Introduction to Dexamethasone Phosphate vs Acetate
When comparing Dexamethasone Phosphate vs Acetate, the key differences involve solubility, pharmacokinetics, ester hydrolysis rate, and formulation applications. Although both derivatives eventually release the same active corticosteroid, their chemical modifications affect how the drug dissolves, how quickly it becomes active, and how long it remains effective in the body. Because of these differences, each form is used for specific pharmaceutical and clinical purposes.
Both compounds are prodrug forms of dexamethasone, designed to improve drug delivery and formulation performance. The base corticosteroid structure remains the same, but the attached ester group changes the compound’s physical and biochemical behavior. These changes influence absorption, distribution, and drug release, resulting in different therapeutic profiles.
Understanding Dexamethasone Phosphate vs Acetate is important for pharmaceutical scientists, researchers, and drug manufacturers. Selecting the appropriate derivative helps ensure that formulations deliver the desired onset of action, stability, and therapeutic performance in pharmaceutical products.
Explore our expertise in specialized drug development: Learn more about Dexamethasone Injection Generic Development
Share via:
Structural Differences in Dexamethasone Phosphate vs Acetate
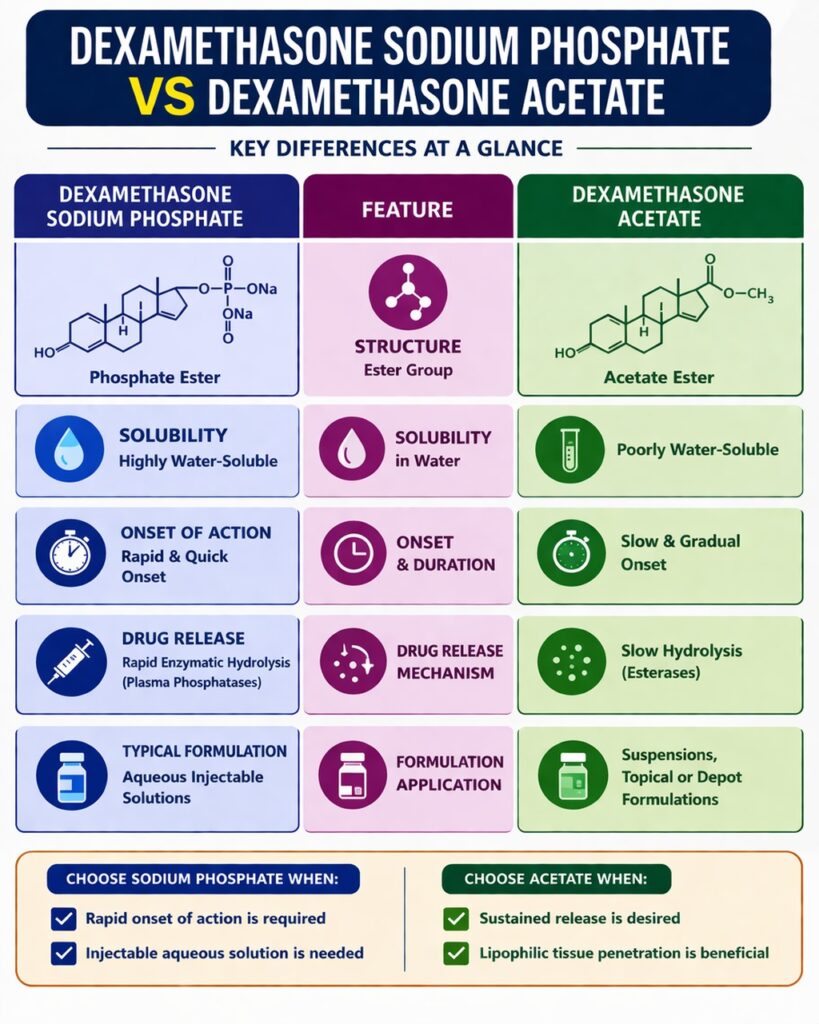
The main structural difference in Dexamethasone Phosphate vs Acetate comes from the ester group attached to the C21 hydroxyl position of the dexamethasone molecule. This chemical modification changes the physical and chemical properties of the steroid while keeping the main pharmacological activity unchanged. The ester group determines how the molecule interacts with water, lipids, and biological membranes. Because of this, it strongly affects formulation strategies and drug release behavior.
| Property | Dexamethasone Sodium Phosphate | Dexamethasone Acetate |
|---|---|---|
| Ester group | Phosphate ester | Acetate ester |
| Solubility | Highly water-soluble | Poorly water-soluble |
| Ionization | Ionic salt form | Non-ionic lipophilic ester |
| Hydrolysis rate | Rapid | Slow |
| Formulation type | Aqueous injectable solutions | Suspensions or topical formulations |
Phosphate esters are widely used in pharmaceutical chemistry because they greatly increase water solubility. When the compound forms an ionic sodium salt, it becomes easier to dissolve in aqueous pharmaceutical preparations. This property allows the drug to be used in intravenous or intramuscular injections. As a result, phosphate derivatives are commonly used in hospital and emergency settings.
Acetate esters, on the other hand, increase the lipophilicity of the molecule. Lipophilic compounds interact more easily with cell membranes and fatty tissues. However, this also reduces their ability to dissolve in water. Because of this property, acetate forms are often used in suspensions or topical formulations where slower drug release is beneficial.
Scientific studies on corticosteroid prodrugs show that phosphate ester modification greatly improves water solubility and allows rapid systemic availability after enzymatic cleavage. This is one reason why phosphate derivatives are preferred for injectable medications. Understanding these structural changes helps scientists predict how the compound will behave in pharmaceutical formulations.
Solubility Differences in Dexamethasone Phosphate vs Acetate
One of the most important differences in Dexamethasone Phosphate vs Acetate is water solubility. Dexamethasone Sodium Phosphate is highly soluble in water, while Dexamethasone Acetate has very limited water solubility. This difference strongly influences formulation design, administration route, and drug absorption. Because solubility determines how easily a drug dissolves in body fluids, it plays a major role in drug delivery.
Dexamethasone Sodium Phosphate
- Exists as a highly ionized sodium salt
- Easily dissolves in aqueous injection solutions
- Suitable for intravenous or intramuscular formulations
Because of its excellent water solubility, dexamethasone sodium phosphate can be used in sterile injectable medications. These formulations allow precise dosing and fast distribution through the bloodstream. The high solubility also helps prevent precipitation during administration. For these reasons, this derivative is widely used in clinical and hospital environments.
Optimize your steroid product profile: Discover our Dexamethasone Sodium Phosphate Formulation Services
Dexamethasone Acetate
- Poorly soluble in water
- Commonly used in suspensions or lipid-based systems
- Dissolves more slowly in biological fluids
The limited solubility of dexamethasone acetate requires special formulation approaches. Suspension systems keep the drug particles dispersed until they slowly dissolve after administration. Lipid-based formulations can also improve delivery in topical or localized treatments. These approaches allow the drug to release gradually over time.
Research shows that adding a phosphate group to corticosteroids can increase aqueous solubility by several orders of magnitude compared with lipophilic esters. This explains why phosphate derivatives are widely used in injectable therapies. In contrast, acetate derivatives are more suitable when slow drug release is desired.
Because of these differences, Dexamethasone Phosphate vs Acetate cannot be directly substituted in pharmaceutical formulations. Each compound requires a different formulation strategy to maintain stability and effectiveness.
Pharmacokinetic Differences Between Dexamethasone Phosphate vs Acetate
The pharmacokinetic behavior of Dexamethasone Phosphate vs Acetate mainly depends on solubility and the rate of ester hydrolysis. These factors control how quickly the active drug appears in the bloodstream and how long therapeutic levels remain. As a result, each derivative shows a different onset and duration of action.
Dexamethasone Sodium Phosphate
- Rapid systemic absorption
- Fast enzymatic conversion to active dexamethasone
- Quick onset of therapeutic effect
Once administered, dexamethasone sodium phosphate dissolves quickly in body fluids and enters systemic circulation. Enzymes called plasma phosphatases rapidly break the phosphate group, releasing active dexamethasone. This rapid conversion leads to a quick increase in drug concentration in the blood. Such pharmacokinetics make it ideal for conditions requiring immediate anti-inflammatory action.
Dexamethasone Acetate
- Slower absorption
- Gradual ester hydrolysis
- Sustained drug release
Dexamethasone acetate is absorbed more slowly because it is lipophilic and poorly soluble in water. Enzymes known as esterases slowly break the acetate group to release the active drug. This delayed conversion results in slower peak plasma levels. However, it also allows the drug to remain active for a longer period.
Studies show that water-soluble phosphate esters of corticosteroids are rapidly converted in plasma, leading to quick therapeutic action. In contrast, acetate esters undergo slower enzymatic hydrolysis, resulting in prolonged drug exposure. This difference explains why each derivative is used in different therapeutic situations.
Drug Release Mechanisms: Dexamethasone Phosphate vs Acetate
The release of active dexamethasone in Dexamethasone Phosphate vs Acetate depends mainly on enzymatic ester hydrolysis. This biochemical reaction converts the prodrug form into active dexamethasone. The speed of this process determines how fast the medication starts working. Therefore, the type of ester group strongly influences the drug release profile.
Phosphate Ester Hydrolysis
- Rapid cleavage by plasma phosphatases
- Immediate release of dexamethasone
- High systemic availability
Phosphate esters are designed to break down quickly after entering the bloodstream. This ensures that active dexamethasone becomes available shortly after administration. Because the conversion happens rapidly, the therapeutic effect begins quickly. This makes phosphate derivatives useful for treatments that require fast anti-inflammatory action.
Acetate Ester Hydrolysis
- Cleaved by esterases
- Slower hydrolysis rate
- Controlled drug release
Acetate esters break down more slowly in the body. Esterase enzymes gradually convert the prodrug into active dexamethasone. This slower process creates a sustained release effect. As a result, the drug remains active in the body for a longer period.
Research on corticosteroid ester derivatives shows that phosphate esters act as rapid-activation prodrugs, while lipophilic esters such as acetates function as slow-release reservoirs. These characteristics are important when designing drug delivery systems and predicting drug exposure.
Formulation Considerations: Dexamethasone Phosphate vs Acetate
When choosing between Dexamethasone Phosphate vs Acetate, pharmaceutical developers must consider several formulation factors. These include solubility, stability, release profile, and the intended route of administration. Each derivative behaves differently in pharmaceutical preparations, so the formulation must be carefully designed.
Key Factors
- Solubility requirements
- Desired release profile
- Stability of the ester bond
- Route of administration
- Tissue penetration
These factors determine whether a formulation should focus on rapid drug action or sustained release. For example, injectable drugs often require high solubility. In contrast, topical or depot formulations may benefit from slower release properties.
Typical Formulation Applications
| Application | Preferred Ester |
|---|---|
| Injectable aqueous solutions | Sodium phosphate |
| Rapid-acting systemic therapy | Sodium phosphate |
| Topical or depot formulations | Acetate |
| Sustained release formulations | Acetate |
Phosphate esters are typically chosen when fast systemic availability is required. Their high solubility allows easy formulation in aqueous injections. This makes them suitable for emergency treatments or hospital-based therapies.
Acetate esters are preferred when prolonged tissue exposure is beneficial. Their lipophilic nature allows the drug to remain in tissues longer. This property supports sustained therapeutic effects in localized treatments.
Partner with an expert team for your next injectable project: Learn about our Dexamethasone Injectable CRO Services
Stability and Hydrolysis Characteristics
Stability differences between Dexamethasone Phosphate vs Acetate arise from variations in ester chemistry and environmental sensitivity. These differences influence how the compounds behave during storage and after administration. Proper formulation design must consider these stability factors to maintain product quality.
Dexamethasone Sodium Phosphate
- Stable in aqueous solutions at controlled pH
- Rapid enzymatic hydrolysis in biological fluids
- Minimal precipitation because of high solubility
Because of its high solubility, dexamethasone sodium phosphate usually remains fully dissolved in aqueous pharmaceutical formulations. Maintaining an appropriate pH helps preserve chemical stability during storage. After administration, enzymes quickly convert it to active dexamethasone.
Dexamethasone Acetate
- More stable in non-aqueous environments
- Slower esterase-mediated hydrolysis
- Risk of precipitation in water-based systems
Dexamethasone acetate is more stable in lipid-based or suspension formulations. Its low water solubility means it can precipitate if not properly formulated. However, well-designed suspension systems can maintain stable drug distribution until administration.
Tissue Penetration Differences in Dexamethasone Phosphate vs Acetate
Another important difference in Dexamethasone Phosphate vs Acetate is tissue penetration. Dexamethasone acetate generally has higher membrane permeability because of its lipophilic nature. Lipophilic molecules interact easily with the lipid bilayers that form biological cell membranes.
Because of this property, acetate esters:
- Cross biological membranes more easily
- Stay longer in lipid-rich tissues
- Provide localized drug retention
These characteristics make dexamethasone acetate suitable for topical or localized treatments. The drug can remain in tissues and release gradually over time, producing longer-lasting effects.
In contrast, phosphate esters are more hydrophilic and remain mainly in aqueous compartments before hydrolysis. Once the ester is cleaved and dexamethasone is released, the active drug can then distribute throughout tissues.
Choosing Between Dexamethasone Phosphate vs Acetate in Drug Development
Selecting between Dexamethasone Phosphate vs Acetate depends on the therapeutic objective and the desired pharmacokinetic profile. Drug developers must evaluate how quickly the medication should act and how long its effects should last. The route of administration also plays an important role in this decision.
Select Sodium Phosphate When
- Rapid onset of action is required
- High aqueous solubility is needed
- Injectable formulations are required
These conditions often occur in acute medical situations where fast therapeutic response is essential.
Select Acetate When
- Sustained release is desired
- Lipophilic tissue penetration is beneficial
- Suspension formulations are acceptable
Acetate derivatives are valuable when prolonged drug exposure is necessary. Their slower hydrolysis allows long-lasting therapeutic activity.
Organizations such as ResolveMass Laboratories Inc. support pharmaceutical developers by providing analytical testing, compound characterization, and purity verification for steroid derivatives. These services help ensure regulatory compliance and consistent pharmaceutical quality.
Conclusion
The differences between Dexamethasone Phosphate vs Acetate are mainly related to ester chemistry, solubility, and the rate of enzymatic hydrolysis. While both derivatives ultimately release the same active corticosteroid, their pharmaceutical behavior is quite different. These differences affect how the drug is formulated, how quickly it acts, and how long its effects last in the body.
When comparing Dexamethasone Phosphate vs Acetate, the phosphate form offers excellent water solubility and rapid systemic activation. Because of these properties, it is widely used in injectable formulations and treatments that require a fast therapeutic response. Its rapid conversion to active dexamethasone makes it especially useful in hospital and emergency settings.
In contrast, dexamethasone acetate is more lipophilic and undergoes slower enzymatic hydrolysis. This results in a longer duration of action and sustained drug release in tissues. For this reason, it is often used in topical, depot, or suspension-based formulations where prolonged therapeutic activity is beneficial.
For pharmaceutical scientists and drug developers, understanding Dexamethasone Phosphate vs Acetate is important when designing effective drug delivery systems. Choosing the correct derivative helps optimize formulation stability, pharmacokinetic performance, and therapeutic outcomes in pharmaceutical products.
For analytical support or research collaboration, Contact ResolveMass Today.
Frequently Asked Questions (FAQs)
The main difference is solubility and drug release speed. Dexamethasone Sodium Phosphate dissolves easily in water and converts quickly into active dexamethasone in the bloodstream. Dexamethasone Acetate is more lipophilic and releases the active drug slowly. Because of this, the phosphate form acts faster while the acetate form provides longer-lasting effects.
Dexamethasone acetate is a corticosteroid medication used to reduce inflammation and suppress immune responses in the body. It is commonly used in topical creams, eye preparations, and injectable suspensions to treat conditions such as skin inflammation, joint pain, and allergic reactions. Because it is more lipophilic, it often provides a slower and longer-lasting therapeutic effect. This makes it useful in treatments where sustained anti-inflammatory action is required.
Dexamethasone sodium phosphate is given to quickly control inflammation, allergic reactions, and certain immune-related conditions. It is highly water-soluble, which allows it to be administered through intravenous or intramuscular injections. After administration, it rapidly converts into active dexamethasone in the body. This fast action makes it useful in emergency care and hospital treatments.
Another common name for dexamethasone acetate is dexamethasone 21-acetate. This name refers to the acetate ester attached to the 21st carbon position of the dexamethasone molecule. It may also appear in pharmaceutical literature simply as Dex Acetate in certain formulations. The compound is widely used in pharmaceutical preparations designed for slower drug release.
Dexamethasone acetate is generally considered more potent than hydrocortisone in terms of anti-inflammatory activity. It can produce stronger effects at lower doses compared with hydrocortisone. However, “better” depends on the medical situation, as hydrocortisone may be preferred for milder conditions or when a shorter-acting steroid is needed. Doctors choose between them based on the patient’s condition and treatment goals.
Yes, dexamethasone sodium phosphate is sometimes administered during surgical procedures. It is often used to help prevent postoperative nausea, reduce inflammation, and support recovery after anesthesia. Because it works quickly, it can provide rapid therapeutic effects during or immediately after surgery. Its injectable form makes it convenient for use in operating rooms.
Yes, dexamethasone acetate is a type of corticosteroid, which is a class of steroid medications. It is derived from dexamethasone and works by reducing inflammation and suppressing immune system activity. The acetate group modifies the drug to change how it dissolves and releases in the body. This modification allows it to provide longer-lasting therapeutic effects in certain formulations.
Reference:
- Al-Owaidi, M. F., Alkhafaji, S. L., & Mahood, A. M. (2021). Quantitative determination of dexamethasone sodium phosphate in bulk and pharmaceuticals at suitable pH values using the spectrophotometric method. Journal of Advanced Pharmaceutical Technology & Research, 12(4), 378–383. https://doi.org/10.4103/japtr.japtr_6_21
- U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2009). Pharmacology review(s): Ozurdex (dexamethasone intravitreal implant), NDA 022315. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2009/022315s000_PharmR.pdf



