
Introduction:
A Dexamethasone Injectable CRO plays a critical role in helping pharmaceutical companies successfully develop generic and complex injectable formulations of dexamethasone. Because dexamethasone exists in multiple injectable forms—including solutions, suspensions, and polymer-based systems—development requires strong analytical expertise, regulatory understanding, and formulation experience.
Pharmaceutical companies often rely on specialized CRO partners to reduce technical risks, accelerate timelines, and ensure regulatory compliance. This article explains how CRO services support dexamethasone injectable development, the key technical challenges involved, and why expertise matters—especially for complex systems such as PLGA-based formulations.
Share via:
Summary:
- A Dexamethasone Injectable CRO provides end-to-end development support including formulation, analytical method development, impurity profiling, and stability studies.
- Generic development is technically challenging due to multiple dosage forms, complex excipients, and polymer-based systems such as PLGA.
- Regulatory success depends heavily on robust impurity control, stability-indicating methods, and reproducible formulation development.
- A specialized CRO like ResolveMass Laboratories Inc. brings deep technical expertise in injectable development and complex impurity characterization.
- Polymer-based systems like the branded product Ozurdex make generic development even more complex, requiring specialized analytical and formulation knowledge.
1: What Does a Dexamethasone Injectable CRO Do?
A Dexamethasone Injectable CRO supports pharmaceutical companies through the complete development lifecycle—from early formulation to regulatory submission.
In simple terms, the CRO ensures that the injectable product is stable, safe, compliant, and scientifically justified before it reaches regulatory authorities.
Core Services Offered
Most specialized CROs provide:
- Pre-formulation studies
- Formulation development (solution, suspension, or complex systems)
- Analytical method development and validation
- Impurity profiling and degradation studies
- Stability studies under ICH conditions
- Extractables and leachables assessment
- Support for regulatory documentation (ANDA/CTA/DMF)
These services significantly reduce the development burden on pharmaceutical companies and help avoid costly delays later in the process.
2: Why Dexamethasone Injectable Development Is Technically Challenging
A Dexamethasone Injectable CRO is required because dexamethasone injectable products are far more complex than standard formulations. The molecule is highly sensitive to formulation variables such as pH, solvent system, excipients, and processing conditions. Even small formulation changes can significantly affect product stability, impurity formation, and long-term shelf life.
Because injectable products are directly administered into the body, regulatory agencies expect extremely high levels of quality, purity, and stability. This is why pharmaceutical companies often rely on a specialized ResolveMass Laboratories Inc. to reduce development risks and generate reliable, submission-ready data.
Key Technical Challenges in Dexamethasone Injectable Development
A specialized Dexamethasone Injectable CRO plays a critical role in overcoming multiple technical challenges that arise during development.
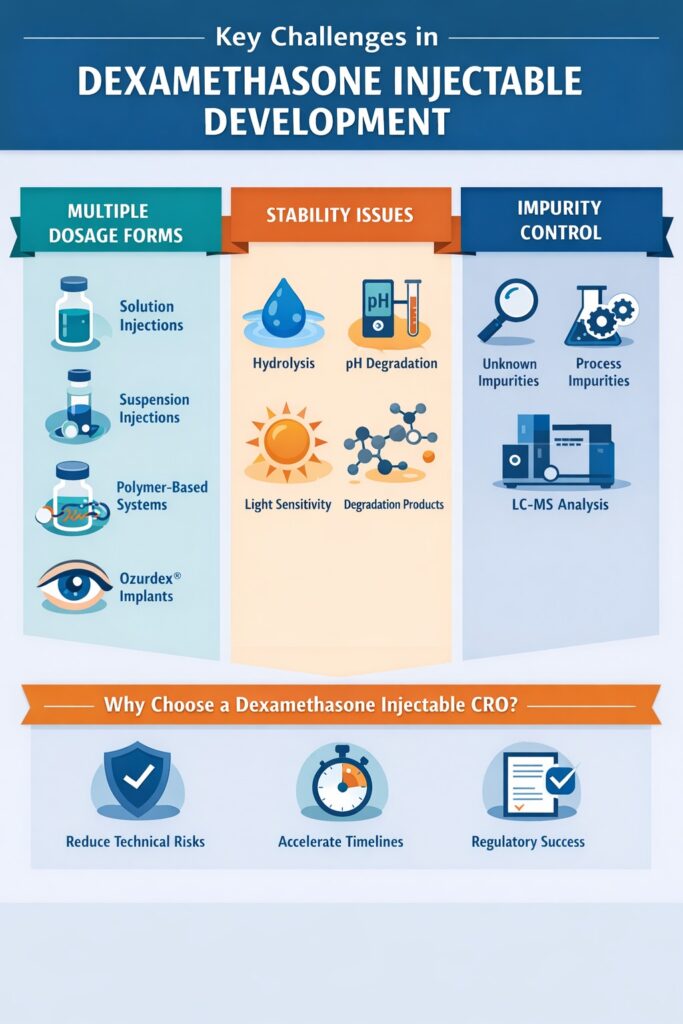
1. Multiple Dosage Forms
Dexamethasone is available in several injectable forms, and each dosage form requires a completely different development strategy:
- Dexamethasone sodium phosphate (solution injection)
- Dexamethasone acetate (suspension injection)
- Controlled-release polymer-based systems
- Long-acting implants such as Ozurdex
Because each form behaves differently in terms of solubility, stability, and degradation pathways, development becomes significantly more complex than a standard injectable product.
2. Stability Challenges
Dexamethasone injectables are highly sensitive to environmental and formulation conditions, which makes stability studies a critical part of development.
The most common stability challenges include:
- Hydrolysis in aqueous media
- pH-dependent degradation pathways
- Light sensitivity in certain formulations
- Excipient-drug compatibility issues
- Formation of new degradation products during long-term storage
A knowledgeable Dexamethasone Injectable CRO can design stability-indicating analytical methods early in development to prevent costly failures later.
3. Impurity Control and Regulatory Expectations
One of the biggest reasons companies work with a Dexamethasone Injectable CRO is the complexity of impurity identification and control.
Regulatory authorities require complete impurity characterization, including:
- Known pharmacopoeial impurities
- Unknown degradation products
- Process-related impurities
- Stability-related impurities formed during storage
- Structural confirmation of unknown peaks
This level of analytical depth typically requires advanced LC-MS/MS expertise and significant experience in impurity profiling.
Why Companies Prefer a Dexamethasone Injectable CRO Instead of In-House Development
Because of the combination of formulation complexity, stability risks, and strict regulatory requirements, many pharmaceutical companies choose to work with a specialized Dexamethasone Injectable CRO rather than developing the product completely in-house.
Working with an experienced CRO not only reduces technical risk but also accelerates development timelines and improves the chances of successful regulatory approval.
3: How a Dexamethasone Injectable CRO Supports Formulation Development
A Dexamethasone Injectable CRO supports formulation development by first defining the target product profile and then designing a scientifically robust formulation strategy based on solubility, stability, and regulatory requirements. This structured approach significantly reduces development risk and improves the chances of successful regulatory approval.
Specialized organizations such as ResolveMass Laboratories Inc. typically combine formulation expertise with advanced analytical capabilities, which is critical for sensitive molecules like dexamethasone.
Typical Formulation Development Workflow Used by a Dexamethasone Injectable CRO
A specialized Dexamethasone Injectable CRO follows a step-by-step workflow to ensure the formulation remains stable, reproducible, and regulatory-compliant. Instead of trial-and-error development, the process is designed to predict potential risks early.
Step-by-Step Workflow
| Development Step | What the Dexamethasone Injectable CRO Does | Why It Matters |
|---|---|---|
| Pre-formulation studies | Solubility studies, pH stability testing, excipient compatibility studies | Prevents long-term degradation and stability failures |
| Prototype formulation development | Multiple formulation trials using different excipients and solvent systems | Helps identify the most stable and reproducible composition |
| Analytical screening | Early impurity profiling and degradation monitoring using advanced analytical tools | Reduces regulatory risk and avoids late-stage surprises |
| Stability screening | Accelerated stability studies and stress testing under multiple conditions | Predicts shelf life early in development |
| Final formulation optimization | Selection of the most robust formulation based on stability and impurity profile | Ensures consistent manufacturing and regulatory success |
Why This Structured Approach Matters in Dexamethasone Injectable Development
Formulation development for injectable corticosteroids is far more complex than standard products. Even small changes in formulation composition can lead to:
- New degradation products
- Reduced stability
- Regulatory rejection due to impurity concerns
- Manufacturing reproducibility issues
This is why pharmaceutical companies often rely on a specialized Dexamethasone Injectable CRO such as ResolveMass Laboratories Inc. to design a structured development strategy rather than relying on trial-based formulation work.
How a Dexamethasone Injectable CRO Reduces Development Failures
An experienced Dexamethasone Injectable CRO reduces the risk of formulation failure by integrating analytical testing directly into formulation development. Instead of testing the product only at the end, stability and impurity monitoring start from the very first formulation trials.
This approach helps companies:
- Improve the chances of regulatory approval
- Avoid long-term stability failures
- Detect degradation pathways early
- Select the most robust formulation faster
- Reduce development time
4: Role of Analytical Expertise in Dexamethasone Injectable CRO Projects
Analytical expertise is one of the most critical components of any Dexamethasone Injectable CRO project because regulatory approval depends primarily on the quality, accuracy, and reliability of analytical data. Regulatory agencies do not approve injectable products based only on formulation—they require scientifically proven evidence of stability, impurity control, and long-term product safety.
This is why pharmaceutical companies prefer to work with specialized organizations such as ResolveMass Laboratories Inc. that combine real laboratory experience with advanced analytical technologies.
Why Analytical Expertise Is Essential in a Dexamethasone Injectable CRO
A Dexamethasone Injectable CRO must demonstrate strong analytical capabilities from the earliest stage of development because injectable products require much stricter quality standards compared to oral formulations.
Regulatory agencies typically expect:
- Highly sensitive stability-indicating methods
- Complete impurity identification
- Accurate assay and degradation monitoring
- Reproducible analytical validation data
- Clear scientific justification for every analytical result
Without strong analytical expertise, even a well-designed formulation can fail during regulatory submission.
Critical Analytical Services Offered by a Dexamethasone Injectable CRO
A specialized Dexamethasone Injectable CRO provides a wide range of advanced analytical services that support formulation development, stability studies, and regulatory documentation.
Core Analytical Services
- Stability-indicating HPLC/UPLC method development
- LC-MS/MS based impurity identification and structural elucidation
- Forced degradation studies under multiple stress conditions
- Residual solvent analysis
- Particle size analysis (especially for suspension injectables)
- Method validation according to ICH guidelines
These services ensure that the injectable product is scientifically characterized and fully compliant with global regulatory expectations.
How Analytical Expertise Reduces Regulatory Risk
One of the biggest advantages of working with a specialized Dexamethasone Injectable CRO is the ability to identify potential risks early in development. Instead of discovering problems during stability studies or regulatory review, experienced CROs detect issues at the analytical stage itself.
Organizations such as ResolveMass Laboratories Inc. typically focus on early-stage impurity profiling, stability-indicating method development, and degradation pathway studies to reduce the risk of:
- Regulatory rejection
- Unknown impurity concerns
- Stability failures
- Delays in submission timelines
- Additional analytical redevelopment
Why Analytical Expertise Matters More for Injectable Products
Analytical expertise is even more important for injectables because these products are administered directly into the body. This means regulatory agencies require extremely high confidence in:
- Product purity
- Long-term stability
- Absence of unknown impurities
- Reproducibility of analytical methods
- Scientific justification of degradation behavior
A specialized Dexamethasone Injectable CRO ensures that every analytical result is reliable, reproducible, and ready for global regulatory submission.
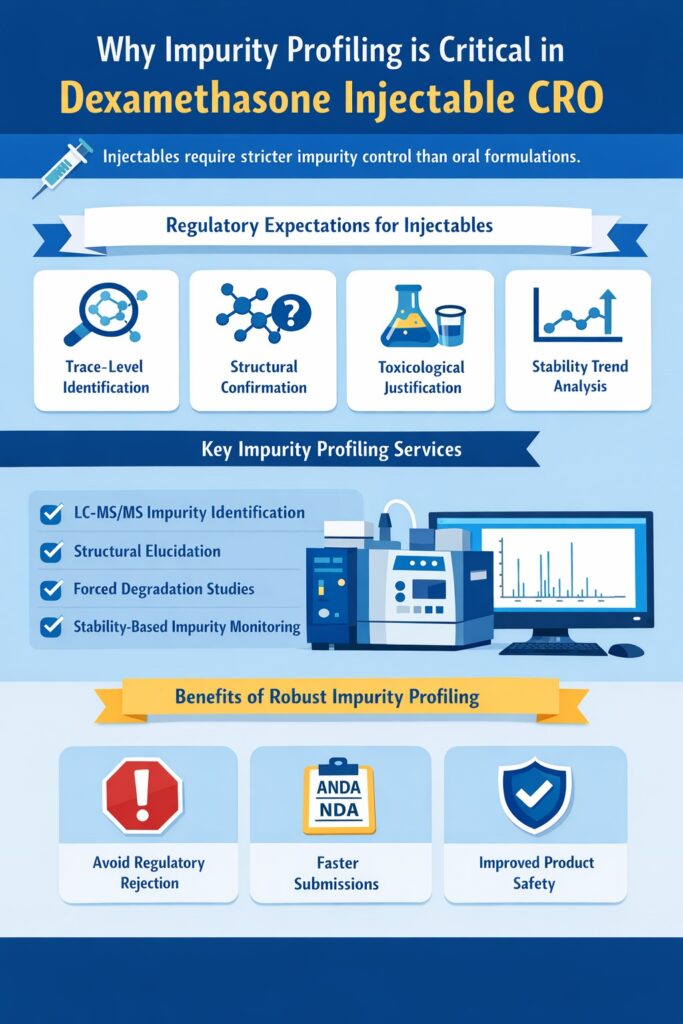
5: Why Impurity Profiling Is Critical in a Dexamethasone Injectable CRO
Impurity profiling is one of the most critical responsibilities of a Dexamethasone Injectable CRO because injectable products are regulated far more strictly than oral formulations. Since these products are administered directly into the body, regulatory agencies require extremely detailed impurity identification, structural confirmation, and stability-based impurity control.
Specialized analytical organizations such as ResolveMass Laboratories Inc. focus heavily on impurity profiling because even trace-level impurities can lead to regulatory questions or delays if they are not properly identified and justified.
Why Regulatory Agencies Focus So Strongly on Impurities in Injectables
Regulatory agencies place special emphasis on impurity profiling in injectable products because there is no natural biological barrier (such as the digestive system) to reduce exposure. This means regulators expect a Dexamethasone Injectable CRO to provide complete scientific justification for every impurity detected during development and stability studies.
Typical regulatory expectations include:
- Identification of impurities at extremely low levels
- Structural confirmation of unknown peaks
- Toxicological justification when required
- Stability-based impurity trend analysis
- Clear differentiation between process impurities and degradation products
Without this level of analytical depth, regulatory approval becomes much more difficult.
Role of a Dexamethasone Injectable CRO in Advanced Impurity Identification
A specialized Dexamethasone Injectable CRO uses advanced analytical techniques—especially LC-MS/MS—to detect and identify impurities that cannot be confirmed using conventional analytical methods.
Key impurity profiling activities include:
- Identification of unknown degradation products
- Forced degradation studies to understand degradation pathways
- Structural elucidation using high-resolution mass spectrometry
- Stability-related impurity monitoring
- Qualification of impurities according to regulatory guidelines
Organizations such as ResolveMass Laboratories Inc. often combine real laboratory experience with advanced mass spectrometry expertise to provide reliable impurity data that is fully suitable for regulatory submissions.
How Strong Impurity Profiling Improves Regulatory Success
One of the main reasons pharmaceutical companies work with a Dexamethasone Injectable CRO is to reduce regulatory risk. When impurities are identified early and supported with strong scientific justification, companies can avoid:
- Regulatory rejection due to unknown peaks
- Delays in ANDA or NDA submission
- Repeated analytical method redevelopment
- Stability failures caused by unidentified degradation products
In short, strong impurity profiling does not just improve product quality—it significantly increases the probability of successful regulatory approval.
6: Complexity of PLGA Polymer in Dexamethasone Injectable Generic Development
A Dexamethasone Injectable CRO becomes even more important when the product involves complex polymer systems such as PLGA (poly-lactic-co-glycolic acid).
PLGA-based injectables are not traditional formulations—they are drug-polymer delivery systems, which means both the drug and the polymer must be carefully controlled.
Why PLGA Makes Dexamethasone Injectable Generic Development Difficult
PLGA-based dexamethasone products introduce multiple technical challenges that significantly increase development complexity. This is one of the strongest reasons companies rely on a specialized Dexamethasone Injectable CRO instead of developing the product entirely in-house.
1. Polymer Variability Creates Major Development Challenges
PLGA is not a single standardized material. Different polymer grades behave very differently, and even small changes in polymer characteristics can significantly affect the drug release profile.
Key parameters that vary between PLGA polymers:
- Molecular weight
- Lactide:glycolide ratio
- End-group chemistry (acid-terminated vs ester-terminated)
- Polymer degradation rate
- Residual monomers and impurities
Because of this variability, a Dexamethasone Injectable CRO must perform detailed polymer characterization before formulation development even begins.
2. Complex Drug Release Mechanism
Unlike conventional injectable solutions or suspensions, PLGA-based systems rely on multiple simultaneous mechanisms. This makes it extremely difficult to match the reference product in generic development.
The release mechanism typically involves:
- Polymer degradation
- Drug diffusion through the polymer matrix
- Matrix erosion over time
- Environmental factors such as pH and temperature
A specialized Dexamethasone Injectable CRO must understand these mechanisms in detail to design a formulation that provides a similar release profile.
3. Advanced Analytical Requirements for PLGA-Based Products
Developing PLGA-based dexamethasone injectables requires far more advanced analytical techniques compared to standard formulations. This is another reason pharmaceutical companies work with specialized organizations such as ResolveMass Laboratories Inc..
Typical analytical requirements include:
- Polymer characterization and molecular weight analysis
- Drug release profile comparison studies
- Degradation pathway analysis
- Stability studies under multiple environmental conditions
- Impurity profiling of both drug and polymer
This level of analytical work requires deep experience in mass spectrometry, polymer chemistry, and long-acting injectable development.
Why Companies Rely on a Dexamethasone Injectable CRO for PLGA-Based Generics
Because of the complexity of PLGA systems, many pharmaceutical companies rely on a specialized Dexamethasone Injectable CRO when developing generic versions of long-acting injectable products. A well-known example is Ozurdex, which uses a biodegradable polymer system to deliver dexamethasone over an extended period.
Working with an experienced CRO helps companies:
- Accelerate the overall development timeline
- Reduce formulation development risk
- Improve drug-release profile matching
- Identify polymer-related impurities early
- Generate regulatory-ready analytical data

7: How a Dexamethasone Injectable CRO Reduces Regulatory Risk
A Dexamethasone Injectable CRO helps pharmaceutical companies avoid regulatory delays by designing development studies that align with global guidelines from the beginning.
How CROs Improve Regulatory Success
They ensure:
- Stability studies follow ICH guidelines
- Analytical methods are stability-indicating
- Impurity thresholds meet regulatory limits
- Data is generated in a submission-ready format
- Documentation supports ANDA or regulatory filings
Instead of fixing issues after submission, companies can avoid problems early by working with a specialized CRO.
8: Why Work With a Specialized Dexamethasone Injectable CRO Instead of an In-House Team?
A Dexamethasone Injectable CRO often provides better results than in-house development because of focused experience in complex injectable products.
Key Advantages
Faster development timelines
CROs already have optimized analytical methods and development workflows.
Lower analytical risk
Experienced scientists can quickly identify degradation pathways and impurities.
Cost efficiency
Instead of investing in new equipment and expertise, companies can outsource development to specialists.
Better regulatory readiness
CRO-generated data is typically more structured and submission-ready.
9: Why ResolveMass Laboratories Inc. Is a Reliable Dexamethasone Injectable CRO
ResolveMass Laboratories Inc. focuses on complex analytical challenges, impurity characterization, and injectable development support. This technical expertise makes the company a reliable Dexamethasone Injectable CRO for pharmaceutical companies working on generic development.
What Makes the Company Stand Out
- Deep expertise in impurity profiling
- Advanced LC-MS/MS capabilities
- Experience with complex injectable products
- Strong understanding of regulatory expectations
- Focus on scientific problem-solving rather than routine testing
This level of specialization helps pharmaceutical companies reduce risk and accelerate development timelines.
10: When Should You Work With a Dexamethasone Injectable CRO?
A Dexamethasone Injectable CRO should be involved early in the development process, especially if the product includes complex formulation challenges.
You Should Consider a CRO If:
- You are developing a generic dexamethasone injection
- You are working with suspension or polymer-based systems
- Unknown impurities are appearing in stability studies
- Analytical methods are not stability-indicating
- You want to accelerate development timelines
Early collaboration reduces costly delays later in the project.
Conclusion:
A Dexamethasone Injectable CRO is essential for pharmaceutical companies developing generic injectable formulations because of the technical complexity, strict regulatory expectations, and high analytical requirements involved. From formulation development to impurity profiling and stability studies, CRO support significantly increases the chances of successful regulatory approval.
The complexity becomes even greater when polymer-based systems such as PLGA are involved, especially for products similar to Ozurdex. In such cases, working with an experienced partner like ResolveMass Laboratories Inc. can help pharmaceutical companies reduce risk, accelerate development, and achieve high-quality, submission-ready data.
Frequently Asked Questions:
A Dexamethasone Injectable CRO is a contract research organization that provides specialized services for developing Dexamethasone injectable products. These services typically include formulation development, analytical method development, impurity profiling, stability studies, and regulatory documentation support.
Pharmaceutical companies use a Dexamethasone Injectable CRO because injectable drug development is more complex than oral dosage forms. A specialized CRO helps reduce stability risks, identify impurities early, develop reliable analytical methods, and prepare regulatory-ready data for faster approvals.
A Dexamethasone Injectable CRO typically provides formulation development, analytical method development and validation, forced degradation studies, stability studies, impurity identification, extractables and leachables (E&L) testing, and regulatory documentation support.
A Dexamethasone Injectable CRO supports regulatory approval by generating high-quality, regulatory-ready data. This includes ICH-compliant stability studies, impurity identification reports, analytical method validation, and technical documentation that meets global regulatory expectations.
Reference
- London NJ, Chiang A, Haller JA. The dexamethasone drug delivery system: indications and evidence. Advances in therapy. 2011 May;28(5):351-66.https://link.springer.com/article/10.1007/s12325-011-0019-z
- Yuen MV, Gianturco SL, Pavlech LL, Storm KD, Mattingly AN. Dexamethasone sodium phosphate: Summary Report.https://archive.hshsl.umaryland.edu/entities/publication/13b8ec2e-af1c-4df7-a478-3684e0f48c82
- Mosoro E, Wilson AN, Homer CS, Vogel JP. Assessing the quality of antenatal corticosteroids in low-and middle-income countries: A systematic review. PloS one. 2020 Dec 3;15(12):e0243034.https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0243034
- Khabale MB, Abale ML, Kasande S. “Key Success Factors of Value Chain Management of Injectable Formulations”: An Expert Opinion Survey. Anveshak International Journal of Management (AIJM). 2025 Jul 1;14(2).https://search.ebscohost.com/login.aspx?direct=true&profile=ehost&scope=site&authtype=crawler&jrnl=22788913&AN=187271579&h=WZcLqtztIasV43cmtZ%2BH32vSi%2FdDDIB5abcNRG4judxtTXe%2BgwLf9A8klBsEzfLZlgRQAl93jzcNnQoxABucXg%3D%3D&crl=c
- Liu H, Liu Y, Tian Z, Li J, Li M, Zhao Z. Coordinating macrophage targeting and antioxidation by injectable nanocomposite hydrogel for enhanced rheumatoid arthritis treatment. ACS Applied Materials & Interfaces. 2024 Jul 10;16(29):37656-68.https://pubs.acs.org/doi/abs/10.1021/acsami.4c06840


