
Introduction:
Forced Degradation Dexamethasone studies are critical to evaluate how dexamethasone injection behaves under extreme stress conditions and to ensure product safety and efficacy.
In pharmaceutical development, especially for injectables, understanding degradation behavior is not optional—it is a regulatory necessity. Dexamethasone, being a corticosteroid, is particularly sensitive to environmental conditions, making forced degradation studies indispensable.
These studies not only help in developing stability-indicating analytical methods but also provide deep insights into impurity formation and degradation mechanisms.
Share via:
Summary:
- Forced Degradation Dexamethasone studies are essential to understand drug stability and degradation pathways.
- These studies help identify critical impurities, degradation products, and stability-indicating methods.
- Dexamethasone injections are highly sensitive to pH, oxidation, light, and temperature.
- Regulatory authorities require comprehensive stress testing data for approval.
- A well-designed study ensures robust formulation, longer shelf life, and regulatory compliance.
1: What is Forced Degradation Dexamethasone?
Forced Degradation Dexamethasone refers to the intentional exposure of dexamethasone to extreme stress conditions to understand its degradation behavior, identify degradation products, and establish stability-indicating analytical methods.
These studies are a critical part of pharmaceutical development, especially for injectable formulations, where even minor degradation can impact safety and efficacy.
Stress Conditions Used in Forced Degradation Dexamethasone
Forced degradation studies typically involve exposing dexamethasone to the following controlled stress conditions:
- Acidic hydrolysis – using acids (e.g., HCl) to evaluate acid sensitivity
- Basic hydrolysis – using bases (e.g., NaOH) to assess alkaline instability
- Oxidative conditions – using oxidizing agents like hydrogen peroxide
- Thermal stress – exposing the drug to elevated temperatures
- Photolytic degradation – exposure to UV or visible light
Purpose of Forced Degradation Dexamethasone Studies
The primary goal of Forced Degradation Dexamethasone studies is to generate meaningful stability data that supports drug development, quality control, and regulatory approval.
Key Objectives
- Identify degradation pathways
Understand how dexamethasone breaks down under different conditions - Establish stability-indicating methods
Develop analytical methods capable of separating drug and degradation products - Detect potential impurities
Identify and quantify degradation-related impurities - Support regulatory submissions
Provide essential data required by ICH guidelines for approval
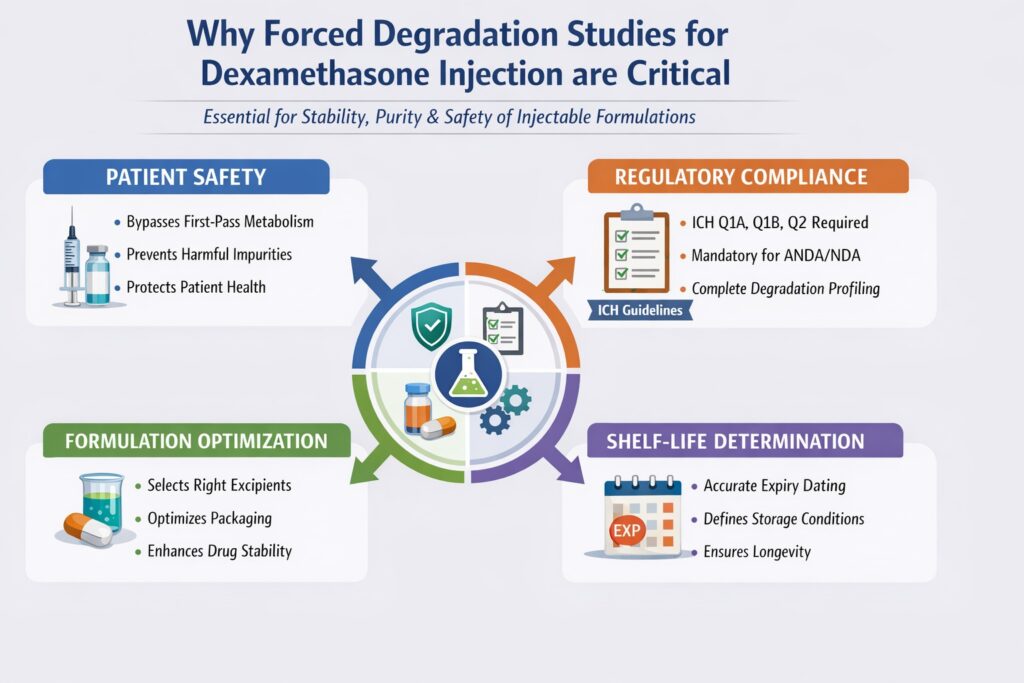
2: Why Forced Degradation Studies for Dexamethasone Injection are Critical
Forced Degradation Dexamethasone studies are essential because injectable formulations require the highest standards of stability, purity, and safety to protect patient health and meet strict regulatory expectations.
Unlike oral dosage forms, injectable drugs are administered directly into systemic circulation. This means there is zero tolerance for instability or harmful degradation products, making forced degradation studies a critical part of development.
Key Reasons
1. Patient Safety
Forced Degradation Dexamethasone studies help ensure that no harmful degradation products compromise patient safety.
- Injectable drugs bypass first-pass metabolism, increasing risk exposure
- Even trace-level impurities can lead to serious adverse effects
- Identifying degradation products early helps in risk mitigation
2. Regulatory Compliance
Forced Degradation Dexamethasone studies are mandatory to meet global regulatory standards and approval requirements.
- Required under ICH guidelines (Q1A, Q1B, Q2)
- Essential for ANDA/NDA submissions
- Regulatory bodies expect complete degradation profiling and impurity data
3. Formulation Optimization
Forced Degradation Dexamethasone studies guide formulation scientists in selecting the most stable composition.
- Helps identify sensitive formulation parameters
- Supports selection of appropriate excipients
- Assists in choosing compatible packaging systems (e.g., glass vials vs. polymer containers)
4. Shelf-Life Determination
Forced Degradation Dexamethasone studies provide critical data for assigning product shelf life and storage conditions.
- Enables accurate expiry date estimation
- Supports stability study design
- Ensures product remains safe and effective throughout its lifecycle
By integrating Forced Degradation Dexamethasone studies early in development, pharmaceutical companies can significantly reduce risks, improve product quality, and ensure smooth regulatory approval for injectable products.
3: Forced Degradation Dexamethasone: Stress Conditions and Their Impact
Forced Degradation Dexamethasone studies use specific stress conditions to reveal how dexamethasone injection degrades, helping identify unique degradation pathways and critical impurities.
Understanding how the molecule behaves under different environments is essential for developing robust, stable, and regulatory-compliant injectable formulations.
1. Acidic Hydrolysis
Acidic conditions break down dexamethasone through hydrolysis, particularly affecting ester functionalities.
- Typically performed using hydrochloric acid (HCl)
- Leads to ester hydrolysis and structural degradation
- Helps evaluate pH sensitivity in acidic environments
Impact:
- Formation of hydrolytic degradation products
- Potential loss of potency
2. Basic Hydrolysis
Basic hydrolysis is one of the most aggressive degradation pathways for dexamethasone.
- Conducted using sodium hydroxide (NaOH)
- Causes rapid degradation compared to acidic conditions
- Breaks down sensitive functional groups quickly
Impact:
- Significant impurity formation
- Useful for identifying worst-case degradation scenarios
3. Oxidative Degradation
Oxidative stress evaluates the susceptibility of dexamethasone to oxidation reactions.
- Uses hydrogen peroxide (H₂O₂)
- Targets oxidizable functional groups within the molecule
Impact:
- Formation of oxidative impurities
- Critical for understanding stability in oxygen-rich environments
4. Thermal Degradation
Thermal stress accelerates chemical reactions, revealing inherent instability in dexamethasone.
- Exposure to elevated temperatures (e.g., 60–80°C)
- Simulates long-term storage conditions in a short time
Impact:
- Accelerated degradation kinetics
- Helps predict shelf-life and storage conditions
5. Photolytic Degradation
Photolytic degradation assesses the effect of light exposure on dexamethasone stability.
- Exposure to UV and visible light
- Conducted as per ICH Q1B guidelines
Impact:
- Formation of photo-degradation and photo-oxidation products
- Important for selecting light-protective packaging
Summary Table: Stress Conditions and Impact
| Stress Condition | Reagent/Condition | Primary Impact |
|---|---|---|
| Acidic Hydrolysis | HCl | Ester hydrolysis, structural breakdown |
| Basic Hydrolysis | NaOH | Rapid degradation, impurity formation |
| Oxidative | H₂O₂ | Oxidative impurities |
| Thermal | High temperature | Accelerated instability |
| Photolytic | UV/Visible light | Photo-degradation products |
By systematically applying these stress conditions, Forced Degradation Dexamethasone studies provide deep insights into degradation behavior, enabling the development of stable, safe, and high-quality injectable products.
4: Analytical Methods for Forced Degradation Dexamethasone
Accurate analytical methods are essential in Forced Degradation Dexamethasone studies to reliably detect, separate, and quantify degradation products and impurities.
A well-developed analytical strategy ensures that all degradation pathways are clearly understood and that the method can distinguish the active drug from its related substances—this is the foundation of a stability-indicating method.
Common Techniques Used in Forced Degradation Dexamethasone
1. HPLC (High-Performance Liquid Chromatography)
HPLC is the most widely used technique for Forced Degradation Dexamethasone due to its robustness and reliability.
- Separates API and degradation products effectively
- Suitable for routine quality control and stability studies
- Compatible with multiple detectors (UV, PDA)
2. LC-MS/MS (Liquid Chromatography–Mass Spectrometry)
LC-MS/MS provides structural insights into degradation products, making it indispensable for impurity identification.
- Enables accurate mass determination
- Helps in structural elucidation of unknown impurities
- Highly sensitive and selective
3. UPLC (Ultra-Performance Liquid Chromatography)
UPLC enhances resolution and reduces analysis time in Forced Degradation Dexamethasone studies.
- Higher resolution and peak capacity
- Faster run times compared to HPLC
- Ideal for complex impurity profiling
4. UV-Visible Spectroscopy
UV-Vis spectroscopy is useful for preliminary analysis and monitoring degradation trends.
- Simple and cost-effective technique
- Useful for kinetic studies and bulk analysis
- Limited specificity compared to chromatographic methods
Key Requirements for Analytical Methods
Analytical methods used in Forced Degradation Dexamethasone must meet strict performance and regulatory criteria to ensure reliability and compliance.
1. Stability-Indicating Capability
- Must clearly separate API from all degradation products
- Should detect even minor impurities
2. High Sensitivity
- Capable of detecting trace-level impurities (ppm level)
- Critical for injectable formulations
3. Selectivity for Impurities
- Ability to distinguish between closely related compounds
- Avoid interference from excipients or matrix components
4. Method Validation (ICH Q2 Compliance)
- Must be validated for:
- Accuracy
- Precision
- Linearity
- Specificity
- Robustness
Summary Table: Analytical Techniques and Their Role
| Technique | Primary Purpose | Key Advantage |
|---|---|---|
| HPLC | Quantification | Reliable and widely accepted |
| LC-MS/MS | Identification | Structural elucidation |
| UPLC | High-resolution analysis | Faster and more sensitive |
| UV-Vis | Screening | Simple and cost-effective |
By implementing these advanced analytical techniques, Forced Degradation Dexamethasone studies can deliver highly accurate, reproducible, and regulatory-compliant data—ensuring the development of safe and stable injectable products.
5: Challenges in Forced Degradation Dexamethasone Studies
Forced Degradation Dexamethasone studies are often complex due to the drug’s inherent chemical sensitivity and the intricate nature of injectable formulations.
Achieving meaningful degradation without compromising analytical clarity requires a carefully balanced experimental approach. If not properly designed, these studies can lead to misleading results or regulatory setbacks.
Major Challenges in Forced Degradation Dexamethasone
1. Low Degradation Levels
Insufficient degradation makes it difficult to detect and characterize degradation products.
- Some stress conditions may produce minimal or no degradation
- Leads to challenges in method development and impurity identification
- May require optimization of stress intensity or duration
2. Co-eluting Impurities
Closely related degradation products may overlap during analysis, complicating separation.
- Common in chromatographic techniques like HPLC/UPLC
- Results in poor resolution and inaccurate quantification
- Requires method optimization and advanced detection techniques (e.g., LC-MS/MS)
3. Matrix Interference from Excipients
Excipients in injectable formulations can interfere with the detection of degradation products.
- Causes peak masking or suppression
- Makes impurity profiling more challenging
- Necessitates highly selective and specific analytical methods
4. Reproducibility Issues
Achieving consistent results across experiments can be difficult in Forced Degradation Dexamethasone studies.
- Variability in stress conditions (pH, temperature, time)
- Instrumental and method-related variability
- Requires strict control of experimental parameters
Practical Considerations for Reliable Studies
To overcome these challenges, Forced Degradation Dexamethasone studies must be carefully designed and controlled.
1. Optimize Stress Conditions
- Start with mild conditions, then increase gradually
- Ensure degradation is controlled and meaningful
2. Avoid Over-Degradation
- Target 5–20% degradation (as per best practices)
- Over-degradation (>20%) can lead to:
- Secondary degradation products
- Misleading impurity profiles
3. Ensure Mass Balance
- Account for total drug content + degradation products
- Confirms method accuracy and completeness
- Essential for regulatory acceptance
Key Takeaway
Successfully executing Forced Degradation Dexamethasone studies requires a balance between sufficient stress and analytical precision.
By addressing these challenges with a scientific and methodical approach, laboratories can generate robust, reproducible, and regulatory-compliant data, ensuring high-quality injectable drug development.

6: Regulatory Expectations for Forced Degradation Dexamethasone
Regulatory agencies expect comprehensive Forced Degradation Dexamethasone data to ensure drug safety and quality.
Key Guidelines
- ICH Q1A – Stability Testing
- ICH Q1B – Photostability
- ICH Q2 – Analytical Validation
Regulatory Requirements
- Identification of degradation products
- Qualification of impurities
- Stability-indicating method validation
- Detailed degradation pathways
7: Best Practices for Forced Degradation Dexamethasone Studies
Following best practices ensures reliable and regulatory-compliant Forced Degradation Dexamethasone results.
Recommended Approach
- Start with mild stress conditions
- Gradually increase stress severity
- Target 5–20% degradation
- Use orthogonal analytical techniques
Documentation
- Record all experimental conditions
- Maintain chromatographic data
- Provide impurity profiling
8: How ResolveMass Laboratories Inc. Ensures Excellence
ResolveMass Laboratories Inc. brings deep expertise in Forced Degradation Dexamethasone studies through advanced analytical capabilities and regulatory experience.
Core Strengths
- Advanced LC-MS/MS platforms
- Expertise in injectable formulations
- Strong track record in impurity profiling
- Regulatory-ready documentation
What Sets Us Apart
- Scientific, data-driven approach
- Custom-designed degradation studies
- Rapid turnaround time
- Global regulatory support
Conclusion:
Forced Degradation Dexamethasone studies are indispensable for ensuring the safety, efficacy, and regulatory compliance of dexamethasone injection products.
By understanding degradation pathways and identifying impurities early in development, pharmaceutical companies can:
- Improve formulation stability
- Reduce regulatory risks
- Ensure patient safety
Partnering with an experienced laboratory like ResolveMass ensures that your Forced Degradation Dexamethasone studies are conducted with the highest level of scientific rigor and compliance.
Frequently Asked Questions:
Forced Degradation Dexamethasone is a stress testing study used to identify degradation pathways and impurities under extreme conditions.
It helps ensure the stability, safety, and quality of dexamethasone injection.
Forced Degradation Dexamethasone studies involve multiple stress conditions to evaluate stability.
These include:
-Acidic hydrolysis
-Basic hydrolysis
-Oxidative degradation
-Thermal stress
-Photolytic degradation
A stability-indicating method in Forced Degradation Dexamethasone can accurately separate and quantify the drug and its degradation products.
It ensures reliable detection of impurities without interference.
Typically, 5–20% degradation is considered optimal in Forced Degradation Dexamethasone studies.
This ensures meaningful results without generating secondary degradation products.
Forced Degradation Dexamethasone studies face challenges related to detection and reproducibility.
Key challenges include:
-Low degradation levels
-Co-eluting impurities
-Matrix interference from excipients
-Reproducibility issues
Reference
- Chen Q, Zielinski D, Chen J, Koski A, Werst D, Nowak S. A validated, stability-indicating HPLC method for the determination of dexamethasone related substances on dexamethasone-coated drug-eluting stents. Journal of pharmaceutical and biomedical analysis. 2008 Nov 4;48(3):732-8.https://www.sciencedirect.com/science/article/pii/S0731708508003853
- Matter B, Ghaffari A, Bourne D, Wang Y, Choi S, Kompella UB. Dexamethasone degradation in aqueous medium and implications for correction of in vitro release from sustained release delivery systems. AAPS PharmSciTech. 2019 Oct 23;20(8):320.https://link.springer.com/article/10.1208/s12249-019-1508-7
- Ummiti K, Vakkala S, Panuganti V, Annarapu MR. Isolation, identification, and characterization of 17-oxo dexamethasone, an oxidative degradation impurity of dexamethasone using flash chromatography and NMR/HRMS/IR. Journal of Liquid Chromatography & Related Technologies. 2014 Oct 21;37(17):2403-19.https://www.tandfonline.com/doi/abs/10.1080/10826076.2013.836712
- Deshmukh R, Sharma L, Tekade M, Kesharwani P, Trivedi P, Tekade RK. Force degradation behavior of glucocorticoid deflazacort by UPLC: isolation, identification and characterization of degradant by FTIR, NMR and mass analysis. Journal of biomedical research. 2016 Feb 20;30(2):149.https://pmc.ncbi.nlm.nih.gov/articles/PMC4820892/
- Wu ZZ, Thatcher ML, Lundberg JK, Ogawa MK, Jacoby CB, Battiste JL, Ledoux KA. Forced degradation studies of corticosteroids with an alumina–steroid–ethanol model for predicting chemical stability and degradation products of pressurized metered-dose inhaler formulations. Journal of pharmaceutical sciences. 2012 Jun 1;101(6):2109-22.https://www.sciencedirect.com/science/article/pii/S0022354915315756


