
Introduction:
When evaluating CRO vs In-house ANDA Development, it’s essential to understand that both approaches aim to successfully develop generic drug products that satisfy FDA requirements for an Abbreviated New Drug Application (ANDA). The choice between an internal team and outsourcing to a Contract Research Organization (CRO) or Contract Development and Manufacturing Organization (CDMO) impacts time to market, budget, scientific expertise, regulatory risk, and product quality.
At ResolveMass Laboratories Inc., our expertise in bioanalytical strategy for drug development and mass spectrometry helps sponsors make informed decisions that improve efficiency, reduce risk, and accelerate approvals.
Share via:
Summary:
- CRO vs In‑house ANDA Development: Choosing between in‑house ANDA teams and external partners (CRO/CDMOs) hinges on cost, experience, speed, risk, and regulatory expertise.
- In‑House ANDA Development offers complete control but demands heavy infrastructure, resources, and regulatory expertise.
- CRO/CDMO partners provide specialized capabilities, scalable resources, and faster timelines with reduced operational burden.
- Best Fit Decision: For most generic drug sponsors, a hybrid model maximizes efficiency, cost‑effectiveness, and compliance.
- Critical Success Factors: Regulatory oversight, data quality, scientific experience, project accountability, timelines, and cost transparency.
- ResolveMass Advantage: Proven experience in ANDA lifecycle support, equipped to guide sponsors through both CRO collaborations and in‑house strategies.
1: What is ANDA Development?
ANDA development is the process of preparing and submitting a generic drug application that proves the product is bioequivalent to the reference listed drug (RLD), meets all required quality standards, and complies with FDA regulatory requirements. This process is a critical step in generic drug commercialization, whether handled in-house or via a CRO/CDMO.
Core ANDA Development Components
- Bioequivalence (BE) Studies: Shows that the generic performs similarly to the RLD (bioanalysis in Canada).
- CMC (Chemistry, Manufacturing & Controls) Documentation: Ensures manufacturing process, composition, and quality control meet regulatory standards (analytical method development for generic drugs).
- Stability Studies: Confirms the product maintains quality, safety, and efficacy over its shelf life.
- Regulatory Strategy and Submission: Includes preparing and filing the ANDA dossier with the FDA.
- Post‑Approval Support: Handles inspections and updates to the product dossier (regulatory support for generic drugs US and Canada CDMO).
Tip: Companies deciding between CRO vs In-house ANDA Development often weigh their internal capabilities in managing these components against the specialized expertise offered by external partners.
2: What Does “CRO vs In‑house ANDA Development” Really Mean?
CRO vs In-house ANDA Development refers to the choice between:
- In‑house ANDA Development – managing all regulatory, scientific, and technical tasks internally.
- CRO/CDMO Support – outsourcing specialized development, regulatory, and analytical services to experienced external partners.
This decision impacts costs, timelines, control, and regulatory compliance, making it a strategic consideration for any generic drug sponsor.
Key Comparison Table
| Category | In‑House ANDA Development | CRO/CDMO Support |
|---|---|---|
| Control | High — full ownership of processes and data | Moderate — depends on vendor agreements |
| Cost | High fixed costs: infrastructure, staff, and regulatory overhead | Variable & scalable: pay-for-service, flexible project budgets |
| Time to Completion | Longer — limited internal resources can slow progress | Often faster — experienced teams and dedicated resources |
| Expertise | Dependent on internal team capabilities | Specialized & focused expertise for ANDA projects |
| Regulatory Knowledge | Requires continuous internal investment and training | Deep regulatory know-how and FDA submission experience |
Tip: Sponsors evaluate this early to balance cost, speed, and regulatory compliance. Learn more about CRO vs CDMO for generic drug development.
3: Why Compare In‑House ANDA Development vs CRO/CDMO
Choosing between in‑house ANDA development and outsourcing to a CRO/CDMO is a critical strategic decision. It directly impacts your operational strategy, project costs, product quality, and time to market for generic drugs.
Key Differences Explained
1. Cost Structure
- In‑house ANDA Development: Requires significant investment in laboratory facilities, equipment, staff salaries, and regulatory affairs expertise.
- CRO/CDMO Support: Offers predictable, project-based budgets with no long-term infrastructure commitments, making costs more scalable and manageable. Predictable budgets, scalable, pay-for-service (outsourcing generic drug development Canada).
2. Regulatory Expertise
- In‑house: Fully dependent on internal team knowledge and experience with FDA ANDA requirements.
- CRO/CDMO: Provides access to seasoned regulatory professionals who have hands-on experience preparing and submitting ANDAs, reducing compliance risk.
3. Scalability
- In‑house: Limited by internal resources, staffing, and operational capacity.
- CRO/CDMO: Easily scalable for multiple ANDA projects (CDMO for generic drug development in Canada).
Tip: Evaluating these differences early helps sponsors determine whether internal investment or external partnership best aligns with project goals, timelines, and regulatory risk management.

4: Benefits of In‑House ANDA Development
In‑house ANDA development offers companies full ownership and direct oversight of all scientific, regulatory, and quality-related activities. This approach provides maximum control over your generic drug development process, though it comes with certain challenges.
Advantages
- Direct Control: Maintain complete authority over intellectual property (IP), data, and quality systems.
- Faster Internal Approvals: Streamlined decision-making allows quicker alignment on project milestones.
- Integrated Communication: Teams work closely together, reducing miscommunication across departments.
- Custom Internal Processes: Tailor workflows and processes to meet specific company standards and priorities.
Challenges
- High Capital and Operational Costs: Significant investment in facilities, equipment, and skilled personnel is required.
- Regulatory Knowledge Maintenance: Continuous training and expertise development are necessary to keep up with FDA regulations.
- Recruitment and Retention: Finding and retaining experienced scientists and regulatory specialists can be difficult.
- Risk of Delays: Limited internal resources may slow down projects, especially complex ANDA submissions.
Tip: Companies weighing CRO vs In‑house ANDA Development often consider internal expertise, available infrastructure, and cost tolerance before choosing this path.
5: Benefits of Choosing a CRO or CDMO for ANDA Development
Partnering with a CRO or CDMO allows companies to access expert scientific and regulatory support while significantly reducing operational burden. For many sponsors, this approach accelerates timelines, ensures compliance, and leverages specialized capabilities that may not exist in-house.
Advantages
- Regulatory Expertise: In the CRO vs In‑house ANDA Development comparison, CROs bring deep knowledge of FDA requirements and ANDA submission strategies (generic drug development CRO for ANDA).
- Specialized Facilities: Access to advanced laboratories, analytical services, and bioequivalence (BE) study capabilities (analytical development for generic drugs Canada).
- Scalable Resources: Teams and resources can scale according to project complexity and timelines.
- Streamlined Processes: Dedicated workflows are optimized for rapid ANDA preparation and submission, reducing delays.
Challenges
- Vendor Oversight: Requires strong project management to monitor vendor performance and deliverables.
- Data Confidentiality: Clear agreements are essential to protect intellectual property and sensitive data.
- Control Limitations: Sponsors may have less direct control compared to fully in-house operations.
Tip: Choosing the right CRO or CDMO can balance speed, expertise, and regulatory compliance, making it a compelling alternative to full in-house ANDA development.
6: How to Choose Between In‑House and CRO/CDMO
Choosing between in‑house ANDA development and a CRO/CDMO partner requires careful evaluation of your strategic goals, internal capabilities, budget, and regulatory risk. The right decision ensures timely ANDA submissions, cost efficiency, and regulatory compliance.
Decision Checklist
When deciding between CRO vs In‑house ANDA Development, consider:
- Project Complexity and Timeline: How complicated is the product and how quickly must it reach the market?
- Internal Scientific Expertise: Does your team have the necessary analytical, regulatory, and technical skills?
- Regulatory Familiarity and FDA Submission Experience: Can your team manage FDA ANDA requirements independently? (reverse engineering for development of generic drugs)
- Budget Forecast and Cost Constraints: Are you prepared for the capital and operational costs of in-house development?
- Scale and Volume of Products Planned: Will your internal resources support multiple simultaneous ANDA projects?
Decision Framework Table
| Factor | In‑House Development | CRO/CDMO |
|---|---|---|
| Early Stage Projects | Sometimes cumbersome due to resource limitations | Recommended due to expertise and speed |
| Limited Regulatory Expertise | High risk of delays or FDA queries | Expert support reduces risk |
| Tight Budget | Not ideal — high fixed costs | Cost-effective and scalable |
| Need for Speed | Limited by internal bandwidth | Accelerated execution with specialized teams |
| High Volume | May justify investment if sustained | Flexible and easily scalable |
Tip: Hybrid models are common, combining internal oversight with CRO/CDMO technical support (CDMO accelerate generic drug development US and Canada).
7: Best Practices for Working With a CRO or CDMO
To maximize the benefits of a CRO or CDMO partnership, sponsors must focus on clear expectations, data quality, and proactive communication. Applying best practices ensures successful ANDA development outcomes while mitigating risks associated with outsourcing.
Effective Collaboration Strategies
When navigating CRO vs In‑house ANDA Development, consider these approaches:
- Define Service Level Agreements (SLAs): Clearly outline deliverables, responsibilities, timelines, and quality metrics.
- Clarify Regulatory Responsibilities: Specify which tasks are managed by the CRO/CDMO and which remain in-house.
- Agree on Timelines and Deliverables: Establish realistic schedules for analytical testing, bioequivalence studies, and regulatory submissions.
- Implement Transparent Data Sharing: Use secure platforms for sharing study results, reports, and regulatory documentation.
- Schedule Regular Progress Reviews: Conduct weekly or biweekly check-ins to ensure alignment and address any issues promptly.
Tip: These practices ensure efficiency and regulatory compliance (CDMO vs CRO for peptide drug development).
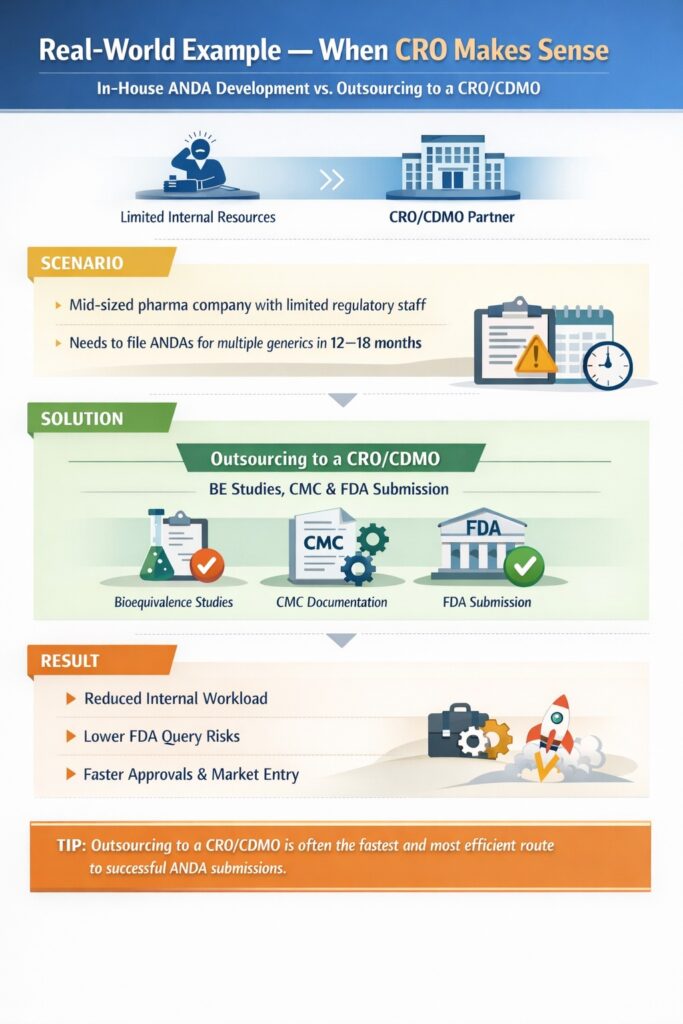
8: Real‑World Example — When CRO Makes Sense
Understanding CRO vs In-house ANDA Development is easier with a practical example. Many mid-sized companies face resource constraints that make outsourcing a strategic choice.
Scenario
- A mid-sized pharmaceutical company has limited regulatory staff.
- Needs to file ANDAs for multiple generic products within a tight timeline of 12–18 months.
Solution
- Partnering with a CRO/CDMO (generic pharmaceutical CDMO Canada) for BE studies, CMC, and FDA submission.
- CRO handles bioequivalence studies, CMC documentation, and FDA submission preparation, while the internal team focuses on strategic oversight.
Result
- Reduced internal workload and operational burden.
- Lower risk of FDA queries due to expert guidance.
- Faster approvals and market entry, leveraging the CRO’s experience and resources.
Tip: Companies with limited internal capabilities often find that outsourcing to a CRO/CDMO is the fastest and most efficient route to successful ANDA submissions.
9: Integrating BOTH — Hybrid Models
Many pharmaceutical sponsors choose a hybrid model, combining the strengths of in-house ANDA development with the specialized expertise of a CRO/CDMO. This approach allows companies to maintain strategic oversight internally while outsourcing complex technical tasks such as bioequivalence (BE) studies, CMC documentation, and analytical testing.
Benefits of a Hybrid Strategy
- Optimized Cost Distribution: Balance internal resource investment with outsourced project costs for greater efficiency (outsourced chemistry in drug discovery)
- Enhanced Internal Knowledge Gain: Internal teams learn from CRO/CDMO experts, building long-term capabilities (peptide characterization in drug development)
- Continuous Regulatory Compliance Support: External partners provide ongoing regulatory guidance while internal oversight ensures alignment with company standards (peptide testing service)
Tip: For companies evaluating CRO vs In-house ANDA Development, hybrid models often offer the best of both worlds—control, cost-efficiency, and access to specialized expertise.
10: Why ResolveMass Laboratories Excels in Both Models
At ResolveMass Laboratories Inc., we leverage decades of hands-on experience in ANDA regulatory submissions, deep knowledge of FDA quality expectations, and a strong commitment to strategic partnerships. Whether your company opts for in-house ANDA development, a CRO/CDMO partnership, or a hybrid approach, ResolveMass ensures your projects are executed efficiently and compliantly.
ResolveMass Laboratories’ Strengths
- Proven ANDA Project Leadership: Successfully guided multiple generic drug submissions from development to approval (generic drug development process ANDA).
- Integrated Scientific and Regulatory Teams: Seamless collaboration across analytical, regulatory, and bioequivalence experts.
- Customized Project Execution Plans: Tailored strategies to match your company’s internal capabilities, timelines, and goals (analytical requirements ANDA generic drugs)
- Focus on Quality, Compliance, and Timely Delivery: Ensures all regulatory and scientific standards are met without delays.
Tip: When comparing CRO vs In‑house ANDA Development, partnering with ResolveMass Laboratories provides a trusted, expert-driven solution that optimizes resources, mitigates regulatory risk, and accelerates ANDA approvals.
Conclusion:
CRO vs In-house ANDA Development is a strategic decision that affects costs, regulatory outcomes, timelines, and overall product success. While in‑house development gives control, it often falls short without extensive expertise and infrastructure. Conversely, experienced CRO/CDMOs like the experts at ResolveMass Laboratories Inc. offer a balanced, scalable, and high‑compliance approach to generic drug development for ANDA filings.
To succeed in today’s competitive generic landscape, understanding the strengths and limitations of each model is crucial — and choosing the right partner makes all the difference.
Frequently Asked Questions:
Not necessarily. Choosing between CRO vs In‑house ANDA Development depends on your company’s internal expertise, resources, project complexity, and timelines. CROs offer specialized knowledge, scalability, and faster execution, while in-house development provides full control over data, processes, and IP. Many sponsors adopt a hybrid approach to balance both advantages.
In-house ANDA development provides:
-Full control over IP, data, and quality systems
-Faster internal approvals and decision-making
-Tailored internal processes
-Close integration of scientific and regulatory teams
Challenges include high capital costs, resource limitations, and the need to maintain updated regulatory expertise.
CRO/CDMO partnerships offer:
-Specialized regulatory and scientific expertise (generic drug development CRO for ANDA)
-Access to advanced labs and bioequivalence study facilities
-Scalable resources based on project complexity
-Streamlined processes for faster ANDA submissions
Challenges include managing vendor performance, protecting confidential data, and having slightly less direct control than in-house teams.
Reference
- Nichol FR. Contract clinical research: value to in-house drug development. InClinical Drug Trials and Tribulations, Revised and Expanded 2002 Mar 26 (pp. 370-380). CRC Press.https://api.taylorfrancis.com/content/chapters/edit/download?identifierName=doi&identifierValue=10.1201/9780203909560-26&type=chapterpdf
- Crouch TJ. The changing role of CROs: The evolution of strategic out-sourcing. Clinical Research and Regulatory Affairs. 1997 Jan 1;14(3-4):205-20.https://www.tandfonline.com/doi/abs/10.3109/10601339709080079
- Calaprice-Whitty D, Hall J. Managing Clinical Development Risk: Pharma and CRO Report. Pharmaceutical Technology. 2014 Feb 1;20(10):s26-34.https://www.pharmtech.com/view/managing-clinical-development-risk-pharma-and-cro-report
- Gad, S.C., Spainhour, C.B. and Serota, D.G., 2020. Functions and types of contract support organizations (including CROs, CMDOs, packagers, and contract formulators). In Contract Research and Development Organizations-Their History, Selection, and Utilization (pp. 109-119). Cham: Springer International Publishing.https://link.springer.com/chapter/10.1007/978-3-030-43073-3_5


