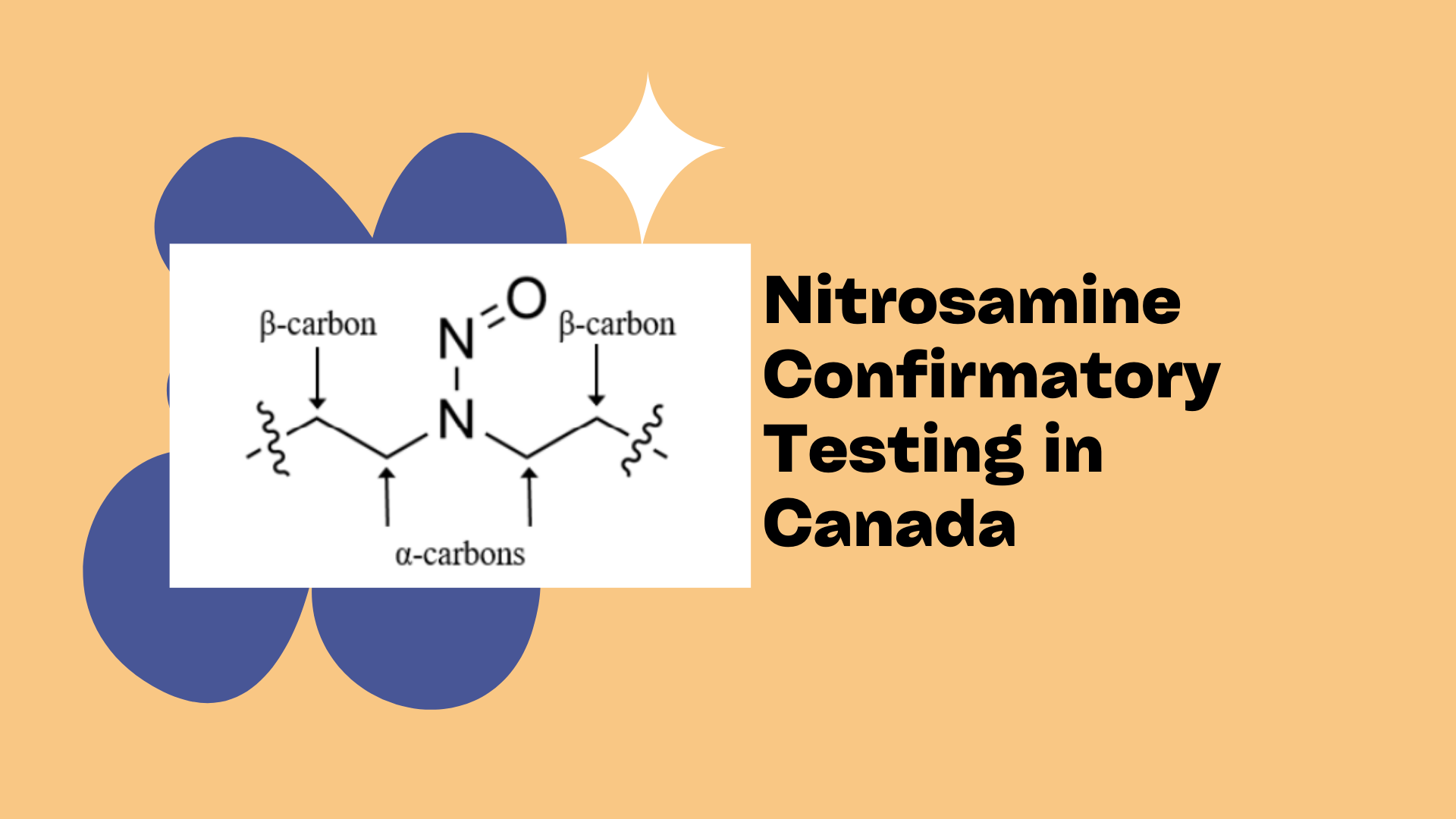
Why You Need a Specialized Nitrosamine Impurity Analysis Lab for Your Pharma Project
In today’s highly regulated pharmaceutical environment, manufacturers are under pressure to ensure that every product batch is free of harmful […]
Why You Need a Specialized Nitrosamine Impurity Analysis Lab for Your Pharma Project Read More »