
Introduction:
Peptide therapeutics are rapidly becoming one of the most promising areas in modern drug development. Pharmaceutical and biotechnology companies are increasingly relying on specialized partners to support complex peptide research, development, and analytical requirements. This is where ResolveMass Laboratories Inc., operating as a Peptide CDMO in United States, plays a critical role.
ResolveMass provides end-to-end peptide development services, helping companies move from early-stage discovery to clinical studies with confidence. With deep expertise in peptide analytical characterization services, mass spectrometry experts in drug development, and advanced bioanalysis, the company delivers high-quality data and scientific insight required for regulatory submissions and successful drug development.
Companies developing peptide therapeutics often rely on a strong bioanalytical strategy for drug development to ensure that clinical and preclinical data meet regulatory expectations.
This article explores how ResolveMass supports the peptide drug development journey through its comprehensive peptide CDMO services in the United States.
Share via:
Summary:
- ResolveMass Laboratories Inc. provides comprehensive end-to-end peptide development services for pharmaceutical and biotech companies.
- As a Peptide CDMO in United States, the company supports projects from early research to clinical development and commercialization.
- Services include peptide synthesis, analytical characterization, bioanalysis, method development, and regulatory support.
- Advanced technologies such as mass spectrometry-based peptide analysis ensure accurate, high-quality data for drug development.
- ResolveMass focuses on scientific expertise, quality, regulatory compliance, and collaborative partnerships to accelerate peptide therapeutics development.
1: Why Choose ResolveMass as a Peptide CDMO in United States?
ResolveMass Laboratories Inc. stands out as a trusted Peptide CDMO in United States by providing specialized peptide analytical expertise, advanced technologies, and collaborative development support for pharmaceutical and biotech companies. The company focuses on delivering reliable scientific data and customized analytical solutions that accelerate peptide drug development.
The company focuses on delivering reliable scientific data and customized analytical solutions that accelerate peptide drug development. Many organizations evaluating peptide CDMO vs CRO models choose ResolveMass because of its specialized analytical capabilities and deep scientific expertise.
ResolveMass also supports complex projects involving peptide characterization in drug development, helping companies ensure structural accuracy and regulatory readiness.
As a dedicated Peptide CDMO in United States, ResolveMass offers comprehensive capabilities tailored specifically for peptide therapeutics. From early research support to complex analytical characterization and bioanalysis, the organization provides the expertise and infrastructure required to meet modern drug development challenges.
Key Advantages of Choosing ResolveMass
- Expert Peptide Analytical Scientists
A team of experienced scientists with deep knowledge in peptide chemistry, bioanalysis, and analytical characterization. - Advanced Mass Spectrometry Platforms
State-of-the-art LC-MS and high-resolution mass spectrometry systems for precise peptide identification, quantification, and structural analysis. - Custom Analytical Method Development
Tailored analytical methods designed specifically for peptide molecules and complex biological matrices. - Regulatory-Ready Bioanalytical Data
High-quality analytical data generated following regulatory expectations to support clinical development and regulatory submissions. - Flexible Collaboration Models
ResolveMass works with biotech startups, pharmaceutical companies, and research organizations through flexible project structures that support different development stages.
ResolveMass works closely with clients to design customized analytical workflows that align with development timelines, regulatory requirements, and scientific objectives. This collaborative approach makes ResolveMass a reliable Peptide CDMO in United States for organizations seeking expert support in peptide therapeutics development.
2: End-to-End Peptide Development Services Offered by ResolveMass as the Peptide CDMO in United States
ResolveMass Laboratories Inc. provides comprehensive end-to-end peptide development services that support pharmaceutical and biotech companies from early discovery to clinical development. As a specialized Peptide CDMO in United States, ResolveMass delivers integrated analytical and bioanalytical solutions designed specifically for peptide therapeutics.
As a specialized Peptide CDMO in United States, ResolveMass delivers integrated analytical and bioanalytical solutions designed specifically for peptide therapeutics. These services often include peptide testing services and advanced analytical characterization.
Companies working on complex biologics or generics frequently rely on expert analytical method development for generic drugs to ensure reliable analytical methods throughout the drug development lifecycle.
ResolveMass also provides expertise in outsourced chemistry in drug discovery, supporting pharmaceutical companies that need specialized analytical capabilities without expanding internal laboratories.
By combining deep scientific expertise with advanced analytical technologies, ResolveMass helps clients move efficiently through each stage of peptide drug development while ensuring data quality, regulatory compliance, and project scalability.
Overview of Peptide Development Services
| Development Stage | ResolveMass Services |
|---|---|
| Discovery | Peptide characterization, structural analysis, and analytical feasibility studies |
| Preclinical Development | Analytical method development, peptide bioanalysis, stability studies |
| Clinical Development | Pharmacokinetics (PK) and pharmacodynamics (PD) bioanalysis, biomarker analysis |
| Regulatory Support | Preparation of analytical data packages and documentation for regulatory submissions |
Integrated Support Across the Peptide Development Lifecycle
ResolveMass provides specialized services tailored to the complex nature of peptide therapeutics. As a trusted Peptide CDMO in United States, the company focuses on generating accurate analytical data that supports critical decision-making during drug development.
Key integrated capabilities include:
- Advanced peptide analytical characterization
- Bioanalytical assay development and validation
- Mass spectrometry-based peptide quantification
- PK/PD analysis for clinical studies
- Regulatory-compliant analytical documentation
This comprehensive approach allows pharmaceutical and biotechnology companies to partner with a single expert provider for their peptide development needs. By supporting every phase of the development lifecycle, ResolveMass strengthens its position as a reliable Peptide CDMO in United States capable of supporting both early-stage research programs and long-term clinical development projects.
3: Peptide Analytical Characterization Services
Peptide analytical characterization ensures that peptide drug candidates are accurately identified, structurally verified, and evaluated for purity and stability before entering clinical development. As an experienced Peptide CDMO in United States, ResolveMass Laboratories Inc. provides advanced analytical characterization services that help pharmaceutical and biotechnology companies gain a complete understanding of their peptide molecules.
ResolveMass provides advanced peptide analytical characterization services that help pharmaceutical and biotechnology companies gain a complete understanding of their peptide molecules.
Peptides are complex biomolecules that require highly sensitive and precise analytical techniques for proper evaluation. ResolveMass uses state-of-the-art instrumentation and deep scientific expertise to generate reliable analytical data that supports peptide development programs.
Advanced Characterization Capabilities
As a trusted Peptide CDMO in United States, ResolveMass offers a wide range of analytical techniques designed specifically for peptide therapeutics.
Key Characterization Techniques include:
- LC-MS Peptide Analysis
Liquid chromatography–mass spectrometry (LC-MS) is used to separate and identify peptide components, enabling accurate molecular weight determination and impurity detection. - High-Resolution Mass Spectrometry
High-resolution mass spectrometry provides precise mass measurement, helping scientists confirm peptide structures and detect minor modifications. - Peptide Impurity Profiling
Comprehensive impurity profiling helps identify synthesis-related impurities, degradation products, and other potential contaminants. - Sequence Confirmation
Advanced analytical techniques confirm the amino acid sequence of peptide molecules, ensuring structural accuracy. - Structural Verification
Detailed structural characterization verifies peptide integrity and supports regulatory documentation.
These services are particularly critical for complex generics and sameness studies, such as those described in generic peptide drug analytical characterization case study and peptide sameness study services in Canada.
ResolveMass has also performed advanced analytical work in projects such as peptide characterization of Ganirelix generic project.
Importance of Peptide Characterization in Drug Development
Accurate peptide characterization is essential for ensuring that drug candidates meet quality, safety, and regulatory requirements. By partnering with an experienced Peptide CDMO in United States, pharmaceutical companies can ensure that their peptide therapeutics are thoroughly evaluated before progressing into clinical development.
ResolveMass combines advanced instrumentation with experienced scientists to deliver high-confidence analytical data, helping clients accelerate peptide drug development while maintaining strict quality standards.
4: Bioanalytical Services for Peptide Therapeutics
Bioanalytical testing is essential for understanding how peptide drugs behave in biological systems, including how they are absorbed, distributed, metabolized, and eliminated. As a specialized Peptide CDMO in United States, ResolveMass Laboratories Inc. offers advanced bioanalytical services that support the evaluation of peptide therapeutics during preclinical and clinical development.
ResolveMass provides specialized bioanalysis in Canada and international bioanalytical support for peptide therapeutics development. These services include pharmacokinetics (PK), pharmacodynamics (PD), biomarker analysis, and LC-MS/MS bioanalysis.
These capabilities are supported by advanced mass spectrometry experts in drug development who ensure accurate peptide detection and quantification.
ResolveMass utilizes highly sensitive analytical platforms and scientifically validated workflows to generate accurate data for pharmacokinetic (PK), pharmacodynamic (PD), and biomarker studies. These insights help pharmaceutical and biotechnology companies better understand the biological performance of peptide-based drugs.
Bioanalytical Capabilities
As an experienced Peptide CDMO in United States, ResolveMass provides a range of bioanalytical services designed to support peptide drug development programs.
Key Bioanalytical Services include:
- Peptide Quantification in Biological Matrices
Accurate measurement of peptide concentrations in biological samples such as plasma, serum, and tissues. - Pharmacokinetics (PK) and Pharmacodynamics (PD) Analysis
Evaluation of how peptide drugs move through and affect biological systems, helping determine dosage and therapeutic effectiveness. - Biomarker Analysis
Identification and quantification of biological markers that indicate drug response or therapeutic activity. - Ligand Binding Assays
Sensitive assays used to measure interactions between peptides and biological targets. - LC-MS/MS Bioanalysis
Advanced liquid chromatography–tandem mass spectrometry methods for precise peptide detection and quantification.
Supporting Clinical Development and Regulatory Requirements
High-quality bioanalytical data plays a critical role in supporting regulatory submissions and clinical trials. By partnering with an experienced Peptide CDMO in United States, pharmaceutical companies can ensure that their peptide therapeutics are evaluated using scientifically robust and regulatory-compliant analytical methods.
ResolveMass combines advanced mass spectrometry technologies, rigorous method validation, and experienced scientific teams to deliver reliable bioanalytical data that helps accelerate peptide drug development.
5: Advanced Mass Spectrometry Expertise
Mass spectrometry is one of the most powerful analytical tools used for accurate peptide identification, characterization, and quantification in drug development. As a specialized Peptide CDMO in United States, ResolveMass Laboratories Inc. leverages advanced mass spectrometry technologies to deliver highly precise and reliable analytical data for peptide therapeutics.
ResolveMass leverages the expertise of mass spectrometry experts in drug development to provide highly sensitive analytical data that supports peptide therapeutics programs.
ResolveMass combines cutting-edge instrumentation with deep scientific expertise to support complex peptide analysis across different stages of drug development. These capabilities enable pharmaceutical and biotechnology companies to gain critical insights into peptide structure, purity, stability, and biological behavior.
Advantages of Mass Spectrometry in Peptide Development
Mass spectrometry plays a crucial role in peptide drug development by enabling detailed molecular analysis. As an experienced Peptide CDMO in United States, ResolveMass uses these technologies to ensure accurate and reproducible results.
Key advantages include:
- High Sensitivity Detection
Mass spectrometry can detect peptide molecules at very low concentrations, making it ideal for biological sample analysis. - Accurate Peptide Quantification
Advanced LC-MS/MS techniques provide precise quantification of peptides in complex biological matrices. - Structural Characterization
High-resolution mass spectrometry helps confirm peptide sequence, structural integrity, and molecular modifications. - Detection of Impurities and Degradation Products
Analytical methods can identify impurities, degradation pathways, and synthesis-related by-products.
Supporting Reliable Peptide Drug Development
The scientific team at ResolveMass applies rigorous analytical workflows to ensure that all mass spectrometry results are accurate, reproducible, and compliant with regulatory expectations. This expertise strengthens ResolveMass’s role as a trusted Peptide CDMO in United States, helping pharmaceutical companies generate high-quality analytical data necessary for successful peptide drug development programs.
6: Analytical Method Development and Validation
Accurate and reliable analytical methods are essential for measuring peptide drugs throughout the drug development lifecycle. As a specialized Peptide CDMO in United States, ResolveMass Laboratories Inc. provides customized analytical method development and validation services tailored specifically for peptide therapeutics.
ResolveMass provides customized analytical method development for generic drugs and peptide therapeutics.
Method development includes LC-MS method design, peptide quantification assays, biomarker detection methods, and stability-indicating assays.
For companies developing complex generic drugs, ResolveMass also supports reverse engineering for development of generic drugs and advanced analytical characterization.
These capabilities are often integrated into broader analytical development for generic drugs in Canada programs.
Peptide molecules often require highly sensitive and selective analytical approaches due to their structural complexity and the challenges of analyzing them in biological matrices. ResolveMass develops robust analytical methods that ensure precise quantification, structural verification, and stability evaluation of peptide drugs.
Method Development Services
ResolveMass designs analytical methods that meet the unique requirements of peptide drug development. As a trusted Peptide CDMO in United States, the company focuses on building methods that are sensitive, reproducible, and suitable for regulatory use.
Key method development capabilities include:
- LC-MS Method Development
Development of advanced liquid chromatography–mass spectrometry methods for peptide identification and quantification. - Peptide Quantification Methods
Sensitive analytical techniques to measure peptide concentrations accurately in various sample types. - Biomarker Detection Methods
Development of assays to detect and quantify biomarkers related to peptide drug activity and therapeutic response. - Stability-Indicating Methods
Analytical approaches that detect peptide degradation products and assess drug stability over time.
Method Validation
After development, analytical methods must be validated to ensure they consistently produce accurate and reliable results. ResolveMass performs comprehensive validation studies in accordance with regulatory expectations.
Validation activities include:
- Accuracy and Precision Testing
Ensuring the method produces consistent and reliable results across multiple analyses. - Sensitivity and Specificity Validation
Confirming the method can detect the peptide at required concentrations without interference. - Regulatory Guideline Compliance
Validation processes aligned with international regulatory expectations for pharmaceutical development. - Method Transfer Support
Assistance with transferring validated analytical methods to other laboratories or manufacturing partners when required.
Through rigorous development and validation processes, ResolveMass ensures that analytical methods are fully suitable for clinical studies, regulatory submissions, and long-term drug development programs. This capability further strengthens its reputation as a dependable Peptide CDMO in United States supporting peptide therapeutics development.
7: Supporting Pharmaceutical and Biotech Companies
ResolveMass Laboratories Inc. collaborates closely with pharmaceutical and biotechnology organizations to support peptide drug development through specialized analytical and bioanalytical expertise. As a trusted Peptide CDMO in United States, ResolveMass acts as a scientific partner, helping clients navigate complex analytical challenges while advancing their peptide therapeutics programs.
The company provides flexible collaboration models that allow clients to receive tailored support based on the stage of their drug development program. Whether a project requires early feasibility studies, analytical method development, or clinical bioanalysis, ResolveMass works with clients to create customized solutions that align with project goals and timelines.
Types of Clients Supported
As an experienced Peptide CDMO in United States, ResolveMass supports a wide range of organizations involved in peptide drug research and development.
These include:
- Early-Stage Biotech Startups
Providing analytical expertise and scientific guidance to help emerging companies advance peptide candidates from discovery to preclinical development. - Mid-Size Pharmaceutical Companies
Supporting drug development programs with specialized analytical services and scalable bioanalytical capabilities. - Global Pharmaceutical Organizations
Offering advanced analytical techniques and collaborative project management for complex peptide development programs. - Academic Research Groups
Assisting research institutions with peptide characterization, bioanalysis, and analytical method development for translational research projects.
Many organizations seeking pharmaceutical CDMO in US and Canada rely on ResolveMass for analytical development support.
The company also supports generic drug developers through specialized services such as:
- CDMO for generic drug development in Canada
- CDMO for generic projects in Canada
- generic pharmaceutical CDMO Canada
Many organizations also rely on outsourcing generic drug development Canada to reduce development timelines and improve efficiency.
A Collaborative Scientific Partnership
ResolveMass emphasizes scientific collaboration, transparency, and clear communication throughout every project. By working closely with clients and understanding their development objectives, the company helps streamline analytical workflows and generate reliable data that supports critical decision-making.
This collaborative approach strengthens ResolveMass’s position as a dependable Peptide CDMO in United States, enabling pharmaceutical and biotechnology companies to accelerate their peptide development programs with confidence.
8: Quality and Regulatory Compliance
Regulatory compliance is essential for successful peptide drug development and clinical approval. As a trusted Peptide CDMO in United States, ResolveMass Laboratories Inc. maintains strict quality standards to ensure that all analytical and bioanalytical services meet global regulatory expectations.
ResolveMass places strong emphasis on data integrity, transparent documentation, and rigorous quality control procedures. These practices help pharmaceutical and biotechnology companies generate reliable analytical data that supports regulatory submissions and clinical development programs.
The company also provides regulatory support for generic drugs US and Canada CDMO to help pharmaceutical companies prepare regulatory submissions and clinical documentation.
Quality Framework
As an experienced Peptide CDMO in United States, ResolveMass operates under a structured quality framework designed to maintain accuracy, consistency, and regulatory readiness across all projects.
Key elements of the quality framework include:
- Good Laboratory Practice (GLP) Principles
Implementation of internationally recognized laboratory practices to ensure consistency, traceability, and reliability of analytical data. - Regulatory-Compliant Analytical Methods
Development and validation of analytical methods that align with regulatory guidelines for pharmaceutical development. - Comprehensive Documentation for Regulatory Submissions
Detailed analytical reports and documentation prepared to support regulatory filings and clinical trial requirements. - Robust Quality Management Systems
Structured quality processes that oversee laboratory operations, data management, and continuous improvement initiatives.
Ensuring Trust and Data Reliability
By maintaining rigorous quality and compliance standards, ResolveMass ensures that clients receive high-quality, scientifically robust, and regulatory-ready analytical data. This commitment to quality strengthens ResolveMass’s reputation as a dependable Peptide CDMO in United States, supporting pharmaceutical and biotechnology companies in advancing peptide therapeutics toward successful clinical development and regulatory approval.
9: Benefits of Working with a Specialized Peptide CDMO in United States
Partnering with a specialized Peptide CDMO in United States allows pharmaceutical and biotechnology companies to access advanced scientific expertise, modern analytical technologies, and regulatory-ready development support. Organizations developing peptide therapeutics often face complex analytical and regulatory challenges, which makes collaboration with an experienced CDMO highly valuable.
ResolveMass Laboratories Inc., as a dedicated Peptide CDMO in United States, provides integrated analytical and bioanalytical services that help streamline peptide drug development while maintaining high quality and regulatory compliance.
ResolveMass helps pharmaceutical companies accelerate generic drug development in US and Canada by providing high-quality analytical and bioanalytical services.
Companies evaluating outsourcing models often compare CDMO vs CRO for generic drug development when selecting development partners.
Key Benefits
Working with a specialized Peptide CDMO in United States offers several strategic advantages for companies developing peptide-based drugs.
- Access to Peptide Analytical Experts
Dedicated scientists with deep expertise in peptide chemistry, bioanalysis, and advanced analytical techniques. - Advanced Analytical Instrumentation
Access to modern technologies such as LC-MS/MS and high-resolution mass spectrometry for precise peptide characterization and quantification. - Faster Development Timelines
Specialized expertise and established workflows help accelerate analytical development and reduce delays in drug development programs. - Regulatory-Compliant Data Generation
Analytical data produced according to regulatory expectations to support clinical trials and regulatory submissions. - Reduced Operational Complexity
Outsourcing analytical development allows pharmaceutical companies to focus on core research while the CDMO manages complex analytical processes.
Accelerating Peptide Drug Development
For companies developing peptide therapeutics, partnering with an experienced Peptide CDMO in United States can significantly improve development efficiency. By leveraging the scientific expertise, analytical infrastructure, and regulatory knowledge of ResolveMass, pharmaceutical and biotech organizations can accelerate drug development timelines while ensuring data quality and regulatory readiness.
10: The Future of Peptide Drug Development
Peptide therapeutics are emerging as one of the fastest-growing areas in modern pharmaceutical research due to their high specificity, strong therapeutic potential, and improved safety profiles. As the global demand for innovative peptide-based drugs increases, the role of specialized partners such as ResolveMass as a Peptide CDMO in United States will become even more critical in supporting drug development programs.
Peptides offer unique advantages compared to traditional small-molecule drugs and biologics. Their ability to target specific biological pathways makes them highly effective for treating complex diseases while minimizing unwanted side effects. Because of these benefits, pharmaceutical and biotechnology companies are investing heavily in peptide research and development.
As innovation continues, pharmaceutical companies increasingly partner with specialized providers such as best peptide CDMO organizations to support complex peptide research and development.
ResolveMass provides advanced peptide drug development CDMO services that support pharmaceutical companies throughout the development lifecycle.
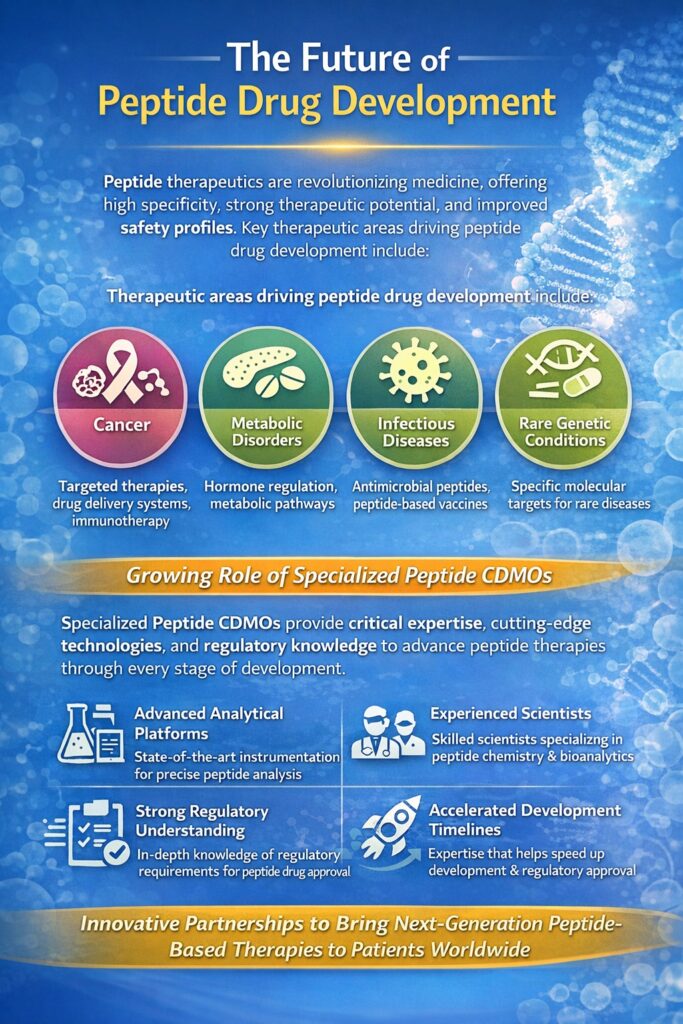
Therapeutic Areas Driving Peptide Drug Development
Peptide-based therapeutics are increasingly being explored for the treatment of several major disease areas, including:
- Cancer
Peptides are being used in targeted therapies, drug delivery systems, and immunotherapy approaches for oncology treatments. - Metabolic Disorders
Many peptide drugs are designed to regulate hormones and metabolic pathways involved in diseases such as diabetes and obesity. - Infectious Diseases
Antimicrobial peptides and peptide-based vaccines are gaining attention for their potential to combat resistant pathogens. - Rare Genetic Conditions
Peptide therapeutics can help address specific molecular targets involved in rare and orphan diseases.
Growing Role of Specialized Peptide CDMOs
As peptide drug development becomes more sophisticated, pharmaceutical companies increasingly rely on specialized service providers to manage complex analytical and bioanalytical requirements. A dedicated Peptide CDMO in United States like ResolveMass provides the expertise, technologies, and regulatory knowledge necessary to support peptide development programs from discovery through clinical trials.
With advanced analytical platforms, experienced scientists, and strong regulatory understanding, ResolveMass helps companies overcome scientific challenges while accelerating development timelines. As innovation in peptide therapeutics continues to expand, partnerships with expert organizations like ResolveMass will play a key role in bringing next-generation peptide drugs to patients worldwide.
Conclusion:
ResolveMass Laboratories Inc. provides comprehensive end-to-end peptide development services, supporting pharmaceutical and biotech companies throughout the drug development lifecycle. With expertise in peptide analytical characterization, bioanalysis, mass spectrometry, and method development, the company has established itself as a reliable Peptide CDMO in United States.
With expertise in peptide analytical characterization services, peptide testing services, bioanalysis, and mass spectrometry, the company has established itself as a reliable Peptide CDMO in United States.
Organizations looking for a trusted Peptide CDMO in United States can rely on ResolveMass to deliver the expertise and analytical capabilities needed for successful peptide therapeutics development.
Frequently Asked Questions:
A Peptide CDMO in United States is a Contract Development and Manufacturing Organization that provides specialized services for peptide drug development. These services may include peptide synthesis, analytical characterization, bioanalysis, method development, and regulatory support to help pharmaceutical and biotech companies advance peptide therapeutics from discovery to clinical development.
A Peptide CDMO typically offers end-to-end development services including:
-Peptide synthesis and purification
-Peptide analytical characterization
-Bioanalytical testing and PK/PD analysis
-Mass spectrometry analysis
-Analytical method development and validation
-Regulatory documentation and support
These services help pharmaceutical companies accelerate peptide drug development.
Pharmaceutical and biotechnology companies often outsource peptide development to a CDMO to access specialized expertise, advanced analytical technologies, and regulatory experience. Outsourcing helps reduce development timelines, optimize resources, and ensure high-quality analytical data required for clinical studies and regulatory submissions.
Peptide analytical characterization is essential for confirming the identity, purity, sequence, and structural integrity of peptide drug candidates. Accurate characterization ensures that peptide therapeutics meet quality and regulatory requirements before entering preclinical or clinical studies.
Mass spectrometry is widely used in peptide drug development for accurate molecular identification, structural verification, impurity detection, and peptide quantification. Advanced LC-MS and high-resolution mass spectrometry techniques provide highly sensitive and reliable analytical data.
Bioanalytical services evaluate how peptide drugs behave in biological systems. These services include pharmacokinetic (PK) analysis, pharmacodynamic (PD) studies, biomarker analysis, and peptide quantification in biological samples such as plasma and tissues.
Working with a specialized Peptide CDMO in United States offers several advantages:
-Access to peptide analytical experts
-Advanced mass spectrometry technologies
-Regulatory-ready analytical data
-Faster development timelines
-Flexible collaboration models for biotech and pharma companies
Reference
- Alex Philippidis. Top 10 Contract Development and Manufacturing Organizations: Most leading CDMOs are reporting revenue growth, but according to the CEO of one of these companies, near-term challenges include declining drug discovery activity.https://journals.sagepub.com/doi/full/10.1089/gen.44.09.06
- The Net Financial Benefits of Single Vendor Integrated CDMO and CRO Drug Development Services.https://www.researchsquare.com/article/rs-6531199/v1
- Catherine Shaffer. Making the Most of a CDMO Relationship: CDMOs and their pharma and biopharma clients could benefit from a little relationship counseling—just enough to clarify key issues like scope of services.https://journals.sagepub.com/doi/full/10.1089/gen.40.04.13


