Introduction:
The shift from conventional injections to long-acting drug delivery systems has transformed modern ophthalmic treatment. A well-designed Dexamethasone Sodium Phosphate Formulation plays a key role in managing inflammation, but developing it for advanced delivery systems requires more than basic formulation knowledge. It involves a clear understanding of sterility, stability, and compatibility with delivery technologies.
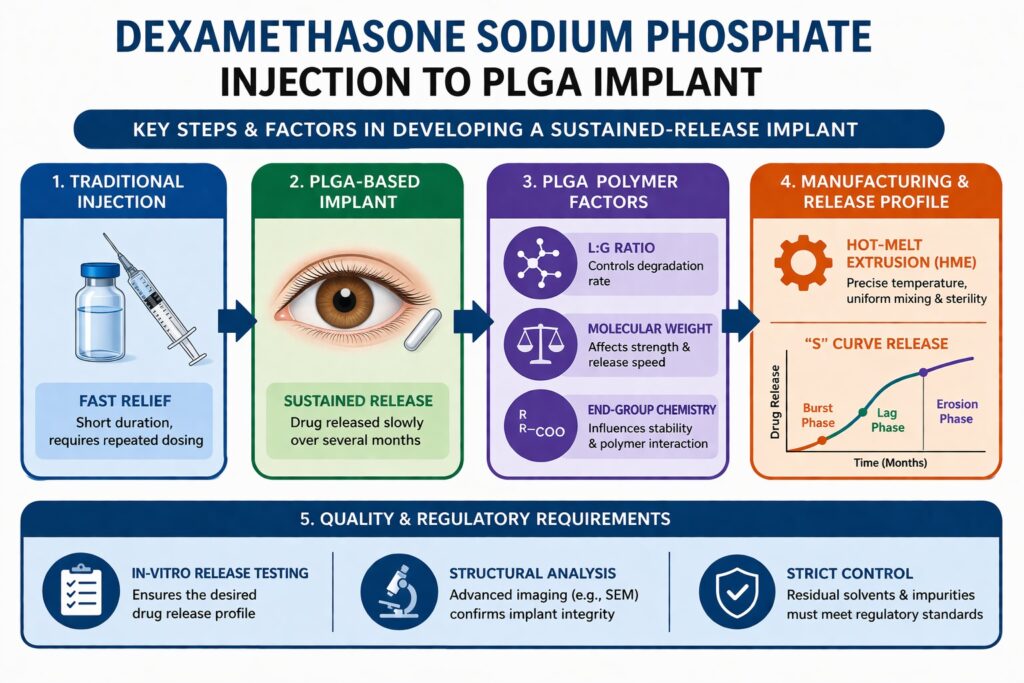
When moving from a simple injectable solution to a sustained-release implant, the level of complexity increases significantly. Developers must not only ensure drug effectiveness but also control how the drug is released over time. This requires careful planning, testing, and optimization at every stage of development. As a result, formulation scientists must combine pharmaceutical science with material science to achieve the desired outcomes.
Share via:
Summary:
- The transition from conventional dexamethasone injections to PLGA-based sustained-release implants represents a major advancement in ophthalmic drug delivery, improving treatment duration and patient compliance.
- Developing a Dexamethasone Sodium Phosphate formulation for implants is significantly more complex, requiring careful control of sterility, stability, drug release, and material compatibility.
- PLGA polymer plays a critical role in drug release behavior, with factors like L:G ratio, molecular weight, and end-group chemistry directly influencing degradation and therapeutic performance.
- Preformulation for implants focuses on solid-state properties and drug–polymer interactions, unlike traditional liquid formulations that emphasize solubility and pH.
- Manufacturing via hot-melt extrusion demands precise temperature control, uniform drug distribution, and strict sterility to ensure consistent and safe implants.
- Achieving the desired “S-curve” release profile and meeting stringent regulatory requirements are essential for developing effective and equivalent long-acting generic implants.
The Evolution from Injection to PLGA-Based Implant
Traditional dexamethasone injections are widely used because they provide fast relief from inflammation. However, their effect is short-lived, which means patients often need repeated dosing. This can be inconvenient and may increase the risk of complications such as infection or eye damage over time.
To overcome these limitations, sustained-release implants were developed. These implants are designed to release the drug slowly inside the eye over several months. When working with a Dexamethasone Sodium Phosphate Formulation in this format, the drug is embedded within a biodegradable polymer matrix. This allows for controlled and predictable drug delivery, improving patient outcomes.
The transition from injection to implant is not just a change in dosage form. It represents a complete shift in formulation strategy. Instead of focusing only on solubility and sterility, scientists must also consider polymer behavior, degradation rate, and long-term stability.
Understanding the Core Complexity: The PLGA Polymer
PLGA is the backbone of most sustained-release implants. It is biodegradable, biocompatible, and widely accepted in pharmaceutical applications. However, it is also highly sensitive to small changes in composition and processing conditions.
Even minor variations in polymer properties can lead to major differences in drug release behavior. Therefore, selecting and controlling PLGA is one of the most critical steps in developing a successful implant formulation. A stable Dexamethasone Sodium Phosphate Formulation depends heavily on how well the polymer is designed and handled.
1. The L:G Ratio (Lactide to Glycolide)
The ratio of lactic acid to glycolic acid in PLGA determines how quickly the polymer degrades. Lactic acid is more hydrophobic, while glycolic acid is more hydrophilic. This means that higher glycolide content usually leads to faster water absorption and quicker degradation.
For developers, matching this ratio to the reference product is essential. Even a small deviation can change the release profile and affect the duration of treatment. Careful sourcing and characterization of PLGA are necessary to maintain consistency.
2. Molecular Weight and Polydispersity
The molecular weight of PLGA affects both the mechanical strength of the implant and the rate of drug release. A lower molecular weight may lead to faster degradation and an initial burst release of the drug. On the other hand, a higher molecular weight can slow down the release process.
For a Dexamethasone Sodium Phosphate Formulation, achieving the right balance is important. The goal is to provide a steady and controlled release without causing sudden spikes in drug concentration. This helps in maintaining safety and effectiveness throughout the treatment period.
3. End-Group Chemistry
PLGA polymers can have different end-group chemistries, such as acid-terminated or ester-terminated forms. These differences may seem small, but they have a significant impact on how the polymer interacts with water and degrades over time.
As the polymer breaks down, it can create an acidic environment inside the implant. This can affect the stability of dexamethasone and potentially reduce its effectiveness. Therefore, understanding and controlling end-group chemistry is essential for long-term formulation stability.
Critical Preformulation for the Generic Ozurdex Model
In traditional formulations, preformulation studies focus on solubility, pH, and compatibility with excipients. However, in implant systems, the focus shifts to solid-state properties and drug-polymer interactions.
Below is a detailed overview of key parameters:
| Parameter | Importance in PLGA Implants | Target/Action |
|---|---|---|
| Glass Transition Temp (Tg) | Determines processing temperature | Must be high enough for stability but low enough to avoid drug degradation |
| Drug Loading | Percentage of API vs. Polymer | Typically ~60-70% drug loading |
| Inherent Viscosity | Relates to polymer chain length | Ensures implant strength and durability |
| Moisture Content | PLGA is sensitive to hydrolysis | Must be kept near 0% during manufacturing |
Strategic Insight: A Dexamethasone Sodium Phosphate Formulation behaves very differently in a solid implant compared to a liquid solution. Inside the implant, the microenvironment can become acidic as the polymer degrades. This can influence drug stability and must be carefully evaluated during development.
Manufacturing Challenges: Hot-Melt Extrusion (HME)
Hot-Melt Extrusion is a key technique used in the production of PLGA-based implants. In this process, the drug and polymer are heated, mixed, and shaped into a small implant. This method ensures uniform distribution of the drug within the polymer matrix.
Temperature control is one of the biggest challenges in this process. If the temperature is too high, the drug may degrade and lose its effectiveness. If it is too low, the polymer may not melt properly, leading to poor mixing and inconsistent results.
Another challenge is maintaining content uniformity. Since the implant is very small, even slight variations in drug distribution can affect performance. Additionally, the process must be carried out under strict sterile conditions to ensure product safety.
Mastering the Release Profile (The “S” Curve)
The release profile of a PLGA-based implant typically follows three phases. Each phase must be carefully controlled to achieve the desired therapeutic effect.
- Burst Phase: Initial release of drug from the surface of the implant.
- Lag Phase: A slower phase where water penetrates the polymer and prepares it for degradation.
- Erosion Phase: The polymer breaks down, releasing the remaining drug.
For a Dexamethasone Sodium Phosphate Formulation, the timing and duration of each phase are critical. A well-designed formulation ensures that the drug is released steadily over the intended treatment period without sudden changes in concentration.
Quality Control and Regulatory Requirements
Developing PLGA-based implants requires strict adherence to regulatory guidelines. These products are classified as complex generics, which means they require detailed evaluation beyond standard bioequivalence studies.
In-vitro release testing is used to simulate how the drug will be released in the eye over time. Advanced imaging techniques, such as scanning electron microscopy, are used to study the internal structure of the implant. These tests help ensure that the generic product matches the reference product in both structure and performance.
Additionally, all residual solvents and impurities must be carefully controlled. Since the implant is used in the eye, even small amounts of contaminants can cause serious issues.
Overcoming the “Generic” Hurdles
One of the biggest challenges in developing a generic implant is understanding that PLGA is not just an inactive ingredient. It plays an active role in controlling drug release and overall product performance.
To overcome this, developers must focus on controlling every aspect of the formulation process. This includes polymer selection, drug loading, and manufacturing conditions. A well-optimized Dexamethasone Sodium Phosphate Formulation can achieve the same safety and effectiveness as the original product.
Common Pitfalls in PLGA Development
- Variability in polymer batches can affect consistency.
- Exposure to moisture can start premature degradation.
- Differences in implant size and shape can alter drug release.
Understanding and avoiding these issues is essential for successful product development.
Conclusion
The development of a PLGA-based implant using a Dexamethasone Sodium Phosphate Formulation is a complex but rewarding process. It requires a deep understanding of both pharmaceutical and material science.
By focusing on polymer behavior, manufacturing precision, and controlled drug release, developers can create effective long-acting treatments. These innovations help improve patient compliance and provide better therapeutic outcomes. As the demand for advanced drug delivery systems continues to grow, expertise in this area will become increasingly important.
For expert guidance and formulation development support, connect with specialists here:
Frequently Asked Questions:
Dexamethasone sodium phosphate injection is used to reduce inflammation and suppress the immune system in serious medical conditions. It is commonly given in cases like severe allergies, asthma attacks, autoimmune diseases, and certain infections. It is also used in hospital settings for conditions such as brain swelling and some cancers. The injection works quickly, making it useful in emergency situations.
Dexamethasone may cause side effects such as increased appetite, weight gain, mood changes, and difficulty sleeping. Some people may also experience high blood sugar, fluid retention, or increased risk of infections. Long-term use can lead to bone weakness or hormonal imbalance. The severity of side effects depends on the dose and duration of use.
Yes, dexamethasone is considered a strong corticosteroid with powerful anti-inflammatory effects. It is more potent than many other steroids, which means smaller doses can produce significant results. Because of its strength, it must be used carefully under medical supervision. Incorrect use can increase the risk of side effects.
Dexamethasone injections are generally safe when administered by a healthcare professional and used for the right condition. They are often used in controlled medical settings where patients are monitored. However, like all medications, they carry some risks, especially with repeated or high-dose use. Safety depends on proper dosing and patient health conditions.
People with untreated infections, certain fungal diseases, or known allergies to corticosteroids should avoid dexamethasone. It should also be used with caution in patients with diabetes, high blood pressure, or stomach ulcers. Pregnant or breastfeeding women should consult a doctor before use. Medical guidance is important to prevent complications.
Dexamethasone helps reduce inflammation, relieve pain, and control immune system overactivity. It can quickly improve symptoms in conditions like severe allergies or respiratory distress. In critical care, it may help reduce complications in certain illnesses. Its fast and strong action makes it highly effective when used correctly.
The safe duration of dexamethasone use depends on the condition being treated and the prescribed dose. Short-term use is generally safe and commonly recommended for acute conditions. Long-term use requires close medical supervision due to potential side effects. Doctors usually aim to use the lowest effective dose for the shortest time possible.
Reference:
- Toledo, A., Amato, C. S., Clarke, N., Reitz, R. E., & Salo, D. (2015). Injectable dexamethasone sodium phosphate administered orally? A pharmacokinetic analysis of a common emergency department practice. Journal of Pediatric Pharmacology and Therapeutics, 20(2), 105–111. https://doi.org/10.5863/1551-6776-20.2.105
- Al-Owaidi, M. F., Alkhafaji, S. L., & Mahood, A. M. (2021). Quantitative determination of dexamethasone sodium phosphate in bulk and pharmaceuticals at suitable pH values using the spectrophotometric method. Journal of Advanced Pharmaceutical Technology & Research, 12(4), 378–383. https://doi.org/10.4103/japtr.japtr_6_21
- U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2009). Pharmacology review(s): Ozurdex (dexamethasone intravitreal implant), NDA 022315. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2009/022315s000_PharmR.pdf



