
INTRODUCTION
Selecting a partner for Extractables and Leachables (E&L) Testing in the United States is one of the most important decisions for any pharmaceutical, biotech, or medical device company preparing for U.S. FDA submissions. The quality of E&L data directly influences approval timelines, risk assessments, and patient safety decisions.
For deeper insights into U.S.-specific E&L requirements, refer to:
- https://resolvemass.ca/extractables-and-leachables-el-requirements-for-u-s-market-authorization/
- https://resolvemass.ca/extractables-and-leachables-el-testing-in-the-united-states/
Summary
- Choosing a laboratory for Extractables and Leachables (E&L) Testing in the United States requires evaluating scientific expertise, FDA experience, instrumentation, and documentation quality.
- The lab you choose should provide full extractables profiling, leachables monitoring, toxicology, and FDA-ready reports.
- U.S.-based labs offer an advantage because they understand evolving FDA expectations and help avoid regulatory deficiencies.
- Key factors include experience with your product type, comprehensive analytical platforms, data integrity systems, and strong scientific communication.
Get in Touch with Us
Why Choosing the Right E&L Lab Matters in 2026
The success of your regulatory submission depends heavily on the reliability, completeness, and scientific credibility of the data generated by your E&L laboratory. Incorrect or incomplete studies are among the leading causes of FDA information requests and approval delays.
Understanding the U.S. Regulatory Framework for E&L Testing
To choose the right service provider, you must first understand what U.S. FDA expects from Extractables and Leachables (E&L) testing in the United States.
Core U.S. Requirements
- Consistent with the global ICH Q3E guideline
https://resolvemass.ca/ich-q3e-guideline-for-extractables-and-leachables/ - Requires systematic identification and qualification of chemical migrants
- Mandatory for:
• Parenterals • Biologics • ATMPs • Ophthalmics • Inhalation products • Single-use systems • Devices
To compare U.S. vs. EU expectations, see:
https://resolvemass.ca/el-testing-usa-vs-europe/
Key reasons lab selection matters:
- The new ICH Q3E guideline increased the technical depth expected in E&L submissions.Compliance with ICH Q3E E&L study requirements.
- FDA now expects more detailed compound identification, exposure risk justification, and structural elucidation.
- Container–closure systems are more complex today, including multilayer polymers, adhesives, coatings, and drug–device interfaces.
- Only experienced U.S.-aligned E&L laboratories understand the interpretation differences between global regulators and the FDA.
- For undersanding more ICH Q3 Guidelines:https://resolvemass.ca/ich-q3e-extractables-and-leachables-el-study-requirements/
- https://resolvemass.ca/ich-q3e-extractables-and-leachables-el-testing-services/
- https://resolvemass.ca/ich-q3e-guideline-for-extractables-and-leachables/
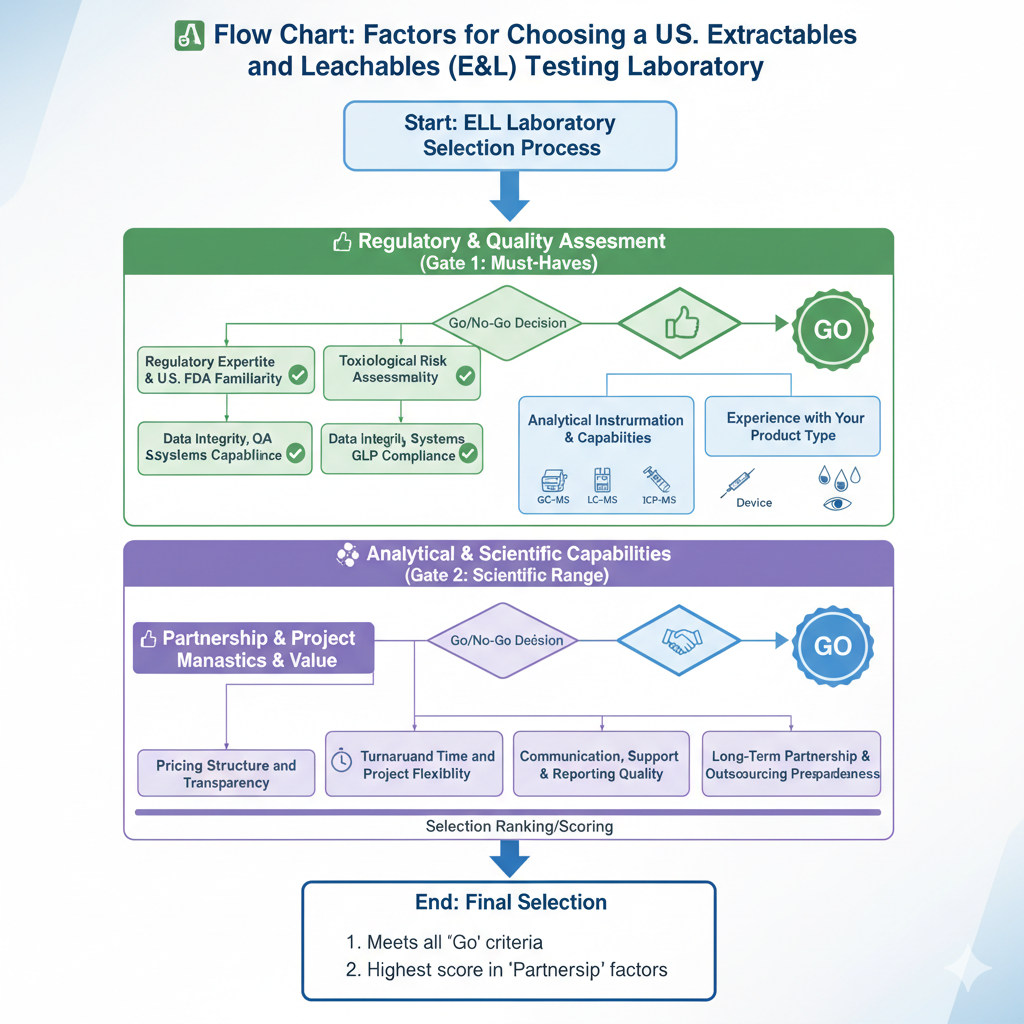
Factors to Evaluate When Choosing an Extractables and Leachables (E&L) Testing Laboratory in the United States
1. Regulatory Expertise and U.S. FDA Familiarity
A strong E&L lab must demonstrate deep understanding of U.S. FDA expectations. The first 1–2 sentences should clearly reveal how capable the laboratory is at handling regulatory-facing work.
A laboratory with strong FDA experience ensures your Extractables and Leachables (E&L) Testing in the United States meets evolving regulatory expectations without gaps.
What to evaluate:
- Experience with FDA submissions across injectables, inhalation products, ophthalmics, biologics, and medical devices.
- Knowledge of ICH Q3E, USP <1663>, USP <1664>, ISO 10993-18, and ISO 10993-17.
- Experience supporting DMFs, ANDAs, NDAs, and combination product approvals.
- Demonstrated ability to interpret FDA comments and justify scientific decisions.
2. Analytical Instrumentation & Capabilities
Your lab must have a full complement of analytical instruments to detect volatile, semi-volatile, non-volatile, and elemental extractables — and monitor leachables over time.
A comprehensive instrumentation suite ensures the laboratory can detect the full chemical profile required for Extractables and Leachables (E&L) Testing in the United States.
Required capability list:
- GC-MS / GC-MS-MS – Volatile & semi-volatile compounds
- LC-MS/MS – Non-volatiles & higher molecular weight extractables
- ICP-MS – Metals & elemental impurities
- HS-GC – Residual solvents
- FTIR, TGA, DSC – Polymer characterization
- NMR (if available) – Confident structural elucidation
Comparison Table
| Capability | Full-Service U.S. E&L Lab | Limited Capability Lab |
|---|---|---|
| GC-MS, LC-MS/MS, ICP-MS | ✔ | ❌ / Partial |
| Extractables & Leachables | ✔ | ❌ |
| Toxicological Support | ✔ | ❌ |
| FDA reporting alignment | ✔ | ❌ |
| Method Validation | ✔ | ❌ |
3. Experience With Your Product Type
Your laboratory must have direct experience with the specific container–closure system (CCS) and dosage form you are developing, because every system behaves differently.
Look for experience with:
- Parenterals – Require high sensitivity and low thresholds.
- Biologics – Risk of leachables interacting with proteins.
- Ophthalmics – Extremely low exposure limits.
- Inhalation drug–device systems – Complex pathways and multi-material components.
- Medical devices – Require both chemical characterization and biocompatibility integration.
4. Turnaround Time and Project Flexibility
Turnaround time directly affects your clinical or commercialization timeline, making it essential to assess a lab’s scheduling capacity.
Evaluate:
- Standard TAT for extractables studies (usually 4–8 weeks)
- Leachables study setup and stability pull support
- Ability to accommodate accelerated or priority timelines
- Availability of scientific support during the study
5. Pricing Structure and Transparency
Transparent pricing allows you to compare laboratories fairly and avoid hidden costs during Extractables and Leachables (E&L) Testing in the United States.
Typical pricing models include:
- Per-study bundled package
- Per-sample cost
- Additional charges for toxicology, method development, or structural elucidation
Always request:
- Itemized quotation
- Instrumentation list
- Scope of work
- Reporting deliverables
6. Toxicological Risk Assessment Capability
Without in-house toxicology, your E&L project becomes fragmented and risks regulatory rejection.
A complete toxicology package includes:
- Identification of compounds
- Threshold of Toxicological Concern (TTC) assessment
- Permitted Daily Exposure (PDE) calculations
- Exposure modeling
- Safety justification for each leachable
This is essential for FDA review.
7. Data Integrity, QA Systems & GLP Compliance
Strong data integrity ensures the E&L report is defensible to regulatory authorities.
Look for:
- GLP compliance
- 21 CFR Part 11 electronic data integrity
- Internal QA audits
- Documented SOP structure
- Peer-reviewed reporting
- Second-scientist verification
ResolveMass provides detailed documentation aligned with ICH Q3E E&L study requirements:
https://resolvemass.ca/ich-q3e-extractables-and-leachables-el-testing-services/
8. Communication, Scientific Support & Reporting Quality
Clear communication and interpretation of findings improve the quality of regulatory submissions.
Strong reports include:
- Identification confidence levels
- Mass spectral justification
- Extractables vs. leachables correlation
- Toxicological summaries
- FDA-ready narrative
9. Long-Term Partnership & Outsourcing Preparedness
Answer upfront: Your E&L partner should support not just one test, but your entire drug development cycle.
A strong partner offers:
- Method development
- Forced extraction
- Migration studies
- Long-term stability support
- On-call scientific consultation
Conclusion
Choosing the right provider for Extractables and Leachables (E&L) Testing in the United States is essential for regulatory success. Your selection should be grounded in scientific expertise, instrumentation range, FDA-aligned study design, toxicological capability, and strong communication. By evaluating the criteria above, you can confidently select a testing partner who will support your 2025 regulatory and commercialization plans.
Get in Touch with Us
FAQs on Extractables and Leachables (E&L) Testing
E&L testing in the United States is considered more stringent because the U.S. FDA expects complete toxicological justification and comprehensive chemical identification for any potential migrant. This includes structural elucidation, toxicological risk assessment, threshold-based justification, and an evaluation of patient exposure under worst-case conditions.
For high-risk products such as sterile injectables, inhalation systems, and ophthalmic formulations, the FDA often requires more sensitive analytical methods, lower detection thresholds, and a deeper understanding of the chemical origin of each impurity. U.S. reviewers also expect laboratories to provide detailed study designs, extractable profiles, analytical validation, and toxicological interpretation as part of the submission.
Any container–closure system (CCS) that directly contacts the drug product automatically falls under extractables testing. The need for full, long-term leachables testing depends on product risk.
High-risk products — such as injectables, ophthalmic solutions, inhalation products, biologics, and products with long shelf life — typically require both extractables and full leachables studies.
Factors influencing the testing requirement include:
-Dosage form and route of administration
-Drug product pH, polarity, strength, and formulation
-Material type (elastomers, plastics, adhesives, coatings, metals, glass)
-Temperature, storage, and shipping conditions
-Patient population (pediatric, geriatric, chronic use)
Sample counts vary based on study phase and regulatory expectations.
For extractables studies, laboratories typically require 3–6 units per extraction condition, depending on the number of solvents, temperatures, and material components being tested.
For leachables studies, the requirement is higher because testing occurs over real-time and accelerated durations. Most programs need 10–20 units per time point, depending on:
-The number of stability intervals
-The complexity of the dosage form
-Whether the product uses multiple packaging components
-The sensitivity of the method
The timeline for leachables studies depends on the product’s shelf life. Most leachables studies run in parallel with long-term stability programs.
Typical timelines include:
3–6 months for accelerated intervals
6–12 months or longer for long-term intervals
The total duration can extend to 18–24 months for biologics or products with extended shelf life. Interim data is often needed for regulatory submissions.
The process of developing a new drug usually takes a total of 10 to 15 years on average. The process can be divided into three main phases: Drug discovery, the first phase in which candidate compounds are selected based on their pharmacological properties
The FDA expects the highest possible degree of identification for any compound detected above reporting thresholds.
-Level 1 (confirmed structure) is preferred — the structure is validated using orthogonal techniques.
-Level 2 (probable structure) may be acceptable if justified scientifically and supported by spectral data.
Compounds with uncertain identity must be tracked, justified, and evaluated toxicologically. For high-risk dosage forms, identification requirements are typically stricter.
Laboratories in the United States implement the principles of ICH Q3E, which outline extractables profiling, analytical method considerations, and leachables monitoring.
However, U.S. labs typically design studies with additional FDA-aligned elements, such as:
-Lower reporting thresholds
-More detailed structural elucidation requirements
-Thorough toxicology packages
-Greater emphasis on method validation and documentation
This ensures the results match FDA expectations during regulatory review.
Get in Touch with Us
Reference
- United States Pharmacopeial Convention. (n.d.). Extractables and leachables. Retrieved October 10, 2025, from https://www.usp.org/impurities/extractables-and-leachables
- Balfour, H. (2022, April 29). Advancing extractables and leachables testing. European Pharmaceutical Review. https://www.europeanpharmaceuticalreview.com/article/170814/advancing-extractables-and-leachables-testing/
- Rozio, M. G. (2025). Correcting detection and quantitation bias in extractables and leachables testing. Journal of Pharmaceutical Analysis, 15(2), 123–134. https://doi.org/10.1016/j.jpha.2025.04.004
- Balfour, H. (2022, April 29). Advancing extractables and leachables testing. European Pharmaceutical Review. https://www.europeanpharmaceuticalreview.com/article/170814/advancing-extractables-and-leachables-testing/

