
Introduction:
E&L Sample Preparation Best Practices play a crucial role in ensuring reliable results in extractables and leachables testing. Even the most advanced LC-MS or GC-MS system cannot produce accurate results if the sample is not prepared correctly.
In regulatory environments, sample preparation is not just a technical step — it is a scientific process that directly impacts regulatory acceptance, data integrity, and patient safety. Companies preparing for global submissions often also follow the latest regulatory frameworks such as the ICH Q3E guideline for Extractables and Leachables and global expectations described in Extractables and Leachables (E&L) Testing in the United States.
Share via:
Summary:
- Sample preparation is the most critical step in extractables and leachables (E&L) studies because poor preparation can invalidate the entire dataset.
- Following E&L Sample Preparation Best Practices ensures accurate identification of leachables, regulatory acceptance, and reproducible results.
- Proper selection of extraction solvents, containers, filtration techniques, and storage conditions significantly improves analytical reliability.
- Regulatory-focused laboratories such as ResolveMass Laboratories Inc. use validated, scientifically controlled sample preparation protocols to ensure submission-ready data.
- This article provides practical, real-world strategies used by experienced analytical laboratories to improve E&L study outcomes.
1: What Are E&L Sample Preparation Best Practices?
E&L Sample Preparation Best Practices are scientifically validated procedures used to prepare pharmaceutical products, packaging materials, and medical device components before extractables and leachables testing. Proper preparation ensures that analytical results accurately reflect the true chemical profile of the product and meet regulatory expectations.
These practices become even more important when companies are preparing studies aligned with the ICH Q3E Extractables and Leachables (E&L) study requirements or performing a full ICH Q3E Extractables & Leachables risk assessment.
In simple terms, these best practices ensure that:
- The sample truly represents the final marketed product
- No contamination is introduced during preparation
- No analyte is lost before analytical testing
- Results remain reproducible, reliable, and regulatory-acceptable
These procedures are followed by experienced analytical laboratories such as ResolveMass Laboratories Inc., where the focus is not only on performing the analysis but also on generating scientifically robust, submission-ready data.
If you want to understand the financial impact of incorrect preparation, this guide on cost of extractables and leachables testing explains why re-testing significantly increases project cost.
2: Why Sample Preparation Is Critical in E&L Testing
Sample preparation is the most important step in extractables and leachables (E&L) testing because it determines whether the final analytical results truly represent the chemical profile of the product. Even highly advanced analytical instruments cannot produce reliable data if the sample is not prepared correctly.
This becomes even more critical when studies are prepared for global markets such as the U.S. and Europe, where expectations differ. You can understand the differences here: E&L testing USA vs Europe regulatory expectations.
If sample preparation is done incorrectly, the following problems can occur:
- False positive impurity results caused by contamination
- Missing leachables in the final analytical report
- Poor reproducibility between batches or studies
- Higher risk of regulatory rejection during submission
- Time-consuming and costly re-testing
This is exactly why E&L Sample Preparation Best Practices are considered the foundation of every successful extractables and leachables study. Experienced laboratories such as ResolveMass Laboratories Inc. focus heavily on scientifically controlled sample preparation to ensure accurate, reproducible, and submission-ready analytical results.
3: Step-by-Step E&L Sample Preparation Best Practices
E&L Sample Preparation Best Practices follow a structured scientific approach used by regulatory-focused analytical laboratories. Each step directly affects the accuracy, reproducibility, and regulatory acceptance of the final analytical results.
This structured approach is also followed in modern Extractables and Leachables testing services for ICH Q3E compliance.
Below is a practical step-by-step workflow used in experienced laboratories such as ResolveMass Laboratories Inc.
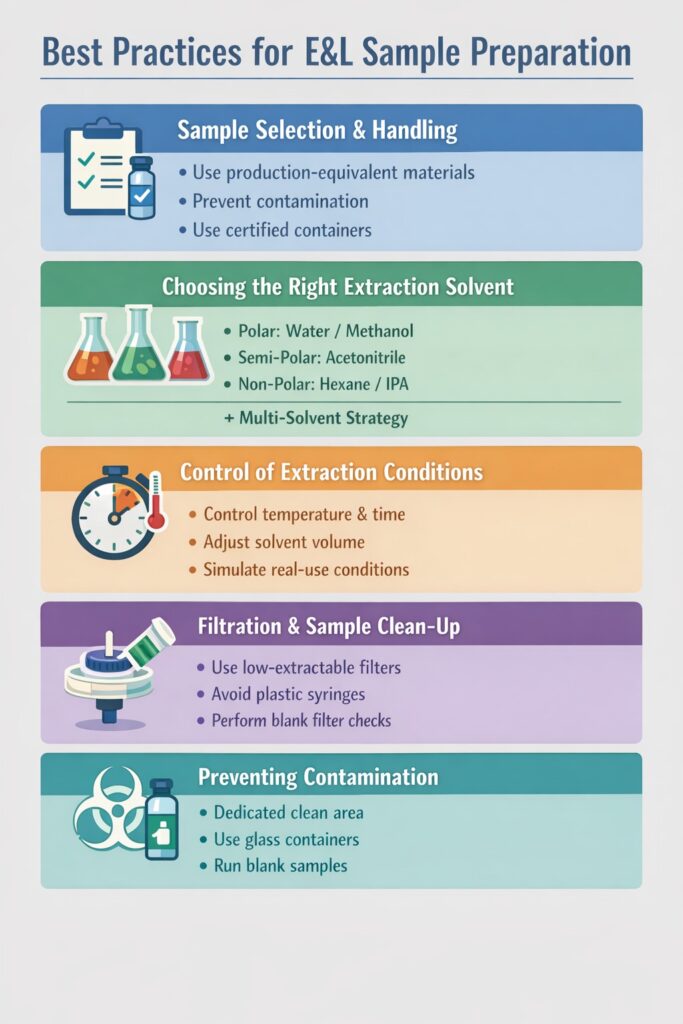
1. Sample Selection and Handling
The first and most critical step in E&L Sample Preparation Best Practices is proper sample selection. The sample must accurately represent the real product and its actual usage conditions.
This is especially important for complex products such as biologics and advanced therapies. If you are working in that space, this guide on Extractables and leachables in biologics and biopharma products explains why sample preparation becomes more complex.
Best practices include:
- Using production-equivalent materials instead of laboratory substitutes
- Ensuring the sample reflects the final marketed product
- Preventing contamination during handling
- Using clean glassware and certified containers
- Maintaining proper labeling and documentation
When this step is performed correctly, the reliability of the entire analytical study improves significantly.
2. Choosing the Right Extraction Solvent
The extraction solvent determines which extractables or leachables will be detected. Selecting the wrong solvent may result in missing critical impurities.
This is particularly important in studies designed for regulatory submissions such as extractables and leachables testing for NDA and ANDA submissions.
To follow E&L Sample Preparation Best Practices, laboratories typically select solvents based on compound polarity and extraction objectives.
Solvent Selection Guide
| Objective | Recommended Solvent Type |
|---|---|
| Polar compounds | Water / Methanol |
| Semi-polar compounds | Acetonitrile |
| Non-polar compounds | Hexane / Isopropanol (IPA) |
| Comprehensive extraction | Multi-solvent extraction strategy |
Using multiple solvents significantly increases the chances of detecting all potential leachables.
3. Control of Extraction Conditions
Extraction conditions must closely simulate real-world product storage and usage conditions. Poorly controlled extraction parameters can create artificial degradation products and misleading analytical data.
This is also critical when performing extractables and leachables requirements for U.S. market authorization, where regulators closely evaluate study design.
E&L Sample Preparation Best Practices recommend controlling:
- Extraction temperature
- Extraction time
- Surface area of materials
- Solvent-to-sample ratio
- Agitation or shaking conditions
For example, excessive heat during extraction can generate degradation compounds that do not actually exist in the final product.
4. Filtration and Sample Clean-Up
Filtration is one of the most sensitive steps in E&L Sample Preparation Best Practices, especially when testing trace-level leachables.
Laboratories performing extractables and leachables testing in the United States typically validate filtration materials before routine testing to avoid false results.
Best practices include:
- Using low-extractable validated filters
- Avoiding plastic syringes when analyzing trace compounds
- Verifying filtration materials before routine use
- Performing blank filter checks
- Using high-purity solvents during sample clean-up
This approach is commonly followed by regulatory-focused laboratories such as ResolveMass Laboratories Inc. to prevent contamination during sample preparation.
5. Preventing Contamination During Preparation
Contamination during preparation is one of the most common reasons E&L studies fail or require re-testing. Even small contamination can lead to false impurity identification.
To maintain E&L Sample Preparation Best Practices, laboratories follow strict contamination-control procedures:
- Using dedicated clean preparation areas
- Preferably using glass containers instead of plastic
- Avoiding perfumes, sanitizers, and volatile laboratory chemicals
- Running blank samples before actual testing
- Storing prepared samples under controlled conditions
This is also a major concern in high-risk products such as inhalation drug products. You can read more here: E&L testing for inhalation and nasal drug products.
These small but critical steps significantly improve analytical accuracy, reproducibility, and regulatory acceptance.
4: Common Mistakes in E&L Sample Preparation
Understanding what not to do is just as important as understanding the correct procedure. Many extractables and leachables studies fail not because of analytical limitations, but because of avoidable mistakes during sample preparation.
Below are the most common errors observed in real regulatory studies handled by experienced laboratories such as ResolveMass Laboratories Inc..
Most Common Sample Preparation Mistakes
- Using unverified or incompatible extraction solvents
- Ignoring blank samples before starting analysis
- Poor storage conditions after sample preparation
- Over-heating samples during extraction
- Using non-validated filtration materials
- Improper labeling of extracted samples
- Contamination caused by improper handling
- Using laboratory-grade materials instead of production-equivalent samples
Even small mistakes at the preparation stage can lead to incorrect impurity identification, missing leachables, or non-reproducible analytical results.
These mistakes become extremely expensive when studies must be repeated. This article on E&L testing cost and project timelines explains how incorrect preparation increases overall testing cost.
This is exactly why E&L Sample Preparation Best Practices are considered essential for generating reliable, reproducible, and submission-ready data. When these best practices are followed correctly, companies can avoid unnecessary re-testing, reduce regulatory risk, and significantly improve the success rate of extractables and leachables studies.
5: Practical Example of Correct Sample Preparation
The importance of E&L Sample Preparation Best Practices becomes much clearer when we look at a real-world scenario from regulatory testing.
Case-study-based learning is also covered in this article: FDA extractables and leachables case studies.
Scenario
A pharmaceutical company was performing leachable testing for a drug product packaged in plastic containers. During the initial analysis, several unexpected unknown peaks were observed in the chromatogram. These peaks created serious concerns regarding product safety and regulatory acceptance.
The company initially suspected the packaging material. However, a deeper investigation revealed that the issue was not related to the packaging at all.
Root Cause
The unexpected peaks were caused by poor sample preparation rather than actual leachables from the product packaging. The main issues identified were:
- Non-validated syringe filters were used during sample filtration
- Extraction temperature was significantly higher than required
- Blank samples were not prepared before analysis
- Sample handling was not performed under controlled conditions
This situation clearly shows how even small deviations from E&L Sample Preparation Best Practices can lead to misleading analytical results.
Solution
The study was repeated using a scientifically controlled preparation approach similar to the one followed by experienced laboratories such as ResolveMass Laboratories Inc..
The corrective actions included:
- Using validated low-extractable syringe filters
- Performing extraction under controlled temperature conditions
- Preparing and analyzing blank samples before the actual study
- Following proper contamination-control procedures
Final Outcome
Once E&L Sample Preparation Best Practices were properly followed:
- The unknown peaks completely disappeared
- The chromatographic results became reproducible
- The analytical report became regulatory-acceptable
- The study did not require further re-testing
This example clearly demonstrates that accurate sample preparation is not just a technical step — it is the foundation of reliable extractables and leachables testing.

6: Table: Correct vs Incorrect Sample Preparation Practices
Understanding the difference between incorrect practices and E&L Sample Preparation Best Practices helps prevent analytical errors and ensures reliable, regulatory-acceptable results.
This structured approach is also part of modern Extractables and Leachables (E&L) testing services at ResolveMass Laboratories Inc..
| Parameter | Incorrect Approach | Best Practice Approach |
|---|---|---|
| Sample handling | Open laboratory environment | Clean, controlled sample preparation area |
| Extraction solvent | Single solvent used without validation | Multi-solvent extraction strategy based on polarity |
| Filtration | Standard laboratory filters | Low-extractable, validated filters |
| Storage of prepared samples | Plastic containers | Certified glass containers |
| Blank testing | Not performed | Mandatory before analytical testing |
| Extraction conditions | Uncontrolled temperature and time | Scientifically controlled extraction parameters |
| Sample labeling | Manual, inconsistent labeling | Properly documented and traceable labeling system |
This table clearly demonstrates how following E&L Sample Preparation Best Practices improves analytical accuracy, reproducibility, and regulatory acceptance. Laboratories such as ResolveMass Laboratories Inc. follow structured and validated preparation procedures to ensure that every analytical result is scientifically reliable and submission-ready.
7: How Regulatory-Focused Laboratories Ensure Reliable Results
Not all analytical laboratories follow the same level of scientific rigor. Laboratories that specialize in regulatory submissions follow highly controlled and scientifically validated workflows to ensure that every result is accurate, reproducible, and acceptable to global regulatory agencies.
If companies prefer outsourcing instead of building in-house capability, this guide explains: outsourcing extractables and leachables testing laboratory in the United States.
Regulatory-focused laboratories typically ensure reliability through:
- Standardized sample preparation protocols that are designed specifically for extractables and leachables testing
- Scientifically validated analytical methods to ensure reproducibility and accuracy
- Advanced contamination-control systems to prevent false impurity results
- Submission-ready documentation that meets regulatory expectations
- Expert scientific review of every sample preparation step before final reporting
Because extractables and leachables studies are highly sensitive, even small preparation errors can lead to misleading results or regulatory delays. This is why many pharmaceutical companies prefer working with specialized laboratories such as ResolveMass Laboratories Inc., where the focus is not just testing but delivering reliable, regulatory-ready analytical data.
8: Benefits of Following E&L Sample Preparation Best Practices
Following E&L Sample Preparation Best Practices significantly improves the accuracy, reliability, and regulatory acceptance of extractables and leachables studies. The right preparation strategy not only improves analytical performance but also reduces the risk of costly regulatory delays.
These benefits are especially important in modern areas such as future of extractables and leachables testing and advanced biologic products.
Implementing scientifically controlled sample preparation provides advantages in both technical and regulatory aspects.
Technical Benefits
When E&L Sample Preparation Best Practices are followed correctly, laboratories can achieve:
- More accurate impurity and leachable identification
- Improved reproducibility across multiple studies
- Better sensitivity in advanced techniques such as LC-MS and GC-MS
- Reduced risk of false positive or false negative results
- More reliable analytical data for long-term stability studies
Experienced laboratories such as ResolveMass Laboratories Inc. focus heavily on sample preparation because it directly affects the quality of analytical results.
Regulatory Benefits
From a regulatory perspective, proper sample preparation plays a major role in submission success. Following E&L Sample Preparation Best Practices helps companies achieve:
- Faster approval timelines
- Reduced need for repeat studies
- Lower overall development costs
- Higher credibility of analytical data during regulatory review
- Stronger and more submission-ready regulatory documentation
This is why E&L Sample Preparation Best Practices are not optional — they are essential for any successful extractables and leachables study and for ensuring long-term regulatory compliance.
9: Final Checklist for E&L Sample Preparation
Following a structured checklist is one of the easiest ways to ensure that E&L Sample Preparation Best Practices are implemented correctly from the beginning of the study to the final analytical report. A well-defined checklist helps prevent contamination, improves reproducibility, and ensures regulatory-ready results.
Before starting E&L testing, make sure the following checklist is followed:
Sample Preparation Checklist
- Production-equivalent samples are used
- Solvent compatibility is verified before extraction
- Extraction conditions (temperature, time, and solvent ratio) are validated
- Blank samples are prepared and tested before analysis
- Low-extractable validated filters are used
- Proper labeling and storage conditions are followed
- Sample handling is performed in a controlled preparation area
- Complete documentation is recorded for regulatory review
Companies planning global submissions can also follow the detailed workflow explained in ICH Q3E extractables and leachables study requirements.
Laboratories specializing in regulatory submissions, such as ResolveMass Laboratories Inc., rely on structured preparation checklists to ensure that every study follows E&L Sample Preparation Best Practices from start to finish.
Conclusion:
In extractables and leachables testing, sample preparation determines the success or failure of the entire study. Even highly advanced analytical instruments cannot compensate for poorly prepared samples.
By following E&L Sample Preparation Best Practices, pharmaceutical companies can improve analytical accuracy, reduce regulatory risks, and ensure faster approval timelines. Laboratories such as ResolveMass Laboratories Inc. specialize in scientifically controlled sample preparation methods that produce reliable, submission-ready results.
Frequently Asked Questions:
E&L Sample Preparation Best Practices are scientifically validated procedures used to prepare pharmaceutical samples, packaging materials, and medical device components before extractables and leachables testing. These practices ensure accurate results, prevent contamination, and improve regulatory acceptance.
Sample preparation is critical because it determines whether the analytical results truly represent the chemical profile of the product. Poor preparation can lead to false impurity results, missing leachables, and regulatory rejection.
The most common mistakes include using unverified extraction solvents, ignoring blank samples, using non-validated filters, overheating samples, and poor storage of prepared samples. Following E&L Sample Preparation Best Practices helps avoid these errors.
Regulatory-focused laboratories follow standardized preparation protocols, validated analytical methods, contamination-control procedures, and submission-ready documentation. Laboratories such as ResolveMass Laboratories Inc. specialize in producing reliable and regulatory-acceptable data.
Common solvents used in E&L Sample Preparation Best Practices include water and methanol for polar compounds, acetonitrile for semi-polar compounds, and hexane or isopropanol for non-polar compounds. A multi-solvent approach is often used to ensure complete extraction.
Reference
- Manual EP. INTRODUCTION TO GOOD LABORATORY PRACTICES 2 (0+ 2).https://buat.edu.in/wp-content/uploads/2025/08/E-Manual-Content-Introduction-to-Good-Laboratory-Practices.pdf
- Abou Mrad C. Best practices for preparation of samples for laboratory analysis (Doctoral dissertation, Politecnico di Torino).https://webthesis.biblio.polito.it/29747/?template=default
- Cecchini I, Mastroianni D, Rocchi S, Angiuoni G. Evaluating E&L Studies for Single-Use Systems. BioPharm Intl. 2017 Feb;30(2):38-44.http://alfresco-static-files.s3.amazonaws.com/alfresco_images/pharma/2018/09/11/5667b8e4-47e0-4584-9038-801d6feaf320/BioPharm_Intl_Feb2017.pdf#page=40
- Stone J. Sample preparation techniques for mass spectrometry in the clinical laboratory. InMass spectrometry for the clinical laboratory 2017 Jan 1 (pp. 37-62). Academic Press.https://www.sciencedirect.com/science/article/pii/B9780128008713000031


