
Introduction:
The generic pharmaceutical market in the United States and Canada is highly competitive and strictly regulated, making speed and compliance critical for successful market entry. One of the most effective strategies companies use today is partnering with a Contract Development and Manufacturing Organization (CDMO).
A specialized CDMO accelerate generic drug development US and Canada by providing expertise in analytical testing, bioanalysis, regulatory strategy, and method development. Instead of building expensive in-house infrastructure, pharmaceutical companies can leverage the experience and technology of CDMOs to streamline development and achieve faster regulatory approvals.
This article explains how CDMOs accelerate generic drug market entry in United States and Canada, the services they provide, and why outsourcing has become essential in modern generic drug development.
Share via:
Summary:
- CDMOs accelerate generic drug development in the US and Canada by providing integrated analytical, bioanalytical, and regulatory support.
- Specialized laboratories help reduce development timelines and regulatory risk.
- CDMOs offer advanced analytical technologies such as LC-MS/MS, impurity profiling, and method validation required for regulatory approvals.
- Working with a CDMO ensures compliance with FDA and Health Canada requirements.
- Strategic outsourcing allows pharmaceutical companies to focus on commercialization while experts handle complex analytical work.
- Partnering with experienced laboratories significantly improves the success rate of generic drug approvals.
1: Why the Generic Drug Market Requires Speed and Precision
Speed and precision are critical in the generic drug market because the first approved generic product often captures a major share of the market after patent expiration. Companies that launch earlier can benefit from temporary market exclusivity and stronger commercial positioning.
However, achieving rapid market entry requires overcoming several technical and regulatory challenges. Generic drug developers must produce high-quality analytical and bioequivalence data while meeting strict regulatory expectations in both the United States and Canada.
Key Challenges in Generic Drug Development
- Strict regulatory requirements from the U.S. Food and Drug Administration and Health Canada
- Complex analytical characterization requirements for APIs and finished formulations
- Demonstrating bioequivalence with the reference listed drug
- Detecting impurities and degradation products using advanced analytical techniques
- Developing validated analytical methods that meet regulatory guidelines
- Managing tight development timelines to reach the market quickly
Because of these challenges, many pharmaceutical companies rely on specialized partners that CDMO accelerate generic drug development US and Canada by providing advanced analytical capabilities, regulatory expertise, and integrated development support. These services are often delivered by organizations offering generic pharmaceutical CDMO services in Canada.
2: How CDMOs Accelerate Generic Drug Development US and Canada
A CDMO accelerate generic drug development US and Canada by providing integrated analytical, bioanalytical, and regulatory services required for successful regulatory submissions. By consolidating these capabilities within one organization, CDMOs help pharmaceutical companies reduce development timelines and improve regulatory readiness.
Generic drug development requires multiple complex steps, including method development, bioanalysis, impurity testing, and regulatory documentation. Experienced CDMOs streamline these processes by using specialized expertise and advanced technologies to generate accurate, compliant data efficiently.
In many cases, companies rely on analytical method development for generic drugs to ensure analytical workflows meet regulatory expectations from the beginning.
Key Ways CDMOs Speed Up Development
| Development Area | How CDMOs Help |
|---|---|
| Analytical Method Development | Develop and validate analytical methods for API and drug product analysis |
| Bioanalytical Testing | Demonstrate bioequivalence with the reference listed drug using validated assays |
| Impurity Profiling | Identify, quantify, and characterize impurities and degradation products |
| Regulatory Documentation | Prepare analytical data packages required for submissions to the U.S. Food and Drug Administration and Health Canada |
| Advanced Instrumentation | Provide access to technologies such as LC-MS/MS and high-resolution mass spectrometry (HRMS) |
By integrating these capabilities within a single workflow, a CDMO accelerate generic drug development US and Canada while ensuring the analytical data generated meets strict regulatory expectations. This integrated approach significantly reduces the time required to produce regulatory-ready data packages, helping pharmaceutical companies move closer to market approval.
3: CDMO Accelerate Generic Drug Development US and Canada Through Advanced Analytical Capabilities
One of the most important ways a CDMO accelerate generic drug development US and Canada is through advanced analytical laboratories that generate high-quality, regulatory-ready data. These laboratories use sophisticated technologies and validated workflows to characterize drug substances and drug products in detail, which is essential for regulatory approval.
These laboratories often rely on mass spectrometry experts in drug development to perform complex molecular characterization and impurity identification required during regulatory submissions.
For a generic drug to be approved, developers must demonstrate that their product is equivalent to the reference listed drug in quality, safety, and performance. This requires extensive analytical characterization across multiple parameters.
Critical Areas of Analytical Characterization
Generic drug development typically requires detailed evaluation of:
- Active Pharmaceutical Ingredients (APIs) to confirm identity, purity, and composition
- Impurity profiles to detect and quantify trace contaminants
- Degradation products formed during manufacturing or storage
- Stability data to demonstrate product shelf life under defined conditions
- Bioequivalence results confirming similarity to the reference drug
Some generic programs also require reverse engineering for development of generic drugs to understand the composition and performance characteristics of the reference product.
These analytical studies form a major part of regulatory submissions to agencies such as the U.S. Food and Drug Administration and Health Canada.
Essential Analytical Technologies Used by CDMOs
To support these requirements, CDMOs rely on advanced instrumentation and specialized analytical expertise.
Key technologies include:
- LC-MS/MS (Liquid Chromatography–Mass Spectrometry) for highly sensitive quantification of drug compounds
- High-Resolution Mass Spectrometry (HRMS) for structural identification of impurities and metabolites
- Peptide Mapping for characterization of peptide-based therapeutics
- Nitrosamine Impurity Testing to detect trace carcinogenic impurities
- Stability Studies to evaluate product behavior under various environmental conditions
For peptide-based therapeutics, specialized services such as peptide characterization in drug development and peptide testing services are often required to confirm structural integrity and purity.
These techniques allow scientists to generate highly precise analytical data that regulatory agencies require before approving a generic drug.
By using validated analytical methods, advanced instrumentation, and experienced scientific teams, CDMOs ensure accuracy, reproducibility, and regulatory compliance throughout the development process. This capability is a major reason why pharmaceutical companies rely on partners that CDMO accelerate generic drug development US and Canada.
4: CDMO Accelerate Generic Drug Development US and Canada by Reducing Regulatory Risk
A CDMO accelerate generic drug development US and Canada by ensuring regulatory compliance from the earliest stages of development. Early alignment with regulatory expectations helps pharmaceutical companies avoid costly delays, data deficiencies, and repeated testing.
Regulatory compliance is one of the most complex aspects of generic drug development. Both the U.S. Food and Drug Administration and Health Canada require extensive scientific data demonstrating that a generic drug is equivalent to the reference product in terms of safety, quality, and performance.
Experienced organizations that provide regulatory support for generic drugs in the US and Canada understand how to align analytical strategies with FDA and Health Canada expectations.
Regulatory Requirements Include
Generic drug submissions typically require:
- Bioequivalence studies demonstrating comparable pharmacokinetic performance
- Analytical method validation according to regulatory guidelines
- Impurity and degradation analysis to ensure product safety and quality
- Stability testing to confirm product shelf life and storage conditions
- Quality control documentation supporting manufacturing and analytical processes
By implementing robust analytical methods and validated workflows, a CDMO accelerate generic drug development US and Canada while ensuring that generated data meets regulatory expectations.
How CDMOs Reduce Regulatory Risk
A proactive regulatory strategy helps pharmaceutical companies minimize potential issues during regulatory review.
Key advantages include:
- Reduced regulatory delays during submission review
- Lower risk of data rejection or regulatory queries
- Decreased need for additional confirmatory testing
By aligning development strategies with regulatory requirements from the beginning, CDMOs help companies streamline the approval process and move generic products to market more efficiently.

5: CDMO Accelerate Generic Drug Development US and Canada Through Integrated Services
A CDMO accelerate generic drug development US and Canada by offering integrated development services that streamline project management and reduce coordination delays. Instead of managing multiple vendors for analytical testing, bioanalysis, and regulatory support, pharmaceutical companies can rely on a single experienced partner.
Many pharmaceutical companies now rely on outsourcing generic drug development in Canada to simplify development workflows and access specialized expertise.
Integrated programs often include analytical development for generic drugs in Canada alongside regulatory and bioanalytical support.
This integrated approach simplifies development workflows and ensures that all scientific and regulatory activities are aligned with requirements from agencies such as the U.S. Food and Drug Administration and Health Canada.
When services are managed within one organization, communication between scientific teams becomes more efficient, and development timelines can be significantly reduced.
Benefits of Integrated CDMO Services
- Faster communication between scientific and regulatory teams
- Reduced operational complexity by eliminating the need to manage multiple vendors
- Streamlined workflows across analytical testing, bioanalysis, and documentation
- Faster decision-making during development and troubleshooting
- Consistent regulatory documentation aligned with submission requirements
This integrated approach is a key advantage offered by organizations providing CDMO services for generic drug development in Canada.
By consolidating multiple development functions under one roof, a CDMO accelerate generic drug development US and Canada while improving efficiency, data consistency, and regulatory readiness. Integrated services ultimately help pharmaceutical companies shorten development timelines and reach the market sooner.
6: Role of Bioanalytical Testing in Accelerating Generic Drug Approvals
Bioanalytical testing is critical because it proves that a generic drug performs the same way in the body as the original reference product. These studies generate the pharmacokinetic and bioequivalence data required for regulatory approval.
Specialized laboratories performing bioanalysis in Canada generate pharmacokinetic and bioequivalence data required for regulatory approval.
A CDMO accelerate generic drug development US and Canada by conducting highly sensitive bioanalytical studies that measure drug concentrations in biological samples and confirm therapeutic equivalence. These studies are essential components of regulatory submissions to agencies such as the U.S. Food and Drug Administration and Health Canada.
Key Bioanalytical Activities
Bioanalytical laboratories perform several critical studies during generic drug development:
- Pharmacokinetic studies to understand how the drug is absorbed, distributed, metabolized, and eliminated in the body
- Bioequivalence testing to confirm that the generic drug performs similarly to the reference listed drug
- Drug concentration measurement in biological samples such as plasma, serum, or urine
- LC-MS/MS quantitative analysis for highly sensitive and accurate drug quantification
These studies generate the data required to demonstrate therapeutic equivalence, which is a fundamental requirement for generic drug approval.
Importance of Validated Bioanalytical Workflows
Because bioanalytical data directly supports regulatory submissions, these studies must follow strict regulatory validation guidelines.
Experienced CDMOs maintain:
- GLP-compliant laboratories
- Validated analytical methods
- Standardized sample handling protocols
- Quality-controlled data reporting systems
By ensuring accuracy, reproducibility, and regulatory compliance, CDMOs help pharmaceutical companies generate reliable bioanalytical data efficiently, which is another way a CDMO accelerate generic drug development US and Canada.
7: How Early Analytical Strategy Helps CDMOs Accelerate Generic Drug Development US and Canada
An early analytical strategy allows developers to anticipate technical and regulatory challenges before they cause delays. By planning analytical workflows at the beginning of development, companies can avoid repeated testing, regulatory queries, and method failures.
A CDMO accelerate generic drug development US and Canada by performing a detailed scientific evaluation before analytical testing begins. This early assessment helps ensure that all analytical methods and studies are aligned with regulatory expectations from agencies such as the U.S. Food and Drug Administration and Health Canada.
A structured bioanalytical strategy for drug development helps ensure that all testing workflows are aligned with regulatory expectations.
Key Factors Evaluated During Early Analytical Planning
Before designing an analytical workflow, experienced CDMO scientists typically assess:
- Molecular complexity of the active pharmaceutical ingredient
- Impurity risks that may affect drug safety and regulatory approval
- Analytical challenges related to detection, quantification, or stability
- Regulatory expectations for data required in generic drug submissions
Based on this scientific evaluation, the CDMO develops a custom analytical roadmap that guides testing, method development, and validation throughout the development lifecycle.
Benefits of Early Strategy Planning
Implementing an analytical strategy early in development offers several advantages:
- Fewer analytical failures during testing
- Faster method development and validation
- Reduced need for re-testing or method adjustments
- Improved regulatory acceptance of analytical data
This proactive planning approach ensures that analytical studies are designed efficiently and aligned with regulatory requirements from the beginning. As a result, strategic planning is one of the key reasons a CDMO accelerate generic drug development US and Canada so effectively.
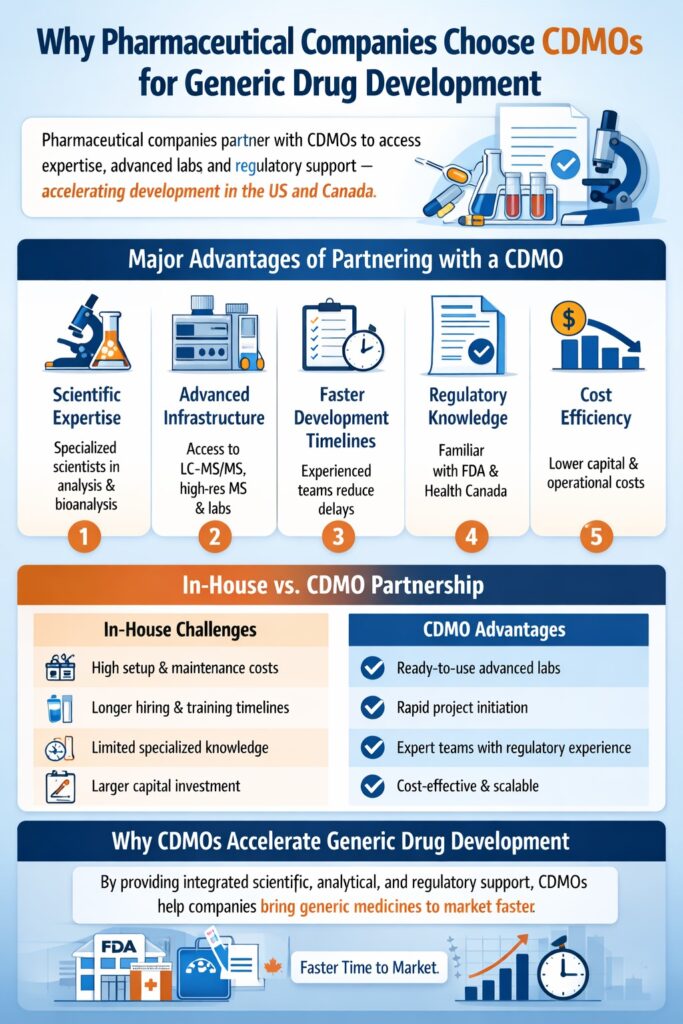
8: Why Pharmaceutical Companies Choose CDMOs for Generic Drug Development
Pharmaceutical companies increasingly partner with CDMOs because outsourcing provides specialized expertise, advanced infrastructure, and faster development timelines. Instead of building expensive in-house laboratories, companies can leverage the experience and resources of dedicated development partners.
Organizations that provide outsourced chemistry in drug discovery and development support help pharmaceutical companies reduce internal resource requirements.
Companies also evaluate CDMO vs CRO for generic drug development when selecting the most suitable development partner.
A CDMO accelerate generic drug development US and Canada by providing integrated scientific, analytical, and regulatory capabilities that support efficient product development and regulatory submissions.
Major Advantages of Partnering with a CDMO
1. Scientific Expertise
CDMOs employ highly trained scientists who specialize in pharmaceutical analysis, bioanalysis, and method development. Their experience helps ensure that analytical strategies are designed correctly from the start.
2. Advanced Infrastructure
State-of-the-art analytical laboratories require significant investment. CDMOs already operate advanced facilities equipped with technologies such as LC-MS/MS and high-resolution mass spectrometry, eliminating the need for pharmaceutical companies to build their own infrastructure.
3. Faster Development Timelines
Experienced CDMO teams understand the technical and regulatory requirements of generic drug development. This expertise helps accelerate project timelines and reduces delays.
4. Regulatory Knowledge
CDMOs regularly support regulatory submissions to agencies such as the U.S. Food and Drug Administration and Health Canada. Their familiarity with these regulatory frameworks helps ensure that analytical data and documentation meet submission standards.
5. Cost Efficiency
Outsourcing analytical and development work reduces capital investment in equipment, laboratories, and specialized personnel. Companies can allocate resources more efficiently while maintaining high scientific standards.
Many organizations also provide CDMO services for generic projects in Canada to support complex development programs.
Because of these advantages, many pharmaceutical developers rely on partners that CDMO accelerate generic drug development US and Canada, enabling them to remain competitive and bring generic medicines to market more efficiently.
9: Why ResolveMass Laboratories Supports Generic Drug Development
ResolveMass Laboratories supports pharmaceutical companies developing generic drugs by providing specialized analytical and bioanalytical services required for regulatory approval. With expertise in complex molecular analysis and advanced mass spectrometry technologies, the laboratory helps generate high-quality data that meets regulatory expectations.
The laboratory has demonstrated expertise through projects such as this generic peptide drug analytical characterization case study, which highlights advanced analytical workflows used during complex drug development programs.
A key way ResolveMass contributes to projects where a CDMO accelerate generic drug development US and Canada is by delivering reliable analytical solutions that support regulatory submissions to agencies such as the U.S. Food and Drug Administration and Health Canada.
Core Analytical Capabilities
ResolveMass Laboratories specializes in several critical analytical services used in generic drug development:
- LC-MS/MS bioanalysis for accurate quantification of drugs and metabolites in biological samples
- Peptide and complex molecule analysis for advanced molecular characterization
- Analytical method development and validation to support regulatory submissions
- Impurity identification and characterization using advanced mass spectrometry techniques
- Regulatory-ready analytical documentation aligned with global regulatory standards
Supporting Regulatory-Ready Drug Development
By combining scientific expertise with state-of-the-art analytical instrumentation, ResolveMass Laboratories helps pharmaceutical companies generate reliable and reproducible analytical data required during drug development.
This approach ensures that drug developers receive:
- Accurate analytical results
- Validated and regulatory-compliant methods
- High-quality data packages suitable for regulatory review
Through this scientific and regulatory support, ResolveMass helps organizations navigate complex development requirements and contribute to projects where a CDMO accelerate generic drug development US and Canada throughout the entire development lifecycle.
Conclusion:
Entering the generic pharmaceutical market in the United States and Canada requires extensive analytical testing, regulatory compliance, and scientific expertise. Without the right support, development timelines can become long and costly.
A specialized CDMO accelerate generic drug development US and Canada by providing integrated services such as analytical testing, bioanalysis, regulatory support, and advanced instrumentation. These capabilities help pharmaceutical companies generate regulatory-ready data faster while reducing development risk.
By partnering with an experienced laboratory such as ResolveMass Laboratories, drug developers can streamline the development process, improve regulatory success rates, and bring generic medicines to market more efficiently.
Frequently Asked Questions:
Generic drug development timelines can vary depending on the complexity of the drug and regulatory requirements. On average, the process may take 3 to 5 years, including analytical testing, bioequivalence studies, stability testing, and regulatory submissions. Partnering with experienced development organizations can significantly reduce delays and streamline the approval process.
Generic drugs are reviewed and approved by:
-U.S. Food and Drug Administration (FDA) in the United States through the Abbreviated New Drug Application (ANDA) process.
-Health Canada in Canada through the Abbreviated New Drug Submission (ANDS) pathway.
Both agencies require comprehensive analytical and bioequivalence data to ensure that generic drugs match the reference product in quality, safety, and efficacy.
Many types of pharmaceuticals can be developed as generic medicines, including:
-Small molecule drugs
-Oral solid dosage forms such as tablets and capsules
-Injectable drugs
-Peptide-based therapeutics
-Complex formulations requiring specialized analytical characterization
The complexity of the drug often determines the level of analytical testing and regulatory scrutiny required.
Generic drug developers may face several technical and regulatory challenges, such as:
-Identifying and characterizing impurities in drug formulations
-Reproducing the performance of the reference listed drug
-Developing validated analytical methods
-Meeting strict regulatory documentation requirements
-Managing tight timelines before market competition increases
Addressing these challenges early helps improve development efficiency.
Regulatory agencies require detailed analytical data to verify that a generic drug matches the reference product in composition, purity, and stability. Analytical characterization helps confirm the identity of active ingredients, detect impurities, and ensure consistent product quality throughout the drug’s shelf life.
Reference
- Kishore Kumar Hotha. Fast-to-Clinic, Fast-to-Market in Biotech Innovation: Optimizing Chemistry Manufacturing & Controls (CMC) Excellence for Clinical and Commercial Success.https://drhothas.com/wp-content/uploads/2024/09/Fast-to-Clinic-Fast-to-Market-in-Biotech-Innovation-Optimizing-Chemistry-Manufacturing-Controls-CMC-Excellence-for-Clinical-and-Commercial-Success.pdf
- CDMO Industry Thrives Amid Consolidation.https://www.pharmamanufacturing.com/sector/contract-manufacturing/article/11309693/cdmo-industry-thrives-amid-consolidation
- CMO Market Report.https://www.aiswariyachidambaram.com/articles/Contract%20Pharma%20Article.pdf
- Jim Huang, PhD and Shaukat Ali, PhD. A CDMO Perspective on Developing Biological Modalities from Discovery to Commercialization.https://pure.rug.nl/ws/portalfiles/portal/495105256/AmericanPharmaceuticalRevew_apr_nov_dec_2022.pdf#page=31

