Introduction
Leuprolide Depot Generic Drug Development Services play a critical role in modern pharmaceutical development, especially for complex injectable products like Lupron Depot® based on PLGA microspheres. These formulations are not simple generics, as they require a deep understanding of both formulation science and drug delivery behavior. Achieving equivalence with the reference listed drug (RLD) involves more than matching ingredients—it requires matching performance.
Unlike traditional dosage forms, depot injections release the drug slowly over time. This makes the development process more demanding, as scientists must carefully control how the drug behaves inside the body. The interaction between polymer, drug, and environment becomes extremely important.
Another major challenge is maintaining the stability of leuprolide acetate within the polymer system. Factors such as moisture, temperature, and pH can affect how the drug degrades over time. Because of this, stability studies are a key part of successful development.
Explore the full scope of the approval process: Learn more about the Generic Drug Development Process for ANDA
Share via:
🔍 Summary
- Leuprolide Depot Generic Drug Development Services require deep reverse engineering of Lupron Depot® with a strong focus on PLGA polymer characterization and microsphere architecture.
- PLGA attributes (MW, lactide:glycolide ratio, end-group chemistry) directly control drug release kinetics and must match the reference product.
- Critical analytical techniques such as NMR, GPC, DSC, SEM, and in vitro release testing are essential for ANDA success.
- Initial burst release and long-term release phases must be precisely controlled to achieve bioequivalence.
- IVIVC (in vitro–in vivo correlation) is a regulatory expectation and a major technical challenge.
- Manufacturing process (spray drying/emulsion) significantly impacts microsphere morphology and performance.
- Regulatory success depends on comprehensive physicochemical + performance characterization, not just Q1/Q2 sameness.
- Advanced analytical method development and PLGA fingerprinting are key differentiators in ANDA approval.
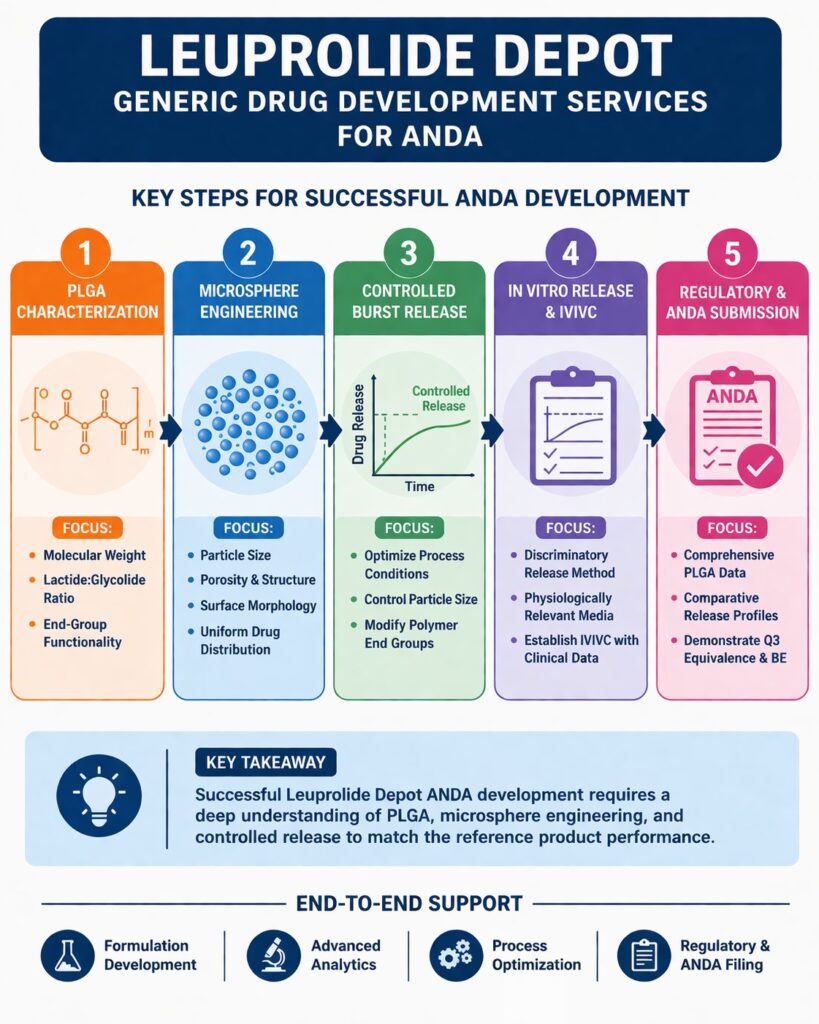
Leuprolide Depot Generic Drug Development Services – Why PLGA Characterization is Important for ANDA
PLGA characterization is one of the most important steps in Leuprolide Depot Generic Drug Development Services, as it directly affects drug release and overall product performance. The polymer controls how the drug is released over weeks or months, making it a critical component of the formulation.
In products like Lupron Depot®, leuprolide acetate is trapped inside PLGA microspheres. The properties of this polymer decide how fast or slow the drug is released. Even small changes in polymer composition can lead to big differences in clinical outcomes.
Key PLGA Attributes That Must Be Matched:
- Molecular weight (Mw, Mn)
- Lactide:glycolide ratio
- End-group functionality (acid vs ester terminated)
- Polymer crystallinity and Tg
- Residual monomers and impurities
To match these properties accurately, advanced analytical tools are required. Consistency between batches is also essential to ensure reliable performance and patient safety.
Ensure your submission meets all regulatory benchmarks: Check the Analytical Requirements for ANDA Generic Drugs
Reverse Engineering Lupron Depot® – Foundation of Leuprolide Depot Generic Drug Development Services
Reverse engineering is a key starting point in Leuprolide Depot Generic Drug Development Services. It helps identify the critical quality attributes (CQAs) needed to match the reference product. This step provides a clear direction for formulation and process development.
The process involves studying the internal structure of the microspheres and understanding how the drug is distributed. Scientists extract and analyze the polymer to recreate a similar formulation.
The process involves:
- Extracting and isolating PLGA from microspheres
- Determining polymer composition and structure
- Studying drug distribution inside microspheres
Reverse engineering also reveals hidden formulation details that are not publicly available. This reduces development risks and improves the chances of regulatory approval.
Key Reverse Engineering Outputs:
| Parameter | Analytical Technique | Importance |
|---|---|---|
| PLGA ratio | ¹H-NMR | Controls degradation rate |
| Molecular weight | GPC/SEC | Impacts release duration |
| Particle morphology | SEM | Affects burst release |
| Drug loading | HPLC | Ensures dose uniformity |
Navigate the complexities of generic drug filings: Review the Requirements for ANDA Submission in Generic Drugs
PLGA Microsphere Characterization in Leuprolide Depot Generic Drug Development Services
Microsphere characterization ensures that the developed product behaves like the reference drug. It is essential for confirming safety, quality, and effectiveness.
Critical Characterization Areas:
Particle Size Distribution
Particle size affects how the drug is injected and released. A consistent size range ensures predictable performance and better patient outcomes. Variations can lead to inconsistent dosing and release patterns.
Porosity & Internal Structure
The internal pore structure controls how the drug moves through the polymer. Properly controlled porosity helps achieve a steady and sustained release over time.
Surface Morphology
The outer surface of microspheres influences the initial drug release. Smoother surfaces usually reduce sudden release at the beginning, which is important for safety.
Drug Distribution
Uniform distribution of the drug ensures consistent release from each microsphere. Uneven loading can result in unpredictable therapeutic effects.
Additional tests like zeta potential and surface energy can provide deeper insights into stability and storage behavior.
Controlling Burst Release – A Core Challenge in Leuprolide Depot Generic Drug Development Services
Burst release is the rapid release of drug immediately after injection. In Leuprolide Depot Generic Drug Development Services, controlling this phase is very important to match the reference product.
This effect happens mainly due to drug present near the surface of microspheres. Polymer hydration and internal pores also contribute to early drug release.
Strategies to Control Burst Release:
- Optimize solvent evaporation conditions
- Control particle size distribution
- Modify polymer end groups
- Adjust drug-polymer interactions
Small changes in process parameters can significantly affect burst release. Therefore, careful optimization and testing are required.
Understand the risks associated with complex generics: Read about Nitrosamine Risk Assessment in Generic Drugs
In Vitro Release Testing & IVIVC in Leuprolide Depot Generic Drug Development Services
In vitro release testing helps predict how the drug will behave inside the body. It is a critical requirement in Leuprolide Depot Generic Drug Development Services for regulatory approval.
Key Requirements:
- Discriminatory release methods
- Physiologically relevant media
- Long-term testing (1–3 months)
Release Phases:
- Initial burst phase
- Diffusion-controlled phase
- Polymer degradation phase
To establish IVIVC (In Vitro–In Vivo Correlation), lab results must match clinical data. This requires advanced modeling and strong scientific justification.
Analytical Techniques Powering Leuprolide Depot Generic Drug Development Services
Advanced analytical tools are essential for understanding and controlling formulation properties. These techniques help ensure product quality and equivalence.
Essential Techniques:
- NMR spectroscopy → PLGA composition
- GPC/SEC → Molecular weight distribution
- DSC → Thermal properties
- SEM → Morphology
- XRPD → Crystallinity
- LC-MS/HPLC → Peptide integrity
Using multiple techniques together gives a more complete understanding of the formulation. This approach is now expected by regulatory authorities.
Accelerate your development timeline in North America: Partner with a Pharmaceutical CDMO in the US and Canada
Manufacturing Process Impact in Leuprolide Depot Generic Drug Development Services
The manufacturing process has a strong impact on the final product. Even with the same formulation, different processes can lead to different results.
Common Methods:
- Oil-in-water (O/W) emulsion solvent evaporation
- Spray drying
Critical Process Parameters:
- Solvent type
- Temperature (ताप)
- Stirring speed
- Polymer concentration
Proper process control ensures consistent quality during scale-up and commercial production.
Decide on the best operational strategy for your project: Compare CRO vs. In-House ANDA Development
Regulatory Expectations for ANDA – Beyond Q1/Q2 Sameness
Regulatory agencies require more than matching ingredients. A deep understanding of product performance is essential for approval.
Key ANDA Expectations:
- Full PLGA characterization
- Comparative in vitro release profiles
- Demonstration of Q3 equivalence
- IVIVC or clinical BE studies
Meeting these expectations helps avoid delays and improves approval success rates.
Leverage expert support for complex formulations: Find the right Generic Drug Development CRO for ANDA
Integrated Workflow for Leuprolide Depot Generic Drug Development Services
A structured workflow is essential for successful development. It ensures smooth coordination between formulation, analysis, and regulatory teams.
Typical Workflow:
- RLD characterization
- PLGA fingerprinting
- Prototype formulation development
- Analytical method development
- In vitro release & IVIVC
- Scale-up and validation
- ANDA submission
This integrated approach reduces risks and improves efficiency throughout the project lifecycle.
Conclusion
Leuprolide Depot Generic Drug Development Services require a highly specialized and scientifically rigorous approach centered on PLGA characterization, microsphere engineering, and controlled drug release mechanisms. Achieving success in ANDA submissions depends on replicating not only the formulation but also the complete performance profile of Lupron Depot®.
As regulatory expectations continue to evolve, the importance of advanced analytical techniques, mechanistic understanding, and integrated development strategies becomes even more significant. Companies that invest in these capabilities are better positioned to achieve bioequivalence and accelerate approval timelines.
📞 Get Expert Support
For specialized support in Leuprolide Depot Generic Drug Development Services, connect with experts:
❓ FAQs (Most Asked Questions)
Leuprolide depot belongs to a group of medicines called gonadotropin-releasing hormone (GnRH) agonists. These drugs act on the pituitary gland to control hormone production. They are commonly used in conditions like prostate cancer, endometriosis, and hormone-sensitive disorders. The depot form allows slow and sustained release over time.
Leuprolide does not increase natural GnRH levels but acts like it in the body. Initially, it overstimulates GnRH receptors, which causes a temporary rise in certain hormones. After continued use, it suppresses hormone production by desensitizing the receptors. This leads to a decrease in testosterone or estrogen levels.
Leuprolide should not be used in patients who are allergic to it or similar drugs. It is also contraindicated during pregnancy because it can harm the developing fetus. Patients with unexplained vaginal bleeding should avoid its use until evaluated. Medical history should always be reviewed before starting treatment.
Patients should avoid missing scheduled doses, as this can affect treatment results. It is also important to avoid medications or supplements that may interfere with hormone levels unless approved by a doctor. Alcohol and smoking should be limited, as they may worsen side effects. Regular monitoring and medical guidance are essential.
Leuprolide is neither chemotherapy nor immunotherapy. It is a hormone therapy that works by lowering certain hormone levels in the body. This helps slow or stop the growth of hormone-sensitive tumors. It is commonly used in prostate cancer and other hormone-related conditions.
The duration of action depends on the specific depot formulation used. Some injections work for one month, while others can last three, four, or even six months. The drug is designed to release slowly over time. This reduces the need for frequent dosing and improves patient convenience.
Reference:
- Chwalisz, K. (2023). Clinical development of the GnRH agonist leuprolide acetate depot. F&S Reports, 4(2 Suppl), 33–39. https://doi.org/10.1016/j.xfre.2022.11.011
- Sethi, R., & Sanfilippo, N. (2009). Six-month depot formulation of leuprorelin acetate in the treatment of prostate cancer. Clinical Interventions in Aging, 4, 259–267. https://doi.org/10.2147/CIA.S4885
- U.S. Food and Drug Administration. (2012). Lupron (leuprolide acetate) injection label (NDA 020011, S-040). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020011s040lbl.pdf
- Swayzer, D. V., & Gerriets, V. (2023). Leuprolide. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK551662/



