Introduction:
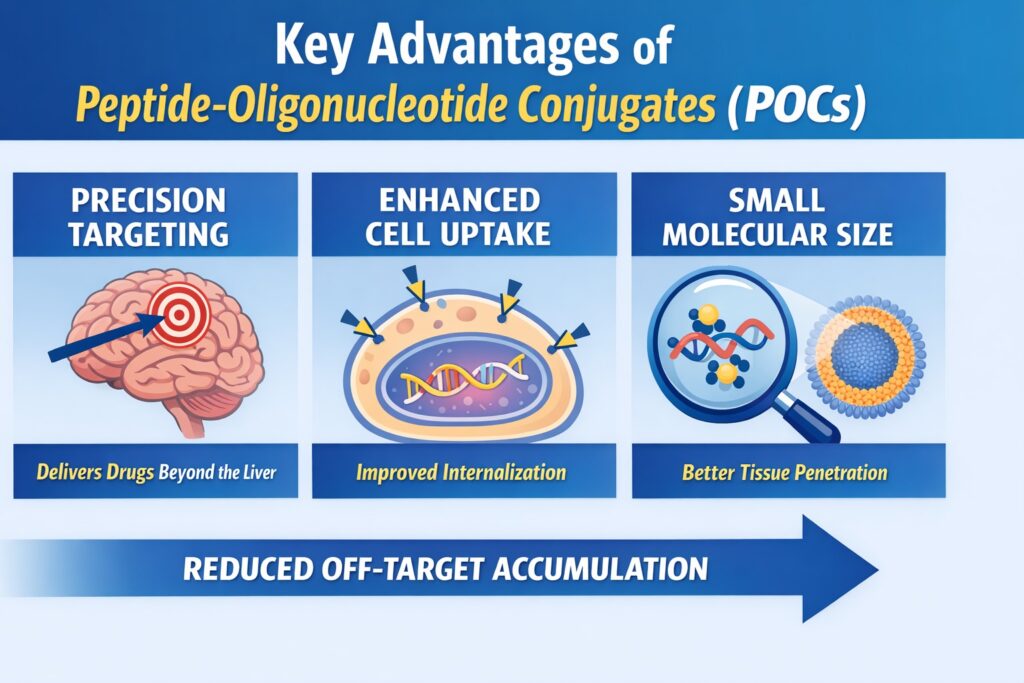
The primary therapeutic advantage of Peptide Oligonucleotide Conjugates Drug Delivery lies in its ability to enable precise, extrahepatic tissue targeting along with enhanced cellular internalization, all while maintaining a relatively small molecular size compared to conventional delivery systems. By covalently linking functional peptides to nucleic acid payloads, these hybrid constructs effectively overcome physiological barriers that typically confine unmodified oligonucleotides and lipid-based nanoparticles to organs like the liver and spleen. This targeted strategy not only improves drug distribution but also minimizes off-target accumulation, which is a major limitation of traditional platforms. Additionally, the reduced molecular footprint enhances tissue penetration and supports better pharmacodynamic outcomes. As a result, POCs are increasingly being recognized as a next-generation solution for precise and efficient nucleic acid delivery.
Explore our specialized solutions for drug development: Custom Synthesis for Drug Discovery
Share via:
Architectural Precision in Peptide Oligonucleotide Conjugates Drug Delivery
One of the biggest advantages of Peptide Oligonucleotide Conjugates Drug Delivery is its well-defined structure. Unlike lipid nanoparticles, which are made from multiple components and can vary between batches, POCs are single molecules with a fixed design. This reduces inconsistencies and makes results more reliable across production batches. It also simplifies regulatory approval because the structure is easier to analyze and confirm.
Learn more about our comprehensive conjugation services: Peptide Oligonucleotide Conjugation Services
In this system, peptides are directly attached to oligonucleotides through strong chemical bonds. These peptides are usually short chains of amino acids that guide the drug to specific cells. This design allows both targeting and delivery to happen at the same time within one molecule. As a result, the drug behaves more predictably inside the body and produces more consistent therapeutic effects.
Another benefit is improved cell entry. Oligonucleotides alone struggle to pass through cell membranes due to their size and charge. By adding cell-penetrating peptides like TAT or polyarginine, the conjugates can interact with the cell surface and enter more easily. This significantly increases how much of the drug reaches inside the cell where it is needed.
The small size of POCs also plays a key role. Compared to larger nanoparticles, they can travel deeper into tissues instead of being trapped in the liver. With additional chemical modifications, these conjugates can stay longer in circulation and avoid rapid removal by the immune system. This makes them highly useful for targeting a wide range of tissues.
| Metric | Lipid Nanoparticles (LNPs) | Peptide-Oligonucleotide Conjugates (POCs) |
| Particle Size (Hydrodynamic) | 60–100 nm | < 10 nm |
| Components | Ionizable lipid, helper lipid, cholesterol, PEG-lipid | Peptide, Linker, Oligonucleotide |
| Manufacturing Method | Microfluidics/Rapid mixing | Stepwise solid-phase or post-synthetic coupling |
| Surface Charge | Neutral/Cationic (pH dependent) | Variable (Typically lower net cationic charge) |
| Renal Clearance | Low (Size exceeds filtration cutoff) | High (Unless modified with PEG/Albumin-binders) |
Mechanisms of Receptor-Mediated Transcytosis in Peptide Oligonucleotide Conjugates Drug Delivery
The targeting ability of Peptide Oligonucleotide Conjugates Drug Delivery largely depends on receptor-mediated transcytosis. In this process, the peptide binds to specific receptors on the cell surface, allowing the drug to pass through biological barriers. This is especially important for reaching areas like the brain, where most drugs cannot enter easily.
Understand the science behind targeted delivery: Peptide Oligonucleotide Conjugates Mechanism of Action
For example, peptides such as Angiopep-2 target receptors that are highly present in brain cells. This helps the drug move from the bloodstream into brain tissue more effectively. Studies show that this method improves drug delivery to the brain compared to older targeting approaches. This opens new possibilities for treating brain diseases and tumors.
This approach is not limited to the brain. It can also be used to target muscle tissues, including skeletal and heart muscle. Specialized peptides help deliver genetic therapies directly into muscle cells, which is important for diseases like muscular dystrophy. This level of precision represents a major step forward in modern medicine.
| Targeted Receptor | Peptide Ligand | Tissue/Cell Target | Transcytosis Efficiency (vs. Naked Cargo) |
|---|---|---|---|
| LRP-1 | Angiopep-2 | BBB, Glioma cells | ~86-fold increase (Brain uptake) |
| nAChR | RVG-29 | Neurons, CNS | High neuronal specificity |
| TfR (Transferrin) | BACE1-siRNA complex | Neurons, Microglia | Targeted neuronal delivery |
| Integrin αvβ3 | RGD motifs | Neoangiogenic vessels, Tumors | Tumor-specific homing |
| ASGPR | GalNAc (Sugar) | Hepatocytes | >90% first-pass extraction |
Structural Optimization and Retro-Inverso Isomerization for Brain Penetration
Improving the stability of these systems is essential for better performance. One useful method is retro-inverso isomerization, where peptides are built using D-amino acids in reverse order. This keeps their function the same but makes them more resistant to breakdown in the body. As a result, the drug remains active for a longer time.
For instance, modified versions of Angiopep-2 show higher stability in blood compared to the natural form. This allows more of the drug to reach its target before being degraded. Increased stability also improves the chances of successful transport across barriers like the brain.
Another important factor is how many peptide ligands are attached. While more ligands can improve binding, too many can reduce the drug’s ability to move through tissues. This balance must be carefully controlled during design. POCs allow this level of precision, making them highly customizable.
Discover how stability impacts therapeutic efficacy: Peptide Oligonucleotide Conjugate Stability
Linker Engineering: Balancing Circulatory Stability with Intracellular Release
Linkers are a key part of Peptide Oligonucleotide Conjugates Drug Delivery systems. They connect the peptide and the oligonucleotide and control when the drug is released. A good linker must stay stable in the bloodstream but release the drug once inside the target cell.
Cleavable linkers respond to conditions inside cells, such as enzymes or pH changes. This ensures that the drug is released only where it is needed. This approach is especially useful in cancer treatment, where targeted release is critical.
Non-cleavable linkers provide higher stability during circulation. They release the drug only after the entire structure is broken down inside the cell. This reduces the risk of unwanted drug release and improves safety.
Deep dive into the chemistry of conjugate links: Peptide Oligonucleotide Conjugate Linker Chemistry
Special spacers can also be added to ensure the drug is released in its original form. This helps maintain full biological activity and improves treatment effectiveness.
| Linker Type | Release Mechanism | Key Chemical Feature | Application |
| Enzyme-Cleavable | Cathepsin B/Proteases | Val-Cit dipeptide | Rapid release in lysosomes |
| pH-Sensitive | Acidic environment | Hydrazone bonds | Endosomal/Lysosomal targeting |
| Reducible | Glutathione (GSH) | Disulfide bonds | Cytosolic release (GSH gradient) |
| Non-Cleavable | Complete degradation | Thioether / Amide | Maximum circulatory stability |
| Self-Immolative | Spontaneous cleavage | PABC spacers | Releases unmodified payload |
Dynamic Modulation of Pharmacokinetics via Linker Selection
The choice of linker affects how long the drug stays in the body and how it behaves. Stable linkers allow the drug to circulate longer, which helps maintain consistent treatment levels. This is important for therapies that require precise dosing.
These linkers can also improve solubility and reduce immune reactions. This makes the treatment safer and more comfortable for patients. For sensitive therapies like RNA-based drugs, this stability is very important.
On the other hand, cleavable linkers allow controlled release based on the target environment. This flexibility helps adapt the drug for different diseases and tissues. As a result, the system becomes more versatile.
Quantitative Comparison of Endosomal Escape Efficiency in Peptide Oligonucleotide Conjugates Drug Delivery
One major challenge in drug delivery is endosomal escape. Many drugs get trapped inside cell compartments and cannot reach their target. This limits their effectiveness.
Peptide Oligonucleotide Conjugates Drug Delivery addresses this issue by using special peptides that help break out of these compartments. These peptides respond to acidic conditions and disrupt the membrane, allowing the drug to enter the cytoplasm.
This method can increase escape efficiency significantly, sometimes up to 50%. Even small improvements in this step can lead to better treatment results. It also allows lower doses to be used, reducing side effects.
Get expert insights into synthesis and characterization: POC Synthesis and Characterization
| Platform | Endosomal Escape Efficiency (%) | Primary Site of Escape | Attrition Factors |
| Traditional LNPs | 1% – 5% | Early/Maturing Endosomes | Recycling, poor disassembly |
| GalNAc Conjugates | 1% – 2% | Late Endosomes | Receptor recycling (90%+) |
| POCs w/ CPPs | 5% – 10% | Endosomes / Direct Entry | Variable depending on CPP type |
| POCs w/ EEPs | Up to 50% | Maturing Endosomes | Optimized histidine switches |
Vesicle Budding-and-Collapse (VBC) vs. Proton Sponge Mechanisms
New research suggests that endosomal escape may happen through a process called vesicle budding-and-collapse. Instead of bursting the membrane, small vesicles form and then collapse, releasing the drug.
Peptides play an important role in this process by helping reshape the membrane. This method is safer and reduces damage to the cell. It also lowers the risk of inflammation.
Understanding these mechanisms helps scientists design better delivery systems. By adjusting peptide structure, they can improve efficiency and safety at the same time.
Advanced Bioanalytical Characterization and Mass Spectrometry Validation
High-quality analysis is essential for developing these systems. Techniques like mass spectrometry and liquid chromatography are used to confirm the structure and purity of the conjugates. This ensures that the final product is safe and effective.
Because POCs combine peptides and nucleic acids, they can be difficult to analyze. Advanced tools like Orbitrap systems help overcome these challenges by providing accurate and detailed data.
Ensure the highest standards for your therapeutic leads: QC Testing for Peptide Oligonucleotide Conjugates
These methods are also important for regulatory approval. They help detect impurities and confirm that the product meets required standards.
| Analytical Challenge | ResolveMass Laboratory Solution | Impact on Drug Development |
| Structural Complexity | Tandem MS (MS/MS) & NMR | Confirm exact sequence & attachment site |
| Low Therapeutic Conc. | LC-MS/MS with 0.1 ng/mL LLOQ | Reliable PK data for clinical trials |
| Impurity Profiling | Peak deconvolution & multi-mode HPLC | Detection of sub-0.1% impurities |
| Stability Assessment | Forced degradation studies (pH/Temp) | Predictive shelf-life & storage protocols |
| Metabolic Mapping | HRMS metabolite identification | Identification of cleavage sites/metabolites |
Precision Peptide Sequencing and Sameness Studies
Accurate sequencing ensures that the peptide part of the conjugate is correct. Techniques like tandem mass spectrometry are used to verify the amino acid sequence. This is especially important for complex designs.
Sameness studies compare new products with existing ones to ensure they perform the same way. These studies are important for developing biosimilars and maintaining quality.
Consistency across batches is critical for success. These analytical methods help ensure that every product meets the same high standards.
Detailed characterization services for complex molecules: Peptide Characterization Service
Overcoming the Limitations of Cationic Charge and Innate Immunogenicity
A key benefit of Peptide Oligonucleotide Conjugates Drug Delivery is reduced immune response. Unlike some delivery systems, POCs do not rely heavily on positive charge, which can trigger inflammation.
Their smaller size also helps them avoid detection by the immune system. This allows them to stay in circulation longer and reach their target more effectively.
Using natural peptide sequences further improves compatibility with the body. This makes the treatment safer and suitable for long-term use.
| Parameter | Cationic LNPs | POC-based Delivery |
| Net Charge Ratio | High (Positive) | Low (Optimized/Minimal) |
| TLR Activation | Significant (TLR 2, 4, 7/8) | Minimal (Depending on sequence) |
| Zeta Potential | Highly positive ($+20$ to $+50$ mV) | Near-neutral to slightly positive |
| Liver Sequestration | High (Driven by opsonization) | Targeted (Driven by ligand) |
| Safety Window | Narrow (Dose-limited toxicity) | Broader (Reduced non-specific uptake) |
Strategic Value of Analytical Precision in Peptide Oligonucleotide Conjugates Drug Delivery
Precise analysis helps identify problems early in development. This reduces the risk of failure and lowers overall costs. It also speeds up the path to market.
Testing for harmful contaminants is another important step. Advanced tools can detect even very small amounts of dangerous substances. This ensures patient safety.
Strong quality control builds trust and supports successful product launch. It is a key part of modern drug development.
Navigate regulatory requirements with our CMC expertise: CMC Services for Peptide Oligonucleotide Conjugates
Scale-Up, GMP Compliance, and the Future of Personalized Nucleic Acid Therapeutics
Scaling up production of POCs requires careful control to maintain quality. Combining peptide and nucleic acid synthesis can be complex, but advanced methods help manage this process.
Technologies like real-time monitoring ensure consistent production. Quality by Design approaches further improve reliability and compliance.
In the future, smaller and more flexible manufacturing systems may allow personalized treatments. However, strong regulations will be needed to maintain quality across all sites.
| Scale-Up Hurdle | Mitigation Strategy | Future Outlook |
| Manufacturing Complexity | Automated, closed-system bioreactors | Reduced manual labor and defect rates |
| Cost of Production | Lean manufacturing & scale-out models | Wider access to personalized therapies |
| Regulatory Variation | Early collaboration with regulators (MHRA/FDA) | International alignment on CMC frameworks |
| Supply Chain Risks | Diversified sourcing & supply chain analytics | More resilient production pipelines |
| Technology Transfer | Standardized SOPs & pilot-scale testing | Faster time-to-market for new TIDEs |
Clinical Landscape and Market Projections (2024–2030)
The clinical progress of Peptide Oligonucleotide Conjugates Drug Delivery is accelerating rapidly. Recent FDA approvals highlight the growing importance of TIDEs in modern therapeutics. These advancements demonstrate the viability of targeted delivery systems.
GalNAc-based therapies have shown strong performance in liver targeting. Meanwhile, peptide-based systems are advancing in neurological and muscular disorders. Ongoing clinical trials are expected to expand these applications further.
Market projections indicate strong growth, driven by increasing demand for RNA-based therapies. POCs are expected to play a central role in this expansion, offering precise and effective treatment options.
Conclusion
The advantages of Peptide Oligonucleotide Conjugates Drug Delivery are reshaping how modern therapies are developed and delivered. With better targeting, improved cell entry, and efficient release mechanisms, these systems overcome many traditional limitations. They make it possible to treat difficult areas like the brain and muscles more effectively.
Recent progress shows that this technology is moving quickly toward wider clinical use. Continued improvements in design and analysis will further strengthen its impact. These systems offer a powerful combination of safety, precision, and performance.
ResolveMass Laboratories Inc. supports this advancement through expert bioanalytical services and scientific guidance. Their work helps bring complex therapies closer to real-world use. As innovation continues, POCs are expected to play a major role in the future of personalized medicine.
Partner with a leading CRO for your next project: Peptide Oligonucleotide Conjugates CRO
For advanced bioanalytical services and custom synthesis support to accelerate your drug development programs, please reach out to our team of specialists: Contact us
Frequently Asked Questions (FAQs)
Peptide Oligonucleotide Conjugates Drug Delivery enables targeting beyond the liver by using peptides that bind to specific receptors on different tissues like the brain or muscles. These peptides guide the drug to the right location and help it cross biological barriers. Through receptor-mediated transcytosis, the drug can move across tight barriers such as the blood-brain barrier. This approach allows effective delivery to areas that are usually difficult to reach.
POCs offer several advantages compared to lipid nanoparticles, mainly due to their smaller size and precise chemical structure. This allows better tissue penetration and more predictable behavior in the body. They also carry less positive charge, which lowers the chances of immune system activation. Because of this, POCs are generally safer and more suitable for repeated or long-term treatments.
The histidine switch works by responding to changes in pH inside the cell. In the bloodstream, the peptide remains stable and inactive at neutral pH. Once inside the acidic environment of the endosome, histidine residues gain a positive charge. This change helps the peptide disrupt the endosomal membrane and release the drug into the cytoplasm.
Certain peptides like Angiopep-2 and RVG-29 are highly effective for brain delivery. Angiopep-2 targets receptors on brain endothelial cells, helping transport drugs into brain tissue. RVG-29 binds to receptors found on neurons, allowing more direct delivery داخل the brain. These ligands improve the efficiency of delivering therapeutic molecules to the central nervous system.
Mass spectrometry is essential for confirming the identity and quality of POCs during development. It helps determine the exact molecular structure and detect even very small impurities. Advanced systems provide highly accurate data, ensuring the product meets strict quality standards. This level of analysis is important for regulatory approval and patient safety.
Producing POCs at a large scale requires careful control of quality and consistency. It is important to maintain the correct ratio between peptide and oligonucleotide components. High purity and efficient chemical bonding must also be ensured during synthesis. Advanced monitoring systems are used to maintain stable production conditions in GMP facilities.
POCs lower immune system reactions by avoiding large and complex particle structures. Their smaller size and simpler design make them less likely to trigger immune responses. They also use lower positive charge, which reduces activation of immune receptors. This results in better safety and improved tolerance in patients.
Reference:
- Li, Y., Zheng, X., Gong, M., & Zhang, J. (2016). Delivery of a peptide-drug conjugate targeting the blood brain barrier improved the efficacy of paclitaxel against glioma. Oncotarget, 7(48), 79401–79407. https://doi.org/10.18632/oncotarget.12708
- Ghorai, S. M., Deep, A., Magoo, D., Gupta, C., & Gupta, N. (2023). Cell-penetrating and targeted peptides delivery systems as potential pharmaceutical carriers for enhanced delivery across the blood-brain barrier (BBB). Pharmaceutics, 15(7), 1999. https://doi.org/10.3390/pharmaceutics15071999
- Yang, L., Ma, J., Liu, B., Li, Y., Ma, Y., Chen, H., & Han, Z. (2025). Recent advances in peptide linkers of antibody–drug conjugates. Journal of Medicinal Chemistry, 68(17), 18099–18113. https://doi.org/10.1021/acs.jmedchem.5c01500
- Jensen, O. N., Kulkarni, S., Aldrich, J. V., & Barofsky, D. F. (1996). Characterization of peptide–oligonucleotide heteroconjugates by mass spectrometry. Nucleic Acids Research, 24(19), 3866–3872. https://doi.org/10.1093/nar/24.19.3866
- Malinowska, A. L., Huynh, H. L., & Bose, S. (2024). Peptide–oligonucleotide conjugation: Chemistry and therapeutic applications. Current Issues in Molecular Biology, 46(10), 11031–11047. https://doi.org/10.3390/cimb46100655



