
Introduction:
Leachable Screening for Parenteral Formulations is one of the most critical quality activities in modern pharmaceutical development. Because parenteral drug products are injected directly into the body, even trace levels of leachables can affect patient safety, product stability, and regulatory approval.
Designing a scientifically sound leachables screening strategy is not just about running analytical tests. It requires deep expertise in material science, analytical chemistry, toxicology risk assessment, and regulatory expectations. This article explains how to design an effective and compliant leachables screening strategy specifically for parenteral drug products.
Share via:
Summary:
- Leachables risk is highest in parenteral products because they bypass the body’s natural defenses.
- A well-designed Leachable Screening for Parenteral Formulations must begin early in development, not just before submission.
- Material selection, extraction strategy, and sensitive analytical techniques all play a critical role.
- Regulatory expectations from global agencies require a science-based and risk-based approach.
- This article explains how to design a robust, regulatory-ready leachables screening program step-by-step.
1: Why Leachable Screening for Parenteral Formulations Is Critical
Leachable Screening for Parenteral Formulations is critical because injectable drug products bypass the body’s natural defense systems and enter directly into systemic circulation. As a result, even trace-level contaminants can pose significant safety risks to patients and may also impact product stability and regulatory approval.
Key Reasons It Is Critical
- No biological barriers: Unlike oral dosage forms, parenteral products do not pass through the digestive system where many impurities may be neutralized or degraded.
- Higher risk from packaging materials: Components such as rubber stoppers, pre-filled syringes, vial coatings, tubing, and filters are potential sources of leachables.
- Stricter regulatory expectations: Regulatory agencies apply much tighter limits and stronger scrutiny for injectable products compared to oral or topical formulations.
- Impact on drug stability and performance: Leachables can interact with the formulation, causing degradation, reduced potency, or physical instability.
- Risk of product recalls and submission delays: Failure to properly identify and control leachables can lead to major regulatory concerns and costly product recalls.
Because of these factors, regulatory authorities now expect a proactive, science-based leachables strategy rather than a reactive testing approach performed late in development. A well-designed screening program not only protects patient safety but also strengthens the overall quality and regulatory confidence in the product.
2: What Is Leachable Screening for Parenteral Formulations?
Leachable Screening for Parenteral Formulations is a systematic analytical study designed to detect, identify, and quantify chemical compounds that migrate from packaging materials, manufacturing components, or drug-device systems into injectable drug products. Because parenteral products are administered directly into the body, even very low levels of leachables must be carefully evaluated and controlled.
This type of screening is not limited to packaging alone. It also includes all materials that come into direct or indirect contact with the drug product during manufacturing, storage, and administration.
Typical Sources of Leachables
| Source Category | Examples |
|---|---|
| Packaging materials | Rubber stoppers, vial coatings, glass vials, syringe components |
| Manufacturing systems | Tubing, connectors, filters, seals, transfer lines |
| Drug-device combination products | Pre-filled syringes, cartridges, autoinjectors, infusion systems |
| Storage conditions | High temperature, long-term storage, light exposure, humidity effects |
A well-planned Leachable Screening for Parenteral Formulations study helps identify potential risks early in development, reduces regulatory concerns during submission, and ensures long-term product safety and stability.
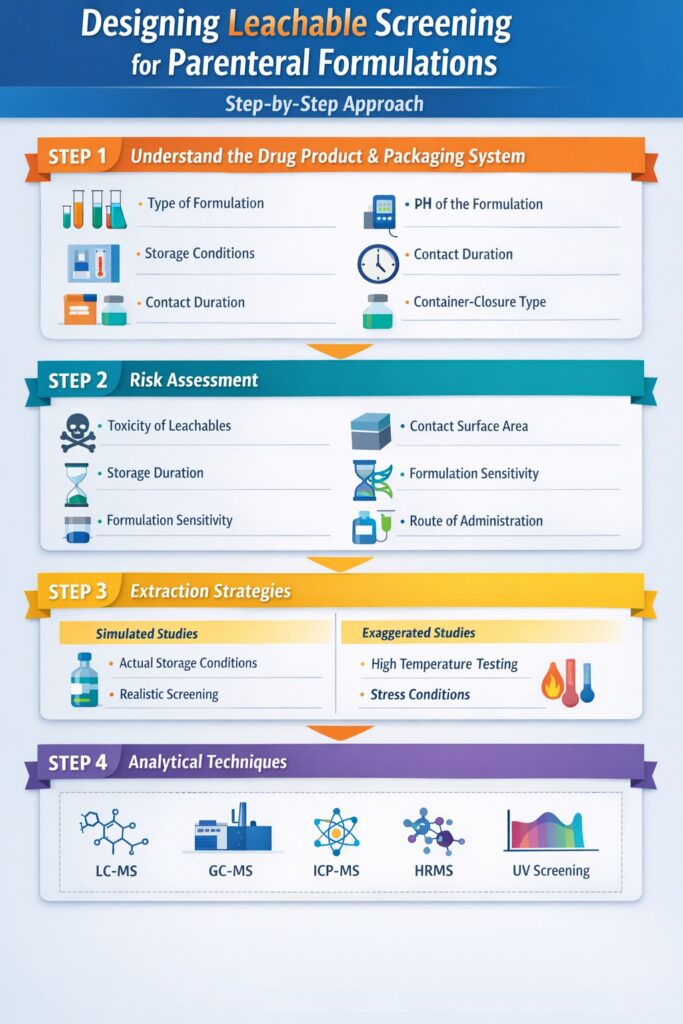
3: Designing Leachable Screening for Parenteral Formulations: Step-by-Step Approach
Designing Leachable Screening for Parenteral Formulations requires a structured and risk-based strategy to generate scientifically reliable and regulatory-acceptable data. A well-planned study does not start in the laboratory — it starts with understanding the product, materials, and potential risks.
Below is a step-by-step framework used in effective leachables screening programs.
Step 1: Understand the Drug Product and Packaging System
The first and most important step in Leachable Screening for Parenteral Formulations is understanding how the drug product interacts with its packaging and delivery system. Different formulations behave very differently when in contact with packaging materials.
Important factors to evaluate
- Type of formulation: Aqueous formulations, lipid-based products, biologics, and small-molecule injectables all have different leachables risk levels.
- pH of the formulation: Highly acidic or alkaline products can increase the probability of leachable formation.
- Storage conditions: Room temperature, refrigerated, or frozen storage can significantly influence leachable migration.
- Duration of contact: Long-term storage increases the risk of leachables compared to short-term products.
- Type of container-closure system: Vials, rubber stoppers, pre-filled syringes, cartridges, and infusion components all contribute differently to risk.
This evaluation forms the scientific foundation of the entire leachables screening strategy.
Step 2: Risk Assessment in Leachable Screening for Parenteral Formulations
A risk-based approach ensures that Leachable Screening for Parenteral Formulations focuses on the most critical risk areas instead of performing unnecessary testing.
Key factors considered in risk assessment
- Toxicity profile of potential leachable compounds
- Contact surface area between formulation and packaging materials
- Storage duration and shelf-life of the product
- Sensitivity of the formulation (biologics and complex formulations are more sensitive)
- Route of administration (IV products generally carry the highest risk)
A scientifically justified risk assessment not only improves data quality but also increases the likelihood of smooth regulatory acceptance.
Step 3: Selecting the Right Extraction Strategy
An effective Leachable Screening for Parenteral Formulations must balance realism with sensitivity. The study should simulate real-world conditions while still detecting low-level contaminants.
Two main study approaches
Simulated Leachables Studies
- Use real drug product or placebo formulation
- Conducted under actual storage conditions
- Provide realistic and regulatory-preferred results
- Essential for submission-ready data
Exaggerated Leachables Studies
- Use elevated temperature or stress conditions
- Help identify worst-case scenarios
- Useful during early product development
- Support risk-based decision making
In most cases, combining both approaches provides the most complete and reliable leachables data.
Step 4: Analytical Techniques Used in Leachable Screening for Parenteral Formulations
The success of Leachable Screening for Parenteral Formulations depends heavily on using advanced and highly sensitive analytical techniques capable of detecting trace-level contaminants.
Common analytical techniques used
- LC-MS: Ideal for semi-volatile and non-volatile organic compounds
- GC-MS: Used for volatile organic compounds and residual solvents
- ICP-MS: Essential for detecting elemental impurities and metal contaminants
- High-resolution mass spectrometry (HRMS): Critical for identifying unknown compounds
- UV-based screening methods: Useful for early-stage screening and trend monitoring
Each technique plays a specific role, and using a combination of complementary methods ensures complete screening coverage.
4: How to Identify Unknown Leachables Effectively
Identifying unknown compounds is one of the most complex steps in Leachable Screening for Parenteral Formulations. Detecting a chromatographic peak alone is not sufficient — the compound must be accurately identified, confirmed, and evaluated from a toxicological perspective.
Because parenteral products are administered directly into the bloodstream, even trace-level unknowns must be carefully investigated and scientifically justified.
A Reliable Process for Identifying Unknown Leachables
A well-structured identification workflow typically includes the following steps:
- High-resolution mass spectrometry (HRMS): Provides accurate mass measurements that help determine the molecular formula of unknown compounds.
- Fragmentation pattern analysis: MS/MS fragmentation data helps confirm structural features and distinguish between similar compounds.
- Database comparison: Spectral libraries and scientific databases are used to match unknown peaks with known chemical structures.
- Structural confirmation using reference standards: Once a likely structure is identified, analytical confirmation using a certified reference standard improves reliability.
- Toxicological risk evaluation: Each identified compound must be evaluated to determine whether it presents a patient safety risk.
Why Experience Matters in Unknown Identification
Unknown identification in Leachable Screening for Parenteral Formulations is not just an analytical task — it requires deep expertise in mass spectrometry, organic chemistry, and regulatory expectations. Misidentification can lead to unnecessary regulatory concerns or, in some cases, missed safety risks.
Laboratories with strong experience in extractables and leachables studies are therefore more likely to identify unknown compounds accurately, justify their safety, and support smooth regulatory submissions.
5: Regulatory Expectations for Leachable Screening for Parenteral Formulations
Regulatory Expectations for Leachable Screening for Parenteral Formulations are focused on a science-based, risk-driven, and well-documented strategy. For injectable products, regulators expect clear evidence that potential leachables have been properly identified, evaluated, and controlled throughout the product lifecycle.
Poorly designed studies or incomplete identification of compounds often result in regulatory questions, data requests, and delays in product approval.
Key Expectations from Regulatory Authorities
A regulatory-ready Leachable Screening for Parenteral Formulations program should include:
- Risk-based justification of the testing strategy: The study design must clearly explain why specific materials, conditions, and analytical methods were selected.
- Identification of unknown compounds whenever possible: Regulatory agencies expect unknown peaks to be investigated and structurally characterized, not ignored.
- Toxicological safety assessment: Each detected leachable should be evaluated to determine whether it presents a potential patient safety risk.
- Clear analytical method validation: Analytical techniques must be properly validated to ensure reliable and reproducible results.
- Long-term stability monitoring of leachables: Leachables must be evaluated throughout the product’s shelf life, not just at the initial stage.
Why a Structured Approach Matters
A structured and scientifically justified Leachable Screening for Parenteral Formulations strategy ensures that the generated data is not only analytically accurate but also fully aligned with global regulatory expectations. This significantly reduces the risk of submission delays, additional studies, and regulatory objections.
6: Common Mistakes in Leachable Screening for Parenteral Formulations
Common mistakes in Leachable Screening for Parenteral Formulations often occur due to poor planning, late study initiation, or lack of technical expertise. These errors may appear small during development but can lead to major regulatory concerns and delays during submission.
Many companies face avoidable challenges simply because the leachables strategy is not designed early or is not supported by a strong scientific justification.
Most Common Errors
Below are the most frequent mistakes observed in Leachable Screening for Parenteral Formulations projects:
- Starting leachables studies too late in development: Waiting until the final stages increases the risk of unexpected findings and costly rework.
- Using non-validated analytical methods: Inaccurate or poorly validated methods can lead to unreliable results and regulatory objections.
- Ignoring unknown peaks in chromatographic data: Even small unknown peaks must be investigated, especially for injectable products.
- Poor documentation of risk assessment: Regulatory agencies expect clear scientific justification for study design and testing strategy.
- Using unrealistic extraction conditions: Overly aggressive or poorly designed extraction conditions can generate misleading results.
- Not involving experienced analytical laboratories: Lack of technical expertise often leads to incorrect interpretation of data and delayed approvals.
Why Avoiding These Mistakes Matters
Avoiding these common errors in Leachable Screening for Parenteral Formulations can significantly improve submission success rates, reduce regulatory queries, and ensure that the product meets the highest safety and quality standards.
7: How Expertise Improves Leachable Screening Results
Expertise plays a critical role in the success of Leachable Screening for Parenteral Formulations. While advanced analytical instruments are essential, accurate interpretation of data is equally important. Without the right experience, even high-quality analytical results can lead to incorrect conclusions or unnecessary regulatory concerns.
An experienced analytical partner understands not only how to detect leachables, but also how to interpret complex analytical data in a way that is scientifically accurate and regulator-ready.
How Expertise Makes a Difference
A laboratory with strong experience in Leachable Screening for Parenteral Formulations can:
- Identify difficult unknown compounds: Experienced analysts can interpret complex mass spectra and distinguish between structurally similar compounds.
- Select the most appropriate analytical techniques: Choosing the right combination of LC-MS, GC-MS, and high-resolution methods improves detection accuracy.
- Reduce false positives and false negatives: Proper method development and data interpretation help avoid misleading results.
- Provide scientifically justified risk assessments: Experienced teams can evaluate whether detected leachables truly present a safety risk.
- Support regulatory documentation and submissions: Well-structured reports and clear scientific justifications improve the chances of smooth regulatory approval.
Why This Is Especially Important for Complex Formulations
Expertise becomes even more important when working with complex drug products such as:
- Biologics
- Liposomal formulations
- Long-acting injectable drugs
- High-sensitivity formulations
- Drug-device combination products
In these cases, Leachable Screening for Parenteral Formulations requires not only advanced analytical tools but also deep scientific understanding and regulatory experience.
8: Best Practices for Designing Leachable Screening for Parenteral Formulations
Best practices for designing Leachable Screening for Parenteral Formulations focus on early planning, scientific justification, and the use of reliable analytical strategies. A structured approach helps ensure accurate results, smoother regulatory submissions, and improved patient safety.
Companies that follow a well-defined strategy from the beginning are far more likely to avoid delays, unexpected findings, and regulatory objections.
Recommended Approach
Below are the most important best practices for Leachable Screening for Parenteral Formulations:
- Start leachables planning during early formulation development: Early planning reduces the risk of late-stage surprises and costly rework.
- Perform risk assessment before selecting analytical methods: A risk-based approach ensures that testing focuses on the most critical areas.
- Use both simulated and exaggerated extraction studies: Combining both approaches provides realistic as well as worst-case scenario data.
- Apply high-sensitivity analytical techniques: Advanced methods such as LC-MS, GC-MS, and high-resolution mass spectrometry improve detection accuracy.
- Confirm unknown compounds with proper structural analysis: Structural confirmation improves data reliability and regulatory acceptance.
- Maintain clear and well-structured documentation: Proper documentation strengthens the scientific credibility of the study.
- Involve regulatory and analytical experts early in the process: Expert involvement improves strategy design and increases submission success rates.
Why Following Best Practices Matters
Following these best practices in Leachable Screening for Parenteral Formulations ensures not only scientific reliability but also strong regulatory compliance. A well-designed screening strategy ultimately protects patient safety, improves product quality, and supports faster regulatory approval.
Conclusion:
Leachable Screening for Parenteral Formulations is not just an analytical requirement — it is a critical patient-safety activity that directly affects regulatory approval and product success. A well-designed screening strategy should begin early, use a science-based risk assessment, apply advanced analytical techniques, and include proper identification of unknown compounds.
Companies that invest in a structured and expert-driven approach to Leachable Screening for Parenteral Formulations can reduce regulatory risks, improve product safety, and accelerate approval timelines. Most importantly, a robust leachables strategy ensures that injectable drug products meet the highest standards of quality and safety.
Frequently Asked Questions:
Leachable Screening for Parenteral Formulations is a systematic analytical study used to detect, identify, and quantify chemical compounds that migrate from packaging materials or manufacturing components into injectable drug products. Since parenteral drugs are administered directly into the bloodstream, even very low levels of contaminants must be evaluated carefully. This screening helps ensure patient safety, product stability, and regulatory compliance. It also supports risk assessment during drug development. A well-designed screening strategy reduces the chances of regulatory delays.
Leachable screening is more critical for parenteral products because these drugs bypass the body’s natural protective systems such as the digestive tract. Any contaminant present in the product directly enters systemic circulation, which increases safety risks. Even trace-level leachables can affect drug stability or cause toxicological concerns. Regulatory agencies therefore apply stricter guidelines for injectable products compared to oral dosage forms. Proper screening helps avoid safety risks and regulatory issues. It also ensures long-term product quality.
Common sources of leachables include packaging materials such as rubber stoppers, glass vials, syringe components, and vial coatings. Manufacturing components like tubing, connectors, filters, and seals can also release chemical compounds into the product. Drug-device combination systems such as pre-filled syringes and cartridges are another major source. Storage conditions such as high temperature and long-term storage may increase leachable formation. Identifying these sources early helps design a better screening strategy. This also improves regulatory acceptance.
Several advanced analytical techniques are used to detect and identify leachables accurately. LC-MS is commonly used for semi-volatile and non-volatile compounds, while GC-MS is ideal for volatile compounds. ICP-MS is used for detecting elemental impurities such as metals. High-resolution mass spectrometry helps identify unknown compounds with better accuracy. These techniques are often used together to ensure complete screening coverage. Using the right analytical methods improves both data quality and regulatory compliance.
Leachable screening should begin as early as the formulation development stage rather than at the final stage of product development. Early planning allows companies to identify potential risks before they become major regulatory issues. It also helps in selecting suitable packaging materials and container-closure systems. Starting early reduces the chances of unexpected findings during submission. It also improves the overall quality of the product. A proactive approach saves both time and cost in the long term.
Reference
- Gusain AS, Chandra S, Araújo IM, de Lima JP, Coutinho HD. Scientifically supported best practices in leachable screening studies for pharmaceutical and parenteral drug products. Future Pharmacology. 2025 Apr 12;5(2):18.https://www.mdpi.com/2673-9879/5/2/18
- Scatena A, Toth S, Chenevert K. Exploring Extractable and Leachable Testing Strategies for Parenterals [Internet].https://www.ondrugdelivery.com/wp-content/uploads/2024/01/156_2024_Jan_PFS_Gateway_Analytical.pdf
- Houston CT, Rodrigues AD, Smith BB, Wang T, Richardson M. Principles for management of extractables and leachables in ophthalmic drug products. PDA Journal of Pharmaceutical Science and Technology. 2022 May 1;76(3):278-94.https://journal.pda.org/content/76/3/278.abstract
- Paskiet D, Jenke D, Ball D, Houston C, Norwood DL, Markovic I. The Product Quality Research Institute (PQRI) leachables and extractables working group initiatives for parenteral and ophthalmic drug product (PODP). PDA Journal of Pharmaceutical Science and Technology. 2013 Sep 1;67(5):430-47.https://journal.pda.org/content/67/5/430.short
- Smith EJ, Paskiet DM, Tullo EJ. The management of extractables and leachables in pharmaceutical products. InParenteral Medications, Fourth Edition 2019 Jul 19 (pp. 535-573). CRC Press.https://www.taylorfrancis.com/chapters/edit/10.1201/9780429201400-29/management-extractables-leachables-pharmaceutical-products-edward-smith-diane-paskiet-erica-tullo


