Introduction
Peptide Oligonucleotide Conjugates Pharmacokinetics focuses on how peptide-linked nucleic acid therapeutics distribute, metabolize, and remain active in biological systems after administration. These advanced molecules combine the targeting ability of peptides with the gene-modifying function of oligonucleotides. Because of this dual structure, their pharmacokinetic behavior reflects both the peptide component and the nucleic acid component interacting with the biological environment.
Unlike many traditional drugs, peptide-oligonucleotide conjugates show complex pharmacokinetic patterns. Their behavior is largely driven by tissue uptake, intracellular trafficking, and slow release inside cells rather than simple plasma concentration changes. In many cases, therapeutic success depends on how efficiently these molecules enter cells and escape intracellular compartments.
Explore our specialized solutions for drug discovery: Custom Synthesis for Drug Discovery
The conjugation of peptides to antisense oligonucleotides (ASOs), small interfering RNA (siRNA), or phosphorodiamidate morpholino oligomers (PMOs) has become an important strategy for improving delivery efficiency. These conjugates help overcome one of the biggest challenges in nucleic acid therapeutics—efficient cellular entry. Peptide ligands can guide these molecules to specific tissues and improve biological uptake.
However, these structural modifications also introduce unique pharmacokinetic dynamics. These include changes in clearance rates, receptor-mediated biodistribution, and extended intracellular half-life. Understanding these features of Peptide Oligonucleotide Conjugates Pharmacokinetics is essential during drug development to achieve optimal therapeutic performance.
Share via:
Summary of Key Insights
- Peptide Oligonucleotide Conjugates Pharmacokinetics is primarily governed by rapid tissue distribution, receptor-mediated uptake, and slow intracellular release rather than classical small-molecule ADME behavior.
- Conjugation of peptides significantly modifies plasma clearance, tissue targeting, and cellular uptake of antisense oligonucleotides (ASOs) and siRNA molecules.
- Pharmacokinetic profiles typically involve a fast plasma distribution phase followed by prolonged tissue retention, especially in liver, kidney, and muscle tissues.
- Peptide ligands influence receptor-mediated transport, endosomal escape, and intracellular trafficking, which directly affect therapeutic exposure.
- Clearance pathways primarily involve nuclease-mediated degradation and renal filtration, but conjugation often reduces renal clearance and increases bioavailability.
- Advanced PK modeling approaches such as PBPK models and cross-species translational modeling are increasingly used to predict human pharmacokinetics.
- Structural design parameters including peptide charge, hydrophobicity, linker chemistry, and oligonucleotide backbone modifications strongly influence PK behavior.
- Understanding Peptide Oligonucleotide Conjugates Pharmacokinetics is critical for optimizing dosing strategies, improving tissue specificity, and accelerating clinical development.
Key Determinants of Peptide Oligonucleotide Conjugates Pharmacokinetics
The pharmacokinetic profile of peptide-oligonucleotide conjugates is mainly influenced by molecular design factors. These include peptide composition, linker chemistry, oligonucleotide backbone modifications, and receptor affinity. Each of these parameters affects how the conjugate interacts with biological membranes, plasma proteins, and intracellular structures.
Even small changes in molecular structure can strongly influence pharmacokinetic behavior. For example, modifications to the peptide sequence may change receptor binding or tissue targeting. Similarly, backbone modifications in the oligonucleotide can increase stability and resistance to enzymatic degradation.
Learn more about different conjugate categories: Types of Peptide Oligonucleotide Conjugates
Physiological factors also play an important role. Tissue perfusion, receptor density, and cellular uptake capacity can affect how these molecules distribute in the body. Variations in receptor expression across organs may lead to selective accumulation in certain tissues.
Major PK Determinants
| Factor | Impact on Pharmacokinetics |
|---|---|
| Peptide ligand structure | Influences receptor targeting and tissue distribution |
| Oligonucleotide backbone modifications | Affects nuclease resistance and plasma stability |
| Linker chemistry | Determines release kinetics and intracellular activation |
| Molecular charge and hydrophobicity | Controls plasma protein binding and renal filtration |
| Receptor affinity | Drives receptor-mediated uptake and tissue accumulation |
Research shows that peptide conjugation can significantly change the volume of distribution and systemic clearance of oligonucleotide therapeutics. These changes often improve pharmacological exposure in target tissues. As a result, peptide conjugation is widely used to enhance delivery in modern oligonucleotide drug development.
For instance, ligand-conjugated antisense oligonucleotides often demonstrate increased cellular uptake through receptor-mediated endocytosis. This shifts the pharmacokinetic behavior from plasma circulation toward intracellular accumulation. As a result, the therapeutic oligonucleotide has a higher chance of reaching its RNA target and producing a sustained biological effect.
Understand the underlying biological processes: Peptide Oligonucleotide Conjugates Mechanism of Action
Tissue Distribution Patterns in Peptide Oligonucleotide Conjugates Pharmacokinetics
A defining feature of Peptide Oligonucleotide Conjugates Pharmacokinetics is the rapid movement of these molecules from plasma into tissues. This process can occur within minutes or hours after administration. The fast distribution happens because peptide ligands interact with cell surface receptors that actively transport the conjugates into tissues.
Once inside tissues, drug concentrations often remain elevated even when plasma levels decrease. This pattern differs from many small-molecule drugs, which rely mainly on passive diffusion across membranes. Peptide-conjugated oligonucleotides instead use active transport mechanisms that improve targeting and intracellular delivery.
Typical Tissue Distribution Profile
Liver–
High uptake due to receptor-mediated transport.
Particularly pronounced in GalNAc-type conjugates.
The liver is often the primary target for many oligonucleotide therapies because hepatocytes express high levels of uptake receptors.
Kidney–
Major site of accumulation due to filtration and tubular uptake.
Renal epithelial cells efficiently internalize oligonucleotides through endocytic pathways.
This accumulation can influence both efficacy and potential toxicity profiles.
Muscle tissues–
Relevant for peptide-PMO conjugates targeting muscular dystrophies.
Specialized cell-penetrating peptides improve delivery into muscle fibers.
Enhanced muscle targeting is particularly important for neuromuscular therapeutic applications.
Central nervous system–
Achievable via specialized targeting peptides.
Certain peptide ligands can facilitate transport across biological barriers.
This capability opens new possibilities for treating neurological diseases using oligonucleotide therapeutics.
Studies indicate that plasma concentrations may decline quickly because tissue distribution dominates systemic clearance. This characteristic is common in many nucleic acid therapeutics. Therefore, measuring tissue concentrations is often more informative than plasma levels when studying Peptide Oligonucleotide Conjugates Pharmacokinetics.
Access our full range of synthesis services: Peptide Oligonucleotide Conjugation Services
Plasma Clearance Mechanisms in Peptide Oligonucleotide Conjugates Pharmacokinetics
Clearance of peptide-oligonucleotide conjugates mainly occurs through nuclease degradation and renal filtration. However, peptide conjugation often slows these processes. Structural stability provided by peptides and backbone modifications helps protect the oligonucleotide from rapid enzymatic breakdown.
Major Clearance Pathways
- Endonuclease and exonuclease degradation
- Renal filtration of smaller fragments
- Hepatic uptake and metabolism
- Intracellular degradation after cellular uptake
Pharmacokinetic studies show several important trends. Conjugated oligonucleotides often display lower renal clearance compared with unconjugated molecules. Their larger molecular size and higher plasma protein binding reduce glomerular filtration.
In many cases, plasma elimination half-life appears short due to rapid tissue uptake. However, intracellular half-life can extend for days or even weeks. Once inside cells, oligonucleotides may remain within intracellular compartments and slowly release over time.
High plasma protein binding further contributes to reduced filtration and improved stability. These combined effects strongly influence the overall Peptide Oligonucleotide Conjugates Pharmacokinetics profile. As a result, dosing strategies often include loading doses followed by less frequent maintenance doses.
Ensure the durability of your therapeutic leads: Peptide Oligonucleotide Conjugate Stability
Cellular Uptake and Intracellular Pharmacokinetics
Cellular uptake plays a central role in Peptide Oligonucleotide Conjugates Pharmacokinetics because therapeutic activity depends on intracellular exposure. For gene modulation to occur, oligonucleotides must reach the cytoplasm or nucleus where RNA targets are located.
Most peptide-conjugated oligonucleotides enter cells through receptor-mediated endocytosis. In this process, the peptide ligand binds to specific receptors on the cell surface. The receptor-ligand complex is then internalized through vesicles formed during endocytosis.
Key Cellular PK Processes
- Cell surface receptor binding
- Endocytosis into early endosomes
- Endosomal trafficking
- Endosomal escape
- Nuclear or cytoplasmic target engagement
A major pharmacokinetic challenge is that only a small portion of internalized molecules escape from endosomes. Many remain trapped inside these compartments and are eventually degraded. Because of this limitation, improving endosomal escape is a major goal in peptide design.
Researchers often design peptides that can disrupt endosomal membranes and release the oligonucleotide into the cytoplasm. Improving this step significantly enhances pharmacodynamic activity and overall therapeutic performance.
Optimize your lead candidates with our expert CRO services: Peptide Oligonucleotide Conjugates CRO
Influence of Peptide Design on Peptide Oligonucleotide Conjugates Pharmacokinetics
Peptide composition strongly influences Peptide Oligonucleotide Conjugates Pharmacokinetics. Different peptide sequences interact with specific receptors and cellular membranes, affecting tissue targeting, cellular uptake, and drug distribution.
Important Design Parameters
Cationic peptides
- Improve cellular penetration
- Increase tissue accumulation
- Enhance interaction with negatively charged cell membranes.
Targeting ligands
- Enable receptor-specific delivery
- Improve tissue selectivity and therapeutic precision.
- Reduce off-target distribution.
Hydrophobic residues
- Enhance membrane interactions
- Facilitate endosomal escape and intracellular delivery.
Peptide length
- Influences molecular stability and PK distribution
- Longer peptides may improve targeting but can also affect metabolic stability.
For example, cell-penetrating peptides used in peptide-PMO conjugates can significantly improve muscle tissue uptake. This property is especially valuable for therapies aimed at treating neuromuscular diseases. As a result, peptide engineering plays a key role in optimizing delivery efficiency.
Review our technical characterization capabilities: Peptide Characterization Service
Pharmacokinetic Modeling of Peptide Oligonucleotide Conjugates
Pharmacokinetic modeling is increasingly used to understand and predict Peptide Oligonucleotide Conjugates Pharmacokinetics during drug development. Because these molecules interact with multiple biological systems, traditional pharmacokinetic models are often not sufficient.
Advanced modeling methods allow researchers to include biological processes such as receptor binding, tissue distribution, and intracellular trafficking.
Common Modeling Approaches
| Modeling Approach | Purpose |
|---|---|
| PBPK (Physiologically Based Pharmacokinetic) Models | Predict tissue distribution |
| PK/PD modeling | Link drug exposure to RNA knockdown |
| Cross-species scaling | Translate animal PK data to humans |
| Mechanistic endosomal trafficking models | Study intracellular exposure |
These models integrate several parameters, including receptor binding kinetics, intracellular degradation rates, and tissue distribution coefficients. By combining experimental data with computational simulations, researchers can better predict human dosing strategies.
This approach is highly valuable during clinical development because it reduces uncertainty before clinical trials begin.
Analytical Strategies for Studying Peptide Oligonucleotide Conjugates Pharmacokinetics
Accurate evaluation of Peptide Oligonucleotide Conjugates Pharmacokinetics requires specialized analytical techniques. Because these molecules contain both peptide and nucleic acid components, conventional bioanalytical methods may not provide sufficient sensitivity or specificity.
Common analytical methods include:
- LC-MS based bioanalysis
- Hybridization ELISA assays
- Mass spectrometry for metabolite identification
- Tissue distribution studies using radiolabeled compounds
These techniques allow researchers to measure several important parameters, including intact conjugate levels, metabolite formation, tissue exposure, and intracellular concentrations.
Reliable analytical data is essential during drug discovery and translational research. It helps scientists understand distribution patterns, degradation pathways, and pharmacokinetic variability. This information supports the development of safer and more effective therapeutic molecules.
Ensure rigorous quality standards for your conjugates: QC Testing for Peptide Oligonucleotide Conjugates
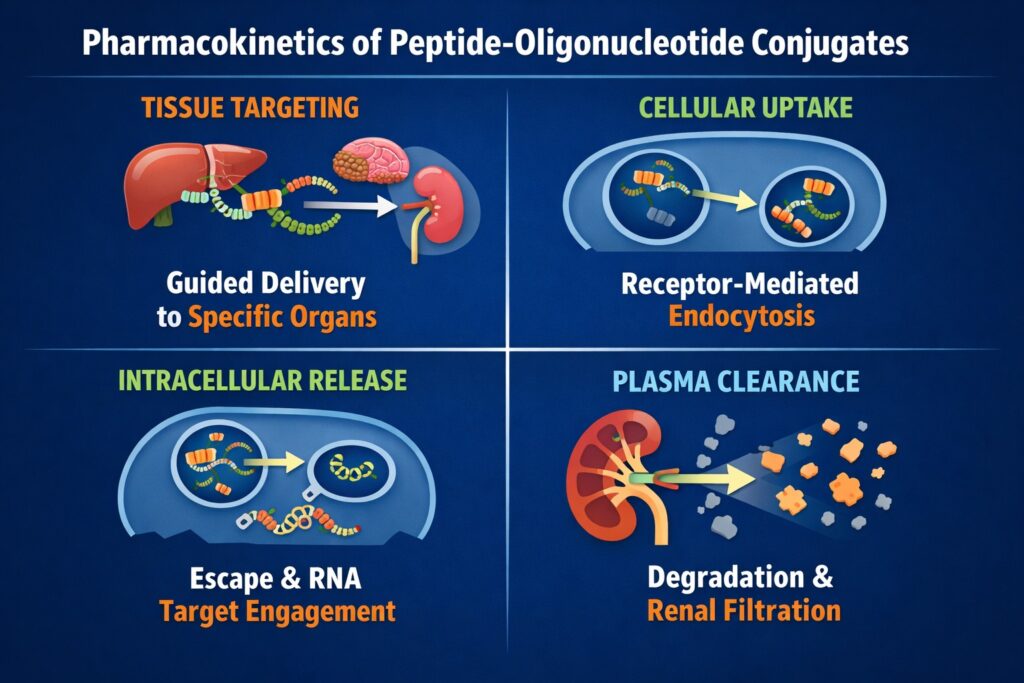
Challenges in Optimizing Peptide Oligonucleotide Conjugates Pharmacokinetics
Despite major progress in nucleic acid therapeutics, several challenges remain in optimizing Peptide Oligonucleotide Conjugates Pharmacokinetics. The complexity of intracellular delivery and tissue targeting makes consistent therapeutic exposure difficult to achieve.
Key Challenges
- Limited endosomal escape efficiency
- Non-specific tissue accumulation
- Immunogenicity of peptide sequences
- Complex intracellular trafficking dynamics
- Difficulties in measuring intracellular exposure
Researchers are actively working on new peptide engineering strategies to overcome these limitations. Advances in oligonucleotide chemistry and delivery technologies are also helping improve intracellular release and stability.
Combining experimental data with computational pharmacokinetic models may further improve design strategies. These integrated approaches allow scientists to better predict how molecular changes affect therapeutic performance.
Future Directions in Peptide Oligonucleotide Conjugates Pharmacokinetics
Future research in Peptide Oligonucleotide Conjugates Pharmacokinetics is focused on improving delivery efficiency and therapeutic precision. Scientists are exploring several innovative strategies to enhance pharmacokinetic performance.
Emerging research focuses on improving PK performance through:
- multivalent targeting ligands
- smart cleavable linkers
- improved peptide engineering
- advanced delivery systems
These technologies aim to achieve enhanced tissue specificity, reduced systemic toxicity, and prolonged intracellular activity. Such improvements could significantly increase the clinical success rate of nucleic acid therapeutics.
Future molecular designs may also respond to specific intracellular conditions. These adaptive systems could release drugs only in targeted cellular environments, improving precision and minimizing off-target effects.
As research progresses, understanding Peptide Oligonucleotide Conjugates Pharmacokinetics will remain essential for the development of effective oligonucleotide-based medicines.
Conclusion
Peptide Oligonucleotide Conjugates Pharmacokinetics plays a crucial role in determining the clinical performance of nucleic acid therapeutics. The interaction between peptide targeting elements and oligonucleotide therapeutic components creates a unique pharmacokinetic profile that differs significantly from traditional drugs.
These conjugates often show rapid tissue distribution, receptor-mediated uptake, prolonged intracellular persistence, and complex clearance pathways. Because of these characteristics, specialized analytical techniques and pharmacokinetic models are required to fully understand their behavior.
Advances in peptide engineering, conjugation chemistry, and modeling approaches are helping researchers design more efficient delivery systems. These developments are addressing long-standing challenges associated with nucleic acid drug delivery.
Continued research into Peptide Oligonucleotide Conjugates Pharmacokinetics will improve tissue targeting, enhance therapeutic efficacy, and accelerate the translation of oligonucleotide therapies into clinical practice. A deeper understanding of these mechanisms will ultimately support the development of safer and more effective treatments.
Discover our integrated CMC support: CMC Services for Peptide Oligonucleotide Conjugates
For scientific collaboration or analytical support related to peptide-oligonucleotide conjugate research, you can connect with our experts:
Frequently Asked Questions (FAQs)
Peptide conjugation changes pharmacokinetic behavior by improving receptor-mediated uptake and directing the oligonucleotide toward specific tissues. These peptides act as targeting ligands that guide the therapeutic molecule to certain cells. As a result, tissue accumulation increases and drug delivery becomes more efficient. This improved targeting can significantly enhance therapeutic exposure and activity.
Rapid plasma clearance often occurs because the molecules quickly move from blood circulation into tissues. Peptide ligands interact with receptors that actively transport the conjugates into cells. This process reduces plasma levels within a short time after administration. However, the molecules may remain inside tissues for extended periods where they continue to produce therapeutic effects.
Receptor-mediated uptake allows selective delivery of oligonucleotides to targeted tissues. The peptide ligand binds to specific receptors present on the cell surface. This interaction triggers internalization through endocytosis. The process strongly influences tissue distribution and improves intracellular drug delivery.
Although plasma half-life can be short, intracellular half-life may last several days or even weeks. Once inside cells, oligonucleotides can remain in intracellular compartments before gradually reaching their RNA targets. This prolonged presence supports sustained gene modulation. As a result, dosing frequency can often be reduced.
Clearance mainly occurs through nuclease-mediated degradation and renal filtration of smaller fragments. Nucleases break down the oligonucleotide into shorter sequences over time. These fragments are then removed through kidney filtration pathways. Hepatic metabolism and intracellular degradation also contribute to the overall elimination process.
Linker chemistry controls how and when the peptide separates from the oligonucleotide after cellular uptake. Cleavable linkers can release the active drug inside cells at the correct time. This improves drug activation and therapeutic efficiency. Proper linker design can also influence stability in circulation and distribution within tissues.
After entering cells through endocytosis, many oligonucleotide conjugates become trapped inside endosomes. Without escape from these vesicles, the molecules cannot reach their RNA targets. Efficient endosomal escape allows the therapeutic agent to access the cytoplasm or nucleus. Improving this step greatly enhances functional drug activity.
Reference:
- Klabenkova, K., Fokina, A., & Stetsenko, D. (2021). Chemistry of peptide-oligonucleotide conjugates: A review. Molecules, 26(17), 5420. https://doi.org/10.3390/molecules26175420
- Venkatesan, N., & Kim, B. H. (2006). Peptide conjugates of oligonucleotides: Synthesis and applications. Chemical Reviews, 106(9), 3712–3761. https://doi.org/10.1021/cr0502448
- Malinowska, A. L., Huynh, H. L., & Bose, S. (2024). Peptide-oligonucleotide conjugation: Chemistry and therapeutic applications. Current Issues in Molecular Biology, 46(10), 11031–11047. https://doi.org/10.3390/cimb46100655



