
Introduction:
The analytical requirements ANDA generic drugs must fulfill are the foundation of successful generic drug approval. In an ANDA submission, regulators are not asking you to prove safety and efficacy again—but they do require strong analytical data to demonstrate that the generic product is truly equivalent to the reference drug.
For pharmaceutical companies entering the generic market, understanding the analytical requirements ANDA generic drugs must meet can prevent regulatory rejection, reduce development costs, and accelerate approval timelines.
This article explains the complete analytical framework required for ANDA submissions and how companies can design a regulatory-ready analytical strategy from the beginning. Learn more about the generic drug development process for ANDA submissions.
Share via:
Summary:
If you are planning an ANDA submission, the analytical requirements are the most critical part of regulatory success. This article explains the analytical requirements ANDA generic drugs must meet and how to avoid costly delays.
Key takeaways:
- Analytical requirements ANDA generic drugs focus mainly on proving equivalence to the reference listed drug (RLD).
- Regulatory agencies expect validated methods, impurity profiling, stability data, and bioanalytical proof of equivalence.
- Poor analytical planning is one of the biggest reasons for ANDA deficiencies and delays.
- Advanced techniques like LC-MS/MS, HRMS, impurity profiling, and method validation are essential.
- A strong analytical strategy can reduce regulatory risk and speed up approval timelines.
1: What Are the Analytical Requirements ANDA Generic Drugs Must Meet?
The analytical requirements ANDA generic drugs must meet are designed to prove that the generic product is pharmaceutically equivalent and bioequivalent to the reference listed drug (RLD). Regulators do not re-evaluate safety and efficacy for generics—but they require strong analytical evidence proving the generic drug performs the same as the original product.
In simple terms, regulatory agencies expect analytical data that clearly demonstrates:
- The active pharmaceutical ingredient (API) is the same
- The impurity profile is comparable to the reference drug
- The formulation behaves the same under test conditions
- The drug performs the same in the human body
Core Analytical Requirements ANDA Generic Drugs Must Fulfill
To meet regulatory expectations, companies must generate scientifically reliable analytical data in the following areas:
- Analytical method development and validation
- Impurity identification and quantification
- Stability studies (long-term and accelerated)
- Bioanalytical method validation for bioequivalence studies
- Dissolution testing and in-vitro performance comparison
- Comparative analytical characterization with the reference drug
Why These Analytical Requirements Matter
These analytical requirements ensure that a generic drug is not just visually or chemically similar—but scientifically proven to be equivalent in quality, performance, and safety. Strong analytical data is what allows regulatory authorities to approve a generic drug without requiring full clinical trials.
2: Why Analytical Requirements ANDA Generic Drugs Depend on Strong Method Validation
The analytical requirements ANDA generic drugs must meet begin with method validation, as regulators rely on validated analytical methods to trust submitted data. Without properly validated methods, even a scientifically equivalent generic drug may face regulatory rejection.
Key Method Validation Parameters
Regulators expect validated methods to demonstrate:
- Accuracy – The method measures the true concentration of the API or impurity
- Precision – Consistent results across repeated measurements
- Specificity – Ability to detect the analyte without interference from excipients or impurities
- Linearity – Accurate response across the intended concentration range
- Robustness – Stability under small method variations
- Detection and quantitation limits – Capability to detect and quantify low-level impurities
Learn more about analytical method development for generic drugs.
Real-World Example
A generic drug formulation may be stable and pharmaceutically equivalent, but if the impurity detection method cannot reliably identify low-level degradants, the ANDA submission may fail. Regulatory agencies consider such gaps critical, often resulting in deficiency letters.
Why Method Validation Is Critical
Strong method validation ensures that all analytical data meets regulatory expectations. It forms the foundation of the analytical requirements ANDA generic drugs must satisfy, enabling regulators to approve the product confidently and preventing costly delays in generic drug development.
3: Analytical Requirements ANDA Generic Drugs: Impurity Profiling and Control
Impurity profiling is one of the most critical analytical requirements ANDA generic drugs must meet because impurities can directly impact drug safety, efficacy, and overall quality. Regulatory agencies require detailed analytical evidence to ensure that any impurities in the generic drug are identified, quantified, and controlled.
Key Impurity Types Regulators Expect
For ANDA submissions, the following impurities must be thoroughly analyzed:
- Process-related impurities – Residues from synthesis or manufacturing
- Degradation impurities – Byproducts formed over time or under stress
- Residual solvents – Traces of organic solvents used in production
- Nitrosamine impurities – Highly controlled due to toxicity concerns
- Unknown impurity identification – Any uncharacterized peaks detected during testing
Why Impurity Profiling Matters
Even when the active ingredient is identical to the reference drug, differences in manufacturing processes or formulation can introduce new impurities. Regulatory expectations require companies to demonstrate that:
- All impurities are identified
- All impurities are controlled through manufacturing and formulation
- Impurity levels remain within acceptable regulatory limits
Typical Analytical Techniques for Impurity Profiling
| Analytical Technique | Purpose in ANDA Submission |
|---|---|
| LC-MS/MS | Detect and quantify impurities accurately |
| High-resolution mass spectrometry (HRMS) | Identify unknown impurities |
| HPLC | Routine impurity testing for consistency |
| Forced degradation studies | Evaluate stability-related impurity formation |
| Stability testing | Monitor impurities over long-term storage |
Advanced analytics like mass spectrometry expertise and bioanalysis services in Canada ensure compliance.
These analytical techniques are essential to satisfy the analytical requirements ANDA generic drugs must comply with and ensure a regulatory-ready submission.
4: How Bioanalytical Testing Supports Analytical Requirements ANDA Generic Drugs
Bioanalytical testing is a crucial part of the analytical requirements ANDA generic drugs must meet, as it provides the evidence needed to demonstrate bioequivalence between the generic product and the reference listed drug (RLD). Regulators rely on robust bioanalytical data to ensure the generic drug behaves in the body just like the original product.
Key Bioanalytical Requirements for ANDA Submissions
To meet regulatory expectations, bioanalytical testing must include:
- LC-MS/MS method development – Sensitive detection of the drug in biological samples
- Bioanalytical method validation – Ensuring accuracy, precision, and reliability of measurements
- Plasma drug concentration measurement – Determining drug levels in human samples during bioequivalence studies
- Stability of the drug in biological matrices – Ensuring sample integrity over the course of testing
- Accuracy and precision in real biological samples – Demonstrating that the method produces reproducible results in clinical settings
Learn more about bioanalytical strategy for drug development.
Why Bioanalytical Testing Matters
Even when a generic formulation appears chemically identical to the reference drug, the true proof of equivalence comes from its in vivo behavior. Bioanalytical testing confirms this by providing precise measurements of drug concentrations in plasma, which regulators use to evaluate bioequivalence.
Without validated bioanalytical methods, a bioequivalence study cannot reliably support an ANDA submission, making this step one of the most critical components of the analytical requirements for generic drug development.
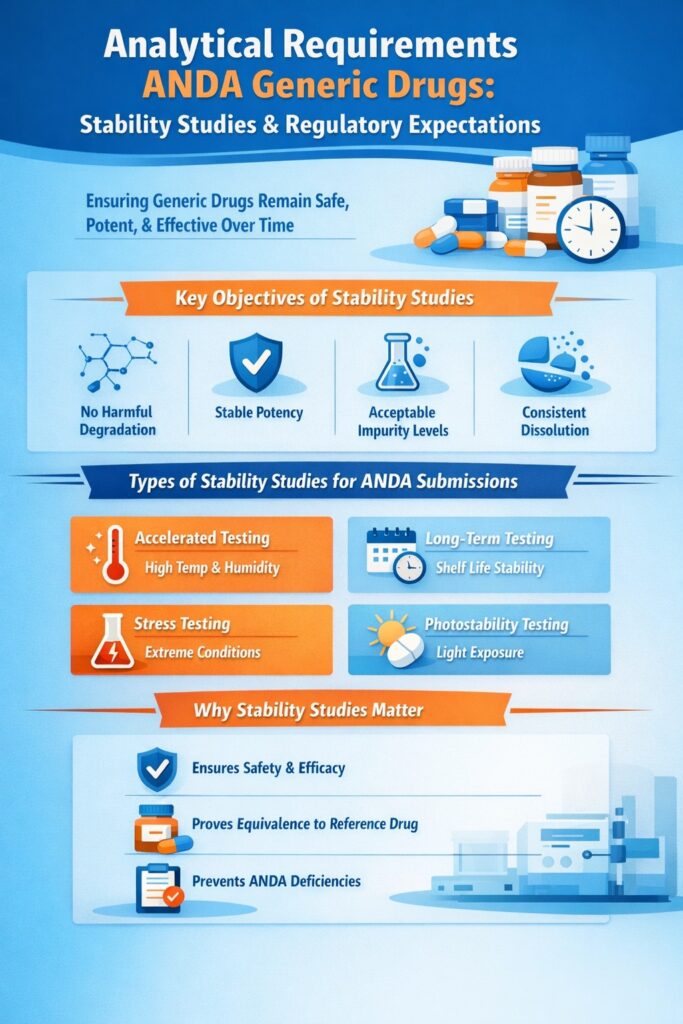
5: Analytical Requirements ANDA Generic Drugs: Stability Studies and Regulatory Expectations
Stability studies are a critical component of the analytical requirements ANDA generic drugs must meet, as regulators need assurance that the generic drug remains safe, effective, and high-quality throughout its shelf life. These studies provide evidence that the drug maintains its potency, purity, and performance under various storage conditions.
Key Objectives of Stability Studies
Regulators expect stability data to demonstrate that the generic drug:
- Does not undergo harmful degradation over time
- Maintains stable potency throughout its shelf life
- Keeps impurity levels within acceptable limits
- Shows consistent dissolution and in-vitro performance
For more details, see analytical development for generic drugs in Canada.
Types of Stability Studies Required for ANDA Submissions
To meet regulatory expectations, the following stability studies are typically conducted:
- Accelerated stability testing – Evaluates drug stability under elevated temperature and humidity to predict long-term behavior
- Long-term stability testing – Confirms stability under recommended storage conditions over the product’s shelf life
- Stress testing – Identifies potential degradation pathways under extreme conditions
- Photostability testing – Assesses the effect of light exposure on drug integrity
- Stability-indicating method validation – Ensures analytical methods can accurately detect degradation products
Why Stability Studies Are Essential
These studies directly support the analytical requirements ANDA generic drugs must satisfy for regulatory approval. By confirming that the generic product remains safe, effective, and equivalent to the reference drug over time, stability studies help prevent ANDA deficiencies and reduce the risk of post-approval quality issues.
6: Common Challenges in Meeting Analytical Requirements ANDA Generic Drugs
Pharmaceutical companies often face delays and regulatory hurdles because they underestimate the complexity of the analytical requirements ANDA generic drugs must satisfy. Meeting these requirements requires careful planning, technical expertise, and regulatory knowledge.
Most Common Challenges
Some of the key challenges in meeting analytical requirements include:
- Poor method validation – Inadequate validation can lead to unreliable data and regulatory deficiencies
- Delays in unknown impurity identification – Unknown or uncharacterized impurities can cause submission delays
- Incomplete stability data – Lack of comprehensive stability studies can trigger regulatory questions
- Weak bioanalytical method development – Ineffective methods fail to demonstrate bioequivalence accurately
- Lack of early regulatory strategy – Failing to align analytical planning with regulatory expectations can result in costly revisions
See our reverse engineering and analytical services for more on developing regulatory-ready strategies.
Real-World Impact of Analytical Challenges
Even a single deficiency in analytical data can delay an ANDA approval by several months. In competitive generic markets, these delays can translate into:
- Financial loss due to postponed market entry
- Increased development costs from additional testing
- Regulatory risk and potential resubmissions
By anticipating these challenges and implementing a strong analytical strategy, companies can mitigate risk, accelerate approvals, and ensure regulatory compliance.
7: How a Strong Analytical Strategy Helps Meet Analytical Requirements ANDA Generic Drugs
A well-designed analytical strategy is critical for meeting the analytical requirements ANDA generic drugs must satisfy. By planning analytical activities early and aligning them with regulatory expectations, companies can reduce development risk, avoid regulatory delays, and ensure submission success.
Learn more about peptide testing services and peptide characterization in drug development.
Key Components of a Strong Analytical Strategy
A robust analytical strategy typically includes:
- Early impurity risk assessment – Identifying potential impurities during early development to mitigate risks
- Forced degradation studies – Understanding degradation pathways to guide stability testing and method development
- Advanced LC-MS/MS method development – Sensitive and specific analytical methods for impurity and bioanalytical testing
- Stability-indicating method design – Ensuring methods can detect degradation products over the product’s shelf life
- Bioanalytical method development aligned with regulatory expectations – Supporting accurate bioequivalence studies
Benefits of a Strong Analytical Approach
Implementing a strong analytical strategy offers multiple advantages, including:
- Faster ANDA approval – Well-prepared submissions reduce the risk of regulatory deficiencies
- Reduced regulatory risk – Early identification of potential gaps prevents delays
- Better product quality – Comprehensive analytical data ensures equivalence and safety
- Lower development costs – Fewer repeated tests and fewer regulatory queries
- Stronger regulatory confidence – Demonstrates technical expertise and thorough preparation
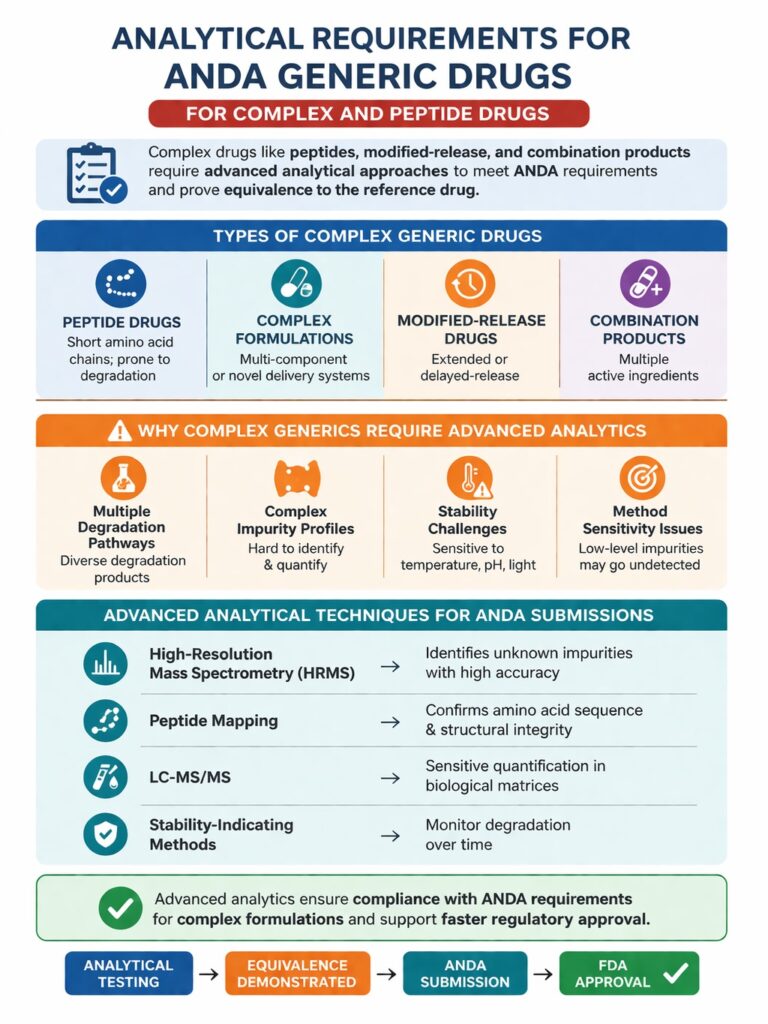
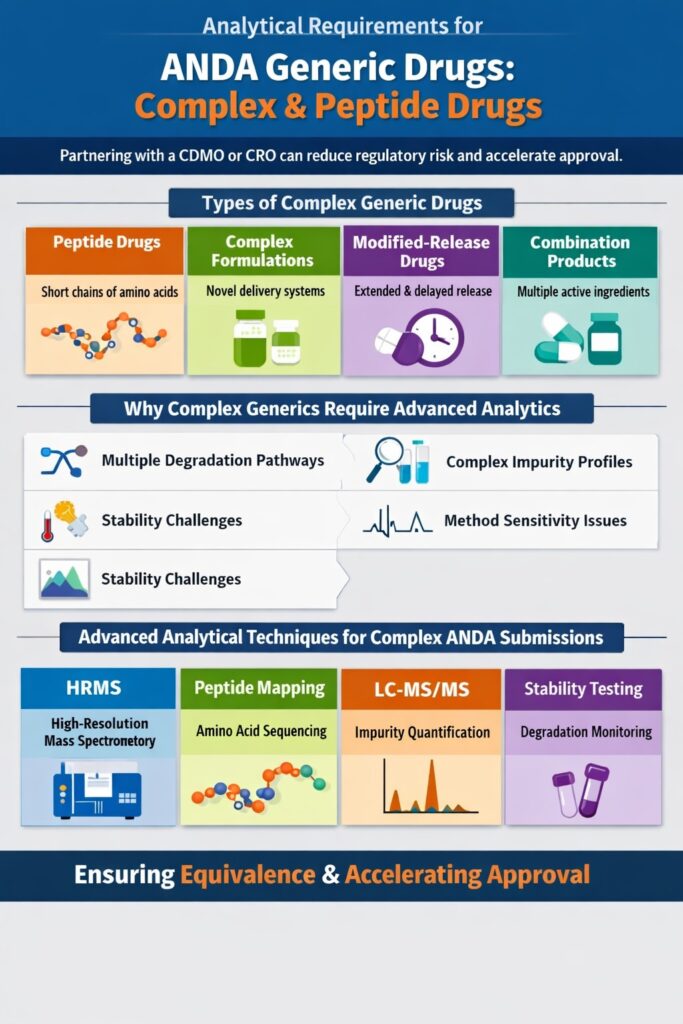
8: Analytical Requirements ANDA Generic Drugs for Complex and Peptide Drugs
The analytical requirements ANDA generic drugs must satisfy become significantly more challenging when the drug is a complex molecule, such as a peptide, modified-release formulation, or combination product. These drugs demand advanced analytical strategies to demonstrate equivalence and ensure regulatory approval.
Partnering with a CDMO for generic drug development in Canada or a generic drug development CRO for ANDA reduces regulatory risk and accelerates approval timelines.
Types of Complex Generic Drugs
Complex generics include:
- Peptide drugs – Short chains of amino acids with high sensitivity to degradation
- Complex formulations – Multi-component or novel delivery systems
- Modified-release drugs – Extended-release or delayed-release formulations
- Combination products – Drugs containing multiple active ingredients
Why Complex Generics Require Advanced Analytics
Complex molecules often present analytical challenges, including:
- Multiple degradation pathways – Leading to diverse degradation products
- Complex impurity profiles – Difficult to identify and quantify without advanced techniques
- Stability challenges – Sensitive to temperature, pH, or light exposure
- Analytical method sensitivity issues – Standard methods may not detect low-level impurities
Advanced Analytical Techniques for Complex ANDA Submissions
To address these challenges, advanced analytical methods are essential:
- High-resolution mass spectrometry (HRMS) – Identifies unknown impurities with high accuracy
- Peptide mapping – Confirms the amino acid sequence and structural integrity of peptide drugs
- LC-MS/MS – Sensitive quantification of impurities and drug levels in biological matrices
- Stability-indicating methods – Detect and monitor degradation products over time
By leveraging these techniques, companies can meet the analytical requirements ANDA generic drugs must satisfy, even for highly complex formulations, and submit regulatory-ready data that supports equivalence to the reference drug.
9: How Regulatory-Focused Laboratories Help Meet Analytical Requirements ANDA Generic Drugs
Pharmaceutical companies often partner with regulatory-focused analytical laboratories because the analytical requirements ANDA generic drugs must satisfy are highly technical, specialized, and subject to strict regulatory scrutiny. Experienced laboratories provide the expertise and infrastructure to ensure that all analytical data is regulatory-ready and submission-compliant.
Key Services Provided by Regulatory-Focused Laboratories
Expert laboratories support ANDA submissions by offering:
- Regulatory-ready method validation – Developing and validating analytical methods that meet FDA and Health Canada standards
- Advanced LC-MS/MS testing – Sensitive detection and quantification of impurities and active pharmaceutical ingredients
- Impurity identification support – Characterization and control of known and unknown impurities
- Stability-indicating method development – Designing methods that can detect degradation products and support long-term stability studies
- Bioanalytical method validation – Ensuring accurate measurement of drug concentrations in biological matrices for bioequivalence studies
Benefits of Working with Specialized Laboratories
Collaborating with a laboratory experienced in regulatory compliance helps companies:
- Reduce the risk of ANDA rejection due to analytical deficiencies
- Accelerate submission timelines by providing validated, regulatory-ready data
- Ensure high-quality data for impurities, stability, and bioanalytical testing
- Strengthen regulatory confidence in the generic product
By leveraging expert laboratories, companies can confidently meet the analytical requirements ANDA generic drugs must fulfill and improve the likelihood of a successful ANDA approval.
Conclusion:
The analytical requirements ANDA generic drugs must meet are not just a regulatory formality—they are the scientific foundation of a successful ANDA submission. From impurity profiling and method validation to stability studies and bioanalytical testing, every analytical step plays a crucial role in proving equivalence to the reference drug.
Companies that invest in a strong analytical strategy early, leveraging specialized CDMO and peptide analytical services, are more likely to achieve faster approval, regulatory confidence, and successful product launch.
If you are planning an ANDA submission and want to ensure your analytical strategy is regulatory-ready, expert guidance can make a significant difference.
Frequently Asked Questions:
ANDA generic drugs must demonstrate equivalence to the reference listed drug through validated analytical methods that ensure quality, purity, potency, and stability. This includes testing for impurities, degradation products, and bioanalytical performance.
Complex molecules, such as peptides, modified-release, or combination products, have multiple degradation pathways, sensitive stability profiles, and complex impurity patterns. These characteristics require advanced analytical techniques to ensure accurate and regulatory-compliant data.
Regulatory-focused laboratories provide expertise in method validation, LC-MS/MS testing, impurity identification, stability-indicating methods, and bioanalytical validation. Their data is submission-ready and meets FDA and Health Canada standards, reducing the risk of rejection.
Techniques include high-resolution mass spectrometry (HRMS) for unknown impurity identification, peptide mapping for sequence verification, LC-MS/MS for sensitive quantification, and stability-indicating methods to monitor degradation over time.
A stability-indicating method is an analytical test that can detect degradation products of a drug over time under various conditions. It ensures the drug maintains quality, safety, and efficacy throughout its shelf life.
Reference
- Pawar J, Hegde N, Sharma S. Regulatory frameworks and filing discrepancies in generic drug approvals: A cross-regional study with analysis of FDA ANDA deficiencies. InAnnales Pharmaceutiques Françaises 2025 Mar 10. Elsevier Masson.https://www.sciencedirect.com/science/article/pii/S0003450925000434
- Hasan M, Shimu S, Akther A, Jahan I, Hamiduzzaman M, Hasan AH. Development of generic drug products by pharmaceutical industries considering regulatory aspects: a review. Journal of Biosciences and Medicines. 2021;9(10):23-39.https://www.researchgate.net/profile/Shahnaz-Akter-4/publication/363844194_Development_of_Generic_Drug_Products_by_Pharmaceutical_Industries_Considering_Regulatory_Aspects_A/links/63313d1686b22d3db4e34f85/Development-of-Generic-Drug-Products-by-Pharmaceutical-Industries-Considering-Regulatory-Aspects-A.pdf
- Pawar J, Hegde N, Sharma S. Focusing on First Cycle Approval in ANDA Submission: Understanding Common Deficiencies & Case Study Insights. Therapeutic Innovation & Regulatory Science. 2025 May;59(3):426-37.https://link.springer.com/article/10.1007/s43441-025-00755-5
- PARASRAMPURIA S, SERTKAYA A, LORD A, BERGER C. Cost of generic drug development and approval final.https://aspe.hhs.gov/sites/default/files/documents/66e13df52a6d314ba4e10c8e55b68b4b/cost-of-generic-drugs-erg.pdf
- Wittayanukorn S, Rosenberg M, Schick A, Hu M, Wang Z, Babiskin A, Lionberger R, Zhao L. Factors that have an impact on abbreviated new drug application (ANDA) submissions. Therapeutic Innovation & Regulatory Science. 2020 Nov;54(6):1372-81.https://link.springer.com/article/10.1007/s43441-020-00163-x


