
Introduction:
Dexamethasone impurities analysis is one of the most critical analytical requirements in the development and quality control of dexamethasone injectable products. Because injectable formulations are directly administered into the bloodstream, even trace-level impurities can create serious safety and regulatory concerns.
Pharmaceutical companies developing generic or innovator dexamethasone injections must perform detailed impurity profiling to meet global regulatory expectations such as ICH guidelines and pharmacopeial standards. This article explains the complete approach to impurity profiling in dexamethasone injectable products, including types of impurities, analytical challenges, regulatory expectations, and best practices used by experienced analytical laboratories.
Companies working on dexamethasone injection analytical methods must also ensure that impurity profiling is integrated early in method development to avoid stability and regulatory issues later.
Share via:
Summary:
- Dexamethasone impurities analysis is essential to ensure safety, efficacy, and regulatory compliance in injectable formulations.
- Injectable products require much stricter impurity control than oral dosage forms due to direct bloodstream exposure.
- Impurity profiling involves identification, qualification, and quantification of related substances, degradation products, and process impurities.
- Advanced analytical tools such as LC-MS, HRMS, and stability-indicating methods are critical for accurate results.
- Working with a specialized analytical CRO like ResolveMass Laboratories Inc. significantly improves reliability, regulatory success, and development speed. Companies involved in dexamethasone injectable CRO services often rely on specialized impurity profiling expertise to support regulatory submissions.
1: What Is Impurity Profiling in Dexamethasone Injectable Products?
Impurity profiling in dexamethasone injectable products is the scientific process of identifying, characterizing, and quantifying all impurities present in the formulation. This includes both known impurities and unknown compounds that may form during API synthesis, formulation development, storage, or product degradation.
Because injectable products are administered directly into the bloodstream, impurity profiling is not only a regulatory requirement but also a critical step in ensuring patient safety, product stability, and long-term quality performance.
In practice, impurity profiling combines advanced analytical techniques with scientific interpretation to understand how impurities form, how they behave during stability studies, and whether they remain within acceptable regulatory limits throughout the product shelf life.
For example, formulation differences between dexamethasone phosphate vs acetate can significantly impact impurity formation and degradation behavior, which makes detailed impurity profiling even more important.
Key Objectives of Impurity Profiling in Dexamethasone Injectable Products
The primary goal of impurity profiling is to ensure that the injectable product remains safe, effective, and compliant with global pharmaceutical regulations.
The main objectives include:
- Identification of all related substances in dexamethasone injection
- Quantification of impurities at trace (very low) levels
- Understanding degradation pathways under stress and stability conditions
- Ensuring product stability throughout the intended shelf life
- Meeting global regulatory requirements such as ICH impurity guidelines
Because injectable formulations require extremely strict impurity limits, dexamethasone impurities analysis must be highly sensitive, selective, and scientifically robust to detect even the smallest impurity levels.
2: Why Dexamethasone Impurities Analysis Is More Critical for Injectables
Dexamethasone impurities analysis is more critical for injectable products because any impurity present in the formulation enters the bloodstream directly. Unlike oral dosage forms, injectable drugs do not pass through the digestive system or liver metabolism before reaching systemic circulation. Even trace-level impurities can therefore have a direct clinical impact.
Because of this higher risk, regulatory agencies require much stricter impurity control and more sensitive analytical testing for injectable formulations compared to oral products.
This is particularly important for products such as dexamethasone sodium phosphate formulation, where formulation stability and degradation behavior can directly affect impurity levels.
Key Reasons Dexamethasone Impurities Analysis Is Critical for Injectables
1. Immediate systemic exposure
Impurities present in dexamethasone injection reach the bloodstream instantly, which increases the importance of accurate impurity identification and control.
2. Much lower acceptable impurity limits
Injectable products must meet significantly stricter impurity limits than oral formulations. This makes highly sensitive dexamethasone impurities analysis essential.
3. Higher regulatory scrutiny
Regulatory agencies carefully evaluate impurity data for injectable drugs because of the higher safety risk associated with direct administration.
4. Greater stability sensitivity
Dexamethasone injectable products can be sensitive to light, temperature, pH, and oxidation. Impurity levels may increase during storage if stability-indicating methods are not properly developed.
5. Sterility and compatibility requirements
Injectable formulations must also remain sterile and compatible with containers, closures, and excipients. Any interaction can create additional impurities that must be detected through advanced analysis.
Comparison: Injectable vs Oral Dosage Forms
| Factor | Oral Products | Injectable Products |
|---|---|---|
| Impurity tolerance | Moderate | Very low |
| First-pass metabolism | Yes | No |
| Stability sensitivity | Moderate | Very high |
| Regulatory expectations | Standard | Very strict |
| Analytical sensitivity required | Medium | Extremely high |
Because of these factors, pharmaceutical companies rely heavily on advanced dexamethasone impurities analysis to ensure the safety, stability, and regulatory compliance of injectable formulations.

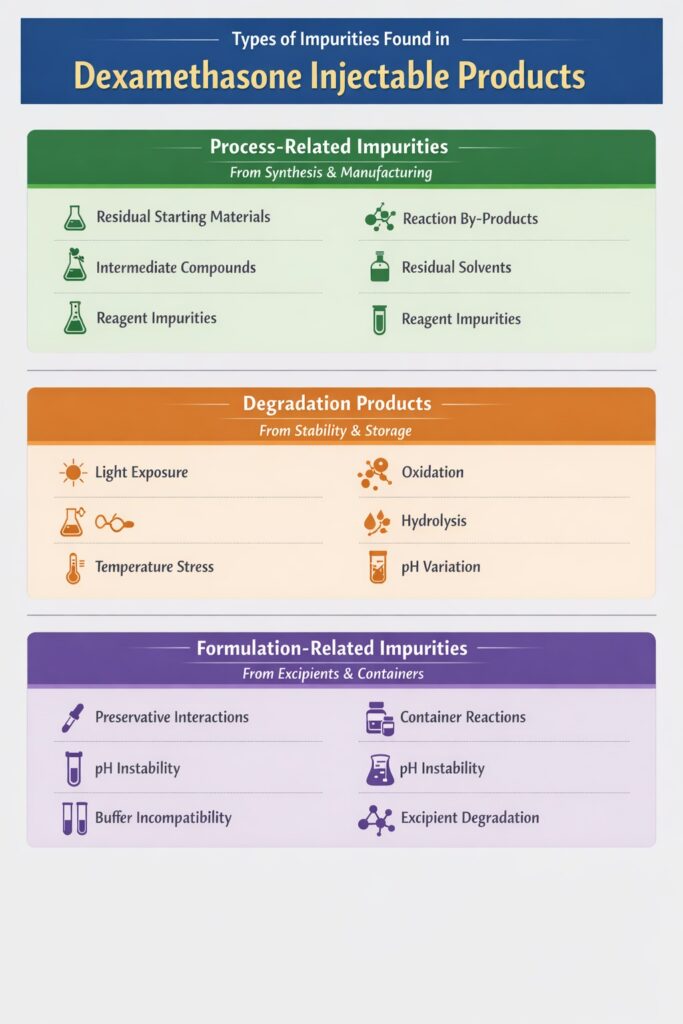
3: Types of Impurities Found in Dexamethasone Injectable Products
Several types of impurities can form in dexamethasone injectable products during manufacturing, formulation, and storage. Understanding these impurity categories is essential for accurate dexamethasone impurities analysis and for developing a reliable impurity control strategy.
In general, impurities in dexamethasone injections fall into three major categories: process-related impurities, degradation products, and formulation-related impurities.
These impurity challenges become even more important during dexamethasone injection generic development, where companies must demonstrate equivalence while meeting strict impurity limits.
1. Process-Related Impurities in Dexamethasone Injectable Products
Process-related impurities originate during the synthesis of dexamethasone API or during the manufacturing process of the injectable formulation. These impurities are typically carried forward from raw materials, intermediates, or chemical reactions used during production.
Common examples include:
- Residual starting materials
- Reaction by-products
- Intermediate compounds
- Residual solvents
- Reagent-related impurities
Because these impurities can remain in very small amounts, dexamethasone impurities analysis must use highly sensitive analytical techniques such as HPLC and LC-MS to detect them accurately.
2. Degradation Products in Dexamethasone Injectable Products
Degradation products are impurities formed when dexamethasone breaks down due to environmental or chemical stress conditions. These impurities often appear during stability studies or long-term storage.
Dexamethasone can degrade due to:
- Light exposure (photodegradation)
- Oxidation
- Hydrolysis
- Temperature stress
- pH variation
Because degradation can occur even at low levels over time, dexamethasone impurities analysis must include stability-indicating analytical methods capable of detecting even minor degradation products before they exceed acceptable limits.
3. Formulation-Related Impurities in Dexamethasone Injectable Products
Formulation-related impurities develop due to interactions between dexamethasone and excipients, containers, or formulation conditions. Injectable products are particularly sensitive to these interactions because of their complex formulation environment.
Common causes include:
- Interaction with preservatives
- Container-closure system reactions
- pH instability
- Buffer incompatibility
- Excipient-related degradation
These impurities are often difficult to predict, which is why advanced dexamethasone impurities analysis and well-designed stability studies are essential for injectable product development.
4: How Dexamethasone Impurities Analysis Is Performed
Dexamethasone impurities analysis is performed using advanced chromatographic separation combined with high-sensitivity mass spectrometry techniques. These analytical tools allow scientists to detect impurities at trace levels and accurately identify both known and unknown compounds present in dexamethasone injectable products.
In modern pharmaceutical development, impurity profiling is no longer limited to simple HPLC testing. Instead, it involves a structured analytical workflow that includes separation, detection, structural identification, and stability-indicating validation.
Common Analytical Techniques Used in Dexamethasone Impurities Analysis
The most reliable impurity profiling studies use a combination of complementary analytical techniques rather than a single method.
The most widely used techniques include:
- High-performance liquid chromatography (HPLC)
- Ultra-high-performance liquid chromatography (UHPLC)
- LC-MS (Liquid Chromatography–Mass Spectrometry)
- High-resolution mass spectrometry (HRMS)
- Stability-indicating analytical methods
- Forced degradation studies
- Photostability and oxidation testing
These techniques help scientists separate closely related steroid compounds and detect impurities at extremely low concentrations.
Typical Workflow for Dexamethasone Impurities Analysis
A structured workflow ensures that all impurities are properly identified and controlled.
Step-by-step process:
- Develop a stability-indicating chromatographic method
- Perform forced degradation studies
- Detect unknown impurities using LC-MS
- Confirm structures using high-resolution mass spectrometry
- Quantify impurities at trace levels
- Validate the analytical method according to regulatory guidelines
This approach ensures that impurity profiling is both scientifically reliable and regulatory-compliant.
Why Advanced Analytical Techniques Are Necessary
Impurities in dexamethasone injectable products often exist at extremely low concentrations, sometimes at trace-level ppm or lower. Conventional analytical methods may not provide sufficient sensitivity, selectivity, or structural confirmation.
Advanced analytical techniques are necessary because they:
- Detect impurities at very low levels
- Separate structurally similar steroid compounds
- Identify unknown degradation products
- Support regulatory submissions
- Improve long-term product stability understanding
For this reason, pharmaceutical companies rely on high-precision dexamethasone impurities analysis using LC-MS-based workflows to ensure the safety and quality of injectable formulations.
5: Regulatory Requirements for Dexamethasone Impurities Analysis
Regulatory authorities require detailed impurity profiling for dexamethasone injectable products to ensure safety, quality, and stability throughout the product lifecycle. Because injectable drugs are administered directly into the bloodstream, impurity data is one of the most closely evaluated sections during regulatory submissions.
Accurate and well-documented dexamethasone impurities analysis is therefore essential not only for compliance but also for faster approval and reduced regulatory risk.
Key Regulatory Expectations for Dexamethasone Impurities Analysis
Regulatory agencies expect pharmaceutical companies to identify, evaluate, and control every impurity that may affect product safety or quality.
The main regulatory expectations include:
- Identification of impurities above reporting thresholds
- Qualification of unknown impurities
- Quantification of related substances at trace levels
- Stability-indicating method development and validation
- Forced degradation studies to understand degradation pathways
- Complete impurity documentation in regulatory submissions
These requirements ensure that the injectable product remains safe and stable throughout its shelf life.
Important Guidelines Followed in Dexamethasone Impurities Analysis
Impurity profiling must follow internationally accepted pharmaceutical guidelines. These guidelines define impurity limits, validation requirements, and stability expectations.
The most important guidelines include:
- ICH Q3A – Impurities in new drug substances
- ICH Q3B – Impurities in new drug products
- ICH Q2 – Analytical method validation
- Stability study guidelines (long-term and accelerated studies)
- Pharmacopeial impurity limits for dexamethasone
Following these guidelines ensures that the analytical data generated during dexamethasone impurities analysis is scientifically reliable and acceptable to global regulatory agencies.
Why Regulatory Compliance Matters
Regulatory agencies place strong emphasis on impurity data because even trace-level impurities in injectable products can impact patient safety. A well-designed impurity profiling strategy helps pharmaceutical companies:
- Reduce regulatory queries
- Avoid submission delays
- Ensure consistent product quality
- Improve approval success rates
For this reason, accurate and well-documented dexamethasone impurities analysis plays a critical role in successful regulatory submissions for injectable products.

6: Challenges in Impurity Profiling of Dexamethasone Injectable Products
Impurity profiling in dexamethasone injectable products is challenging because impurities often exist at extremely low levels and many of them have very similar chemical structures. These factors make dexamethasone impurities analysis more complex compared to many other pharmaceutical products, especially when strict regulatory limits must be met.
In addition, injectable formulations are highly sensitive to stability conditions, which increases the difficulty of detecting and controlling degradation products throughout the product lifecycle.
Major Challenges in Dexamethasone Impurities Analysis
The most common technical challenges include:
- Very low impurity concentrations (trace-level detection required)
- Similar chemical structures among steroid-related compounds
- Stability-sensitive injectable formulations
- Unknown degradation pathways
- Complex chromatographic separation requirements
- Extremely strict regulatory impurity limits
Because of these challenges, impurity profiling requires advanced analytical instruments and strong scientific expertise.
Real-World Analytical Challenges and Their Impact
| Challenge | Impact on Analysis |
|---|---|
| Trace-level impurities | Requires highly sensitive analytical instruments |
| Similar steroid structures | Makes chromatographic separation difficult |
| Degradation during storage | Requires stability-indicating analytical methods |
| Unknown impurities | Requires structural elucidation using LC-MS/HRMS |
| Regulatory pressure | Demands highly accurate and well-documented data |
Why Expertise Matters
Due to the complexity of steroid molecules and the strict impurity limits for injectable products, companies increasingly rely on specialized laboratories that focus on dexamethasone impurities analysis using advanced LC-MS-based techniques.
Working with experienced analytical experts helps pharmaceutical companies:
- Ensure faster and smoother regulatory submissions
- Detect impurities earlier
- Reduce regulatory risk
- Improve product stability understanding
7: Role of Stability Studies in Dexamethasone Impurities Analysis
Stability studies play a critical role in dexamethasone impurities analysis because they help identify degradation products that form during storage. Without stability data, impurity profiling remains incomplete, and it becomes difficult to ensure the long-term safety and quality of dexamethasone injectable products.
Stability studies allow scientists to understand how the drug behaves under different environmental conditions and whether impurity levels remain within acceptable regulatory limits throughout the product shelf life.
Types of Stability Studies Used in Dexamethasone Impurities Analysis
A comprehensive impurity profiling strategy always includes multiple types of stability studies.
The most important types include:
- Accelerated stability studies
- Long-term stability studies
- Stress degradation studies
- Photostability testing
- Thermal degradation studies
- Oxidation and pH stress studies
Each of these studies helps detect different types of degradation products that may form over time.
What Stability Studies Help Scientists Understand
Stability testing provides valuable scientific data that supports both product development and regulatory submissions.
These studies help scientists understand:
- How dexamethasone degrades under stress and storage conditions
- Which impurities increase over time
- Whether the injectable formulation remains safe throughout its shelf life
- The impact of light, temperature, and pH on product stability
- Whether stability-indicating analytical methods are effective
Because impurity levels in injectable products must remain extremely low, dexamethasone impurities analysis combined with well-designed stability studies is essential to ensure product safety, regulatory compliance, and long-term stability.

8: Importance of Expert Analytical Support
Expert analytical support plays a critical role in dexamethasone impurities analysis because many impurities exist at trace levels and some remain unknown without advanced identification techniques. Working with an experienced analytical laboratory significantly improves the accuracy, reliability, and regulatory acceptance of impurity data for dexamethasone injectable products.
When impurity profiling involves complex steroid molecules and strict regulatory limits, analytical expertise becomes just as important as the instruments used.
Why Expertise Matters in Dexamethasone Impurities Analysis
Specialized analytical laboratories have the scientific experience required to handle complex impurity profiling challenges that may not be possible with routine analytical testing.
Expert support becomes especially important when:
- Unknown impurities need structural identification
- Stability-indicating methods must be developed from scratch
- Trace-level impurities must be quantified accurately
- Degradation pathways need to be scientifically understood
- Regulatory submissions require well-documented impurity data
Benefits of Specialized Analytical Expertise
Working with a laboratory that focuses specifically on impurity profiling offers significant advantages for pharmaceutical companies developing dexamethasone injectable products.
Key benefits include:
- Faster impurity identification using advanced LC-MS techniques
- Reliable stability-indicating analytical method development
- Accurate and regulatory-compliant impurity documentation
- Better understanding of degradation pathways
- Reduced analytical risks during product development
- Improved chances of regulatory approval
ResolveMass Laboratories Inc. focuses specifically on advanced dexamethasone impurities analysis using modern LC-MS and HRMS technologies. This specialized expertise helps pharmaceutical companies develop safe, stable, and fully compliant injectable formulations while reducing development risks and regulatory delays.
9: Best Practices for Dexamethasone Impurities Analysis
The best practices for dexamethasone impurities analysis focus on using a step-by-step scientific strategy that ensures accurate impurity identification, reliable quantification, and full regulatory compliance. Following proven analytical approaches helps pharmaceutical companies achieve successful impurity profiling in dexamethasone injectable products.
A structured workflow not only improves data accuracy but also reduces analytical risks during product development and regulatory submission.
Recommended Analytical Approach
The most effective strategy for dexamethasone impurities analysis follows a logical sequence of scientific steps.
Recommended workflow:
- Start with forced degradation studies to understand degradation behavior
- Develop a stability-indicating HPLC or UHPLC method
- Identify unknown impurities using LC-MS techniques
- Confirm impurity structures using high-resolution mass spectrometry (HRMS)
- Validate the analytical method according to regulatory guidelines
- Perform long-term and accelerated stability studies
- Document impurity profiles for regulatory submissions
Why This Approach Works
A structured analytical approach ensures that both known and unknown impurities are properly controlled throughout the product lifecycle.
Key advantages include:
- Detects impurities at very low concentration levels
- Ensures compliance with global regulatory guidelines
- Improves safety and stability of dexamethasone injectable products
- Reduces the risk of regulatory delays or rejections
- Supports faster product approvals
- Provides scientifically reliable impurity data
By following these best practices, pharmaceutical companies can achieve highly accurate dexamethasone impurities analysis while ensuring the safety, quality, and long-term stability of injectable formulations.
10: Future Trends in Dexamethasone Impurities Analysis
Future trends in dexamethasone impurities analysis focus on higher sensitivity, faster analytical workflows, and smarter impurity identification using advanced technologies. As regulatory expectations continue to increase, pharmaceutical companies are shifting toward more precise and automated impurity profiling approaches.
Modern analytical laboratories are no longer relying only on conventional chromatographic methods. Instead, they are adopting advanced technologies that improve both detection accuracy and analytical efficiency.
Emerging Trends in Dexamethasone Impurities Analysis
Several technological and scientific advancements are shaping the future of impurity profiling in dexamethasone injectable products.
Key emerging trends include:
- High-resolution mass spectrometry (HRMS) for trace-level impurity detection
- AI-assisted impurity identification and data interpretation
- Faster UHPLC-based chromatographic methods
- More advanced stability-indicating analytical techniques
- Improved structural elucidation of unknown impurities
- Greater regulatory focus on trace-level unknown impurities
- Automated analytical workflows to reduce human error
What These Trends Mean for Pharmaceutical Companies
Adopting modern analytical strategies early can provide a strong competitive advantage in generic and injectable drug development.
These future trends will help companies:
- Detect impurities at much lower levels than before
- Identify unknown degradation products faster
- Reduce development timelines
- Improve regulatory submission success rates
- Ensure better long-term stability of dexamethasone injectable products
As analytical technology continues to evolve, dexamethasone impurities analysis will become more precise, faster, and more reliable—helping pharmaceutical companies develop safer and more compliant injectable formulations while meeting increasingly strict global regulatory requirements.
Conclusion:
Dexamethasone impurities analysis is essential for ensuring the safety, stability, and regulatory compliance of dexamethasone injectable products. From process-related impurities to degradation products formed during storage, every impurity must be accurately identified and quantified.
Because injectable formulations demand extremely strict impurity control, working with a specialized analytical laboratory is one of the most reliable ways to ensure success in development and regulatory approval. With the right analytical expertise, pharmaceutical companies can reduce risks, improve stability performance, and accelerate time-to-market.
Frequently Asked Questions:
Dexamethasone impurities analysis is the scientific process used to identify, characterize, and quantify impurities present in dexamethasone injectable products. These impurities may form during API synthesis, formulation development, storage, or degradation over time. The goal is to ensure that the product remains safe and stable throughout its shelf life. Regulatory agencies require detailed impurity profiling before approval. Accurate dexamethasone impurities analysis also helps pharmaceutical companies avoid stability and safety issues.
Impurity analysis is more critical for injectable products because the drug is administered directly into the bloodstream. Unlike oral drugs, there is no metabolic barrier before exposure, which increases safety risks. Even very small impurity levels can affect patient safety. Regulatory agencies therefore require stricter impurity limits for injectables. This makes dexamethasone impurities analysis an essential step in development and quality control. It also helps ensure long-term product stability.
Dexamethasone injections may contain process-related impurities, degradation products, and formulation-related impurities. Process impurities originate during API synthesis or manufacturing. Degradation products form due to light, heat, oxidation, or pH changes. Formulation-related impurities may arise from interactions with excipients or containers. Identifying all these impurities is essential for regulatory approval. That is why comprehensive dexamethasone impurities analysis is required.
Several advanced analytical techniques are used in dexamethasone impurities analysis to detect impurities at very low levels. Common methods include HPLC, UHPLC, LC-MS, and high-resolution mass spectrometry (HRMS). These techniques help separate closely related steroid compounds and identify unknown impurities accurately. Stability-indicating analytical methods are also developed to detect degradation products. Using multiple techniques improves the reliability of impurity profiling. This ensures the data meets regulatory expectations.
Stability studies help scientists understand how dexamethasone degrades over time under different conditions. These studies identify degradation products that may not be visible during initial testing. They also help determine whether impurity levels remain within acceptable limits during storage. Long-term and accelerated stability studies are both required by regulatory agencies. Stability data supports the product shelf life and regulatory submissions. Therefore, stability testing is a key part of dexamethasone impurities analysis.
Regulatory authorities require pharmaceutical companies to identify and quantify impurities above specific reporting thresholds. Unknown impurities must also be identified and qualified if they exceed acceptable limits. Analytical methods used must be validated according to ICH guidelines. Stability-indicating methods and forced degradation studies are also required. Complete impurity documentation must be included in regulatory submissions. Accurate dexamethasone impurities analysis plays a major role in approval success.
LC-MS is one of the most important tools used in dexamethasone impurities analysis because it can detect impurities at extremely low concentrations. It also helps identify unknown degradation products by providing structural information. Conventional analytical techniques cannot always identify impurities accurately. LC-MS allows scientists to confirm the molecular structure of impurities. This makes impurity profiling more reliable and scientifically strong. It is especially useful for complex steroid compounds.
Reference
- KUMAR B, MUDHULKAR R, GUNDEKARI S, RAO DV, ANAMALAGUNDAM L. IDENTIFICATION AND CHARACTERIZATION OF NEW IMPURITIES IN DEXAMETHASONE OPHTHALMIC SUSPENSION. Int J App Pharm. 2025;17(5):292-301.https://www.academia.edu/download/124590177/53923.pdf
- Ummiti K, Vakkala S, Panuganti V, Annarapu MR. Isolation, identification, and characterization of 17-oxo dexamethasone, an oxidative degradation impurity of dexamethasone using flash chromatography and NMR/HRMS/IR. Journal of Liquid Chromatography & Related Technologies. 2014 Oct 21;37(17):2403-19.https://www.tandfonline.com/doi/abs/10.1080/10826076.2013.836712
- Raju PM, Shyamala P, Narayana BV, Dantuluri HS, Bhupatiraju RV. A fast, validated UPLC method coupled with PDA-QDa detectors for impurity profiling in betamethasone acetate and betamethasone phosphate injectable suspension and isolation, identification, characterization of two thermal impurities. InAnnales Pharmaceutiques Françaises 2022 Nov 1 (Vol. 80, No. 6, pp. 837-852). Elsevier Masson.https://www.sciencedirect.com/science/article/pii/S0003450922000347
- Chen Q, Zielinski D, Chen J, Koski A, Werst D, Nowak S. A validated, stability-indicating HPLC method for the determination of dexamethasone related substances on dexamethasone-coated drug-eluting stents. Journal of pharmaceutical and biomedical analysis. 2008 Nov 4;48(3):732-8.https://www.sciencedirect.com/science/article/pii/S0731708508003853
- Xu Q. Incorporating solid-phase extraction into compendial procedures for the determination of dexamethasone and impurities in low-dose drug products. Journal of Pharmaceutical and Biomedical Analysis. 2019 Oct 25;175:112773.https://www.sciencedirect.com/science/article/pii/S073170851931427X


