity.
- Biological Assays: Evaluate activity against intended target.
- Stability Studies: Assess peptide integrity under different storage conditions.
Table: Key Analytical Methods
| Method | Purpose |
|---|---|
| HPLC | Purity analysis |
| Mass Spectrometry | Sequence confirmation |
| ELISA/Bioassays | Functional activity |
| Stability Studies | Shelf-life prediction |
For related analytical services, see:
ResolveMass Laboratories Inc. applies rigorous analytical protocols that comply with ICH and FDA standards, reinforcing trustworthiness in peptide development.
6. GMP Manufacturing and Scale-Up
How are peptide drugs manufactured at commercial scale?
GMP manufacturing ensures peptides are produced at high quality, reproducibly, and in compliance with regulatory standards. Partnering with a peptide CDMO enables seamless scale-up from laboratory quantities to commercial production.
Key Considerations in Peptide Formulation:
- Route of Administration: Formulations can be designed for injectable, oral, or topical delivery depending on therapeutic needs.
- Stabilizers: Special excipients and stabilizers prevent peptide degradation during storage and administration.
- Dosage Form Optimization: Peptides can be formulated as liquid solutions, lyophilized powders, or encapsulated products to enhance stability and convenience.
Benefits of Professional Peptide Formulation:
- Enhances shelf-life and long-term stability
- Improves bioavailability for better therapeutic effect
- Reduces immunogenic responses and adverse reactions
By leveraging the expertise of a peptide drug development CDMO like ResolveMass Laboratories Inc., companies can achieve precisely tailored formulations that maximize therapeutic potential and patient compliance.
7. Regulatory Support
How do CDMOs assist with regulatory approval?
Navigating regulatory pathways is essential for the successful development and commercialization of peptide therapeutics. A peptide drug development CDMO provides comprehensive support to ensure that all manufacturing, documentation, and submission processes meet the requirements of regulatory authorities such as the FDA, EMA, or Health Canada.
Key Activities in Regulatory Support:
- CMC (Chemistry, Manufacturing, and Controls) Documentation: Prepare detailed reports covering all aspects of peptide production and quality control.
- Validation of Manufacturing Processes: Ensure reproducibility and compliance with Good Manufacturing Practices (GMP).
- Regulatory Strategy Consultation: Guide clients through clinical trial applications (IND/CTA) and approval pathways.
For regulatory and generic drug support, see:
By partnering with ResolveMass Laboratories Inc., organizations gain access to deep scientific expertise and regulatory knowledge, facilitating smooth submissions, faster approvals, and reduced risk of compliance issues.
Conclusion:
Partnering with a peptide drug development CDMO like ResolveMass Laboratories Inc. ensures comprehensive support from discovery to commercial manufacturing. By integrating advanced AI-driven design, rigorous analytical testing, and GMP-compliant production, ResolveMass accelerates development timelines while maintaining the highest quality standards. For biotech companies and pharmaceutical innovators, this partnership translates to efficient, safe, and scalable peptide therapeutics.
Frequently Asked Questions:
A peptide drug development CDMO (Contract Development and Manufacturing Organization) provides end-to-end services for peptide therapeutics, including discovery, preclinical testing, synthesis, formulation, analytical testing, GMP manufacturing, and regulatory support. Companies partner with CDMOs like ResolveMass Laboratories Inc. to accelerate timelines, reduce costs, and ensure regulatory compliance.
Preclinical studies evaluate safety, efficacy, pharmacokinetics (PK), pharmacodynamics (PD), and stability using in vitro and in vivo models. Partnering with a peptide CDMO ensures standardized protocols, regulatory compliance, and reliable data for IND or CTA submissions. For bioanalytical support, see Bioanalytical Strategy for Drug Development.
Peptides are synthesized mainly via Solid-Phase Peptide Synthesis (SPPS), allowing precise amino acid assembly. High-purity peptides (>95%) are obtained using purification techniques like HPLC and mass spectrometry. For expert analytical characterization, see Peptide Characterization in Drug Development.
Formulation development converts raw peptides into stable, patient-friendly drug products, optimized for injectable, oral, or topical delivery. Professional CDMO formulation improves bioavailability, shelf-life, and reduces immunogenic responses. Learn more about peptide testing and formulation services.
GMP manufacturing ensures consistent, high-quality peptide production at commercial scale, adhering to regulatory standards. CDMOs handle process scale-up, sterile production, and documentation to maintain reproducibility.
A CDMO offers full development and manufacturing capabilities, whereas a CRO typically focuses on research and clinical trials. ResolveMass combines peptide synthesis, formulation, analytical testing, and GMP manufacturing under one roof, streamlining workflows and reducing timelines. Learn more: CDMO vs CRO for Generic Drug Development.
AI accelerates peptide design, in silico screening, and predictive stability analysis, helping identify optimal candidates faster and more accurately. CDMOs leverage AI for efficient discovery, testing, and quality assurance, enhancing success rates and reducing costs.
Reference
- Komal Sharma , Krishna K. Sharma , Anku Sharma , Rahul Jain. Peptide-based drug discovery: Current status and recent advances.https://www.sciencedirect.com/science/article/abs/pii/S1359644622004573
- Markus Muttenthaler, Glenn F. King, David J. Adams & Paul F. Alewood. Trends in peptide drug discovery.https://www.nature.com/articles/s41573-020-00135-8
- Michael W. Pennington , Brant Zell , Chris J. Bai. Commercial manufacturing of current good manufacturing practice peptides spanning the gamut from neoantigen to commercial large-scale products.https://www.sciencedirect.com/science/article/pii/S2590098620300580
- Laszlo Otvos. The latest trends in peptide drug discovery and future challenges.https://www.tandfonline.com/doi/full/10.1080/17460441.2024.2365969
- The Current State of Peptide Drug Discovery: Back to the Future?https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.7b00318


