.
.
.
Generic Drug Development CDMO

A Generic Drug Development CDMO plays a critical role in helping pharmaceutical companies transform drug concepts into commercially viable generic medicines. At ResolveMass Laboratories Inc., we partner with pharmaceutical innovators, generic drug manufacturers, and biotech companies to provide end-to-end development and manufacturing support. Our integrated approach combines analytical science, formulation expertise, regulatory knowledge, and scalable manufacturing capabilities to accelerate generic drug programs while ensuring strict compliance with global regulatory standards. By leveraging advanced technologies and experienced scientific teams, we help clients navigate complex development pathways, reduce development timelines, and bring high-quality generic medicines to market efficiently.
Explore our full suite of professional support: Access Comprehensive Chemistry, Manufacturing, and Controls (CMC) Services
Share via:
What is a Generic Drug Development CDMO?
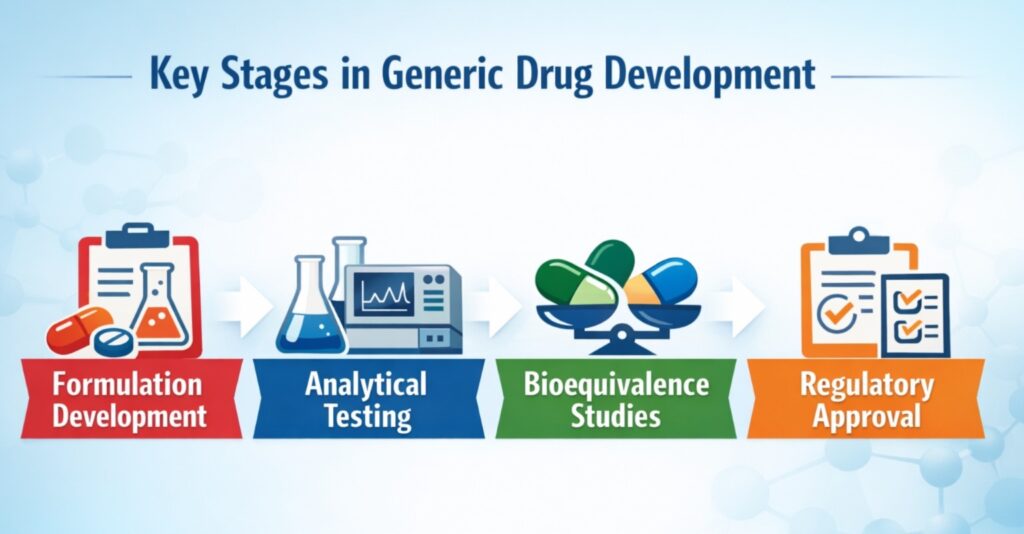
A Generic Drug Development CDMO (Contract Development and Manufacturing Organization) provides specialized services to pharmaceutical companies that aim to develop and commercialize generic versions of branded drugs. These organizations support the entire lifecycle of drug development, including:
- Pre-formulation and formulation development
- Analytical method development and validation
- Bioequivalence study support
- Stability studies and regulatory documentation
- Process optimization and scale-up
- Clinical and commercial manufacturing
By partnering with an experienced CDMO, pharmaceutical companies can leverage established infrastructure, regulatory expertise, and advanced scientific capabilities without the need to build costly in-house facilities.
Optimize your outsourcing strategy: Learn more about our specialized CMC CRO Services
Why Choose ResolveMass as Your Generic Drug Development CDMO
ResolveMass Laboratories Inc. has built a reputation for scientific precision, regulatory expertise, and reliable pharmaceutical development support. Our multidisciplinary teams bring deep experience in analytical chemistry, pharmaceutical formulation, and regulatory compliance to support complex generic drug programs.
Scientific Expertise
Our scientists specialize in analytical characterization, impurity profiling, and formulation optimization to ensure reliable and reproducible generic drug development.
Regulatory Compliance
We design development strategies aligned with global regulatory expectations, including FDA, Health Canada, and international regulatory guidelines.
Advanced Analytical Infrastructure
Our laboratories are equipped with modern analytical instrumentation capable of supporting complex drug product analysis and validation.
Ensure your filing meets every benchmark: Understand the key CMC Requirements for NDA Submissions
Our Generic Drug Development Services
ResolveMass offers a comprehensive suite of services designed to support every stage of generic pharmaceutical development.
- Pre-Formulation Studies
- Understanding the physical and chemical properties of active pharmaceutical ingredients to establish optimal formulation strategies.
- Formulation Development
- Designing stable, bioequivalent formulations that meet regulatory requirements and therapeutic equivalence standards.
- Analytical Method Development
- Developing robust analytical methods for accurate characterization, impurity detection, and quality control.
- Method Validation
- Ensuring analytical procedures meet regulatory guidelines for precision, accuracy, and reliability.
- Stability Testing
- Evaluating drug product stability under various environmental conditions to determine shelf life.
- Regulatory Documentation Support
- Assisting clients with regulatory submission packages including data generation and technical documentation.
Strengthen your technical data: Discover our Analytical Method Development for IND and NDA
Our Development Approach
As a trusted Generic Drug Development CDMO, our development process focuses on scientific rigor, regulatory compliance, and client collaboration. Each project begins with a detailed assessment of the target reference listed drug (RLD), followed by systematic formulation design, analytical characterization, and process optimization.
Our teams emphasize data-driven decision making, ensuring every stage of development is supported by validated scientific evidence. This approach minimizes risk during scale-up and regulatory review.
Identify and mitigate potential hurdles: How to Manage CMC Risk in Drug Development
Industries We Support
- Generic pharmaceutical companies
- Biotech startups entering the generics market
- Pharmaceutical manufacturers expanding product portfolios
- Contract research organizations requiring analytical support
- Global pharmaceutical companies developing complex generics
Benefits of Partnering with a Generic Drug Development CDMO
Collaborating with a specialized CDMO allows pharmaceutical companies to accelerate development timelines while maintaining the highest quality standards.
- Faster development timelines
- Access to specialized scientific expertise
- Reduced infrastructure investment
- Improved regulatory readiness
- Scalable manufacturing solutions
- Advanced analytical capabilities
Why the Global Demand for Generic Drug Development is Growing
The global pharmaceutical industry continues to see significant demand for generic medicines due to patent expirations, rising healthcare costs, and increased access to healthcare worldwide. Generic drugs provide therapeutically equivalent alternatives to branded medications at significantly lower costs.
As more companies enter the generics market, partnering with a knowledgeable Generic Drug Development CDMO becomes essential to ensure successful product development, regulatory approval, and commercialization.
Partner with ResolveMass Laboratories Inc.
ResolveMass Laboratories Inc. is committed to delivering reliable, science-driven solutions for pharmaceutical development. As an experienced Generic Drug Development CDMO, we provide the analytical insight, development expertise, and regulatory understanding required to support successful generic drug programs from concept to commercialization.
Our collaborative approach ensures that each client receives tailored development strategies designed to meet their specific project goals while maintaining strict compliance with global pharmaceutical standards.
Get answers to your technical questions: Read our Chemistry, Manufacturing, and Controls (CMC) Services FAQs
Contact Our CDMO Experts Today – Contact us
Frequently Asked Questions (FAQs)
Pharmaceutical companies often collaborate with CDMOs to access specialized expertise, advanced laboratory infrastructure, and experienced scientific teams. This partnership allows companies to accelerate development timelines without investing in expensive internal facilities. It also helps ensure that development processes align with global regulatory expectations.
Generic drug development usually involves pre-formulation research, formulation optimization, analytical method development, stability studies, and validation testing. CDMOs may also provide regulatory support and assistance with documentation for drug approval submissions. These services help ensure the generic product performs similarly to the reference listed drug.
Generic medicines must demonstrate bioequivalence with the original branded drug and meet strict quality, safety, and efficacy standards. Regulatory agencies such as the FDA, EMA, and Health Canada require extensive analytical data and stability testing. CDMOs help ensure that development programs follow these regulatory guidelines from the beginning.
Analytical testing plays a critical role in verifying the identity, purity, strength, and stability of a generic drug product. Techniques such as chromatography, dissolution testing, and impurity profiling are commonly used. These tests confirm that the generic formulation matches the performance characteristics of the reference drug.
Yes, many CDMOs support clients by preparing scientific data, analytical reports, and technical documentation required for regulatory submissions. Their experience with regulatory guidelines helps ensure that development data is generated in a compliant manner. This support can improve the efficiency of the approval process.
CDMOs support the development of a wide range of pharmaceutical dosage forms, including tablets, capsules, injectables, topical formulations, and modified-release products. Their expertise allows them to address formulation challenges across different drug delivery systems. This flexibility helps pharmaceutical companies expand their generic product pipelines.
Reference:
- Wahlich, J. (2021). Continuous manufacturing of small molecule solid oral dosage forms. Pharmaceutics, 13(8), 1248. https://doi.org/10.3390/pharmaceutics13081248
- Pandhare, S. D., & Wamane, V. B. (2023). Generic drug development and current scenario. International Journal for Multidisciplinary Research, 5(3). https://doi.org/10.36948/ijfmr.2023.v05i03.2784
