
Introduction:
The Generic Drug Development Process ANDA is a scientifically driven pathway that enables pharmaceutical companies to develop affordable and high-quality generic medicines. Instead of conducting full clinical trials, companies must prove that their product is equivalent to the reference listed drug (RLD) in terms of quality, safety, and performance.
However, many companies face delays because they do not fully understand the step-by-step Drug Development Process ANDA. Analytical challenges, impurity issues, bioequivalence failures, and incomplete regulatory planning are among the most common reasons for rejection.
This article provides a detailed, practical, and structured explanation of the Generic Drug Development Process ANDA so that pharmaceutical companies can better plan their development strategy and avoid costly mistakes. If you want to understand how an early bioanalytical strategy in drug development improves success rates, this guide will help you understand the entire process.
Share via:
Summary:
- The Generic Drug Development Process ANDA is a structured pathway used to demonstrate that a generic drug is equivalent to the reference listed drug (RLD).
- The process includes product selection, analytical characterization, formulation development, bioequivalence studies, and regulatory submission.
- Early analytical strategy and impurity profiling significantly reduce regulatory risk. Companies that work with mass spectrometry experts in drug development often improve data quality and reduce development timelines.
- Strong method development and validation are critical for successful ANDA approval. Many pharmaceutical companies also rely on analytical development for generic drugs Canada to strengthen regulatory submissions.
- This article explains the Generic Drug Development Process ANDA step by step in a practical and easy-to-understand format.
1: What is the Generic Drug Development Process ANDA?
The Generic Drug Development Process ANDA is the structured sequence of scientific, analytical, formulation, and regulatory steps required to submit an Abbreviated New Drug Application (ANDA) to the U.S. FDA and obtain approval for a generic drug.
In simple terms, the goal of the Generic Drug Development Process ANDA is to prove that a generic drug is equivalent to the reference listed drug (RLD) in terms of quality, safety, and performance.
To achieve this, companies must demonstrate:
- The same active pharmaceutical ingredient (API)
- The same strength and dosage form
- The same performance in the human body (bioequivalence)
- The same quality, purity, and stability standards
Unlike innovative drug development, the Drug Development Process ANDA focuses heavily on analytical proof of equivalence. This is why many companies rely on bioanalysis in Canada and analytical method development for generic drugs to build a strong scientific foundation.
Step 1: Product Selection in the Generic Drug Development Process ANDA
The first step in the Drug Development Process ANDA is selecting the right product based on scientific feasibility, regulatory strategy, and market potential. If the product selection is wrong, the entire development program may fail before it even begins.
A strong product selection strategy often begins with reverse engineering for development of generic drugs to understand the reference product before development begins.
What happens in this stage?
During this stage, pharmaceutical companies carefully evaluate whether a product is suitable for generic development. The key activities include:
- Identification of the reference listed drug (RLD)
- Evaluation of patent expiry and regulatory exclusivity
- Assessment of formulation complexity
- Initial review of impurity and analytical feasibility
- Market demand and competition analysis
Why this step is critical in the Generic Drug Development Process ANDA
Many generic drug projects fail not because of poor execution, but because the product was not evaluated properly in the beginning. Without a strong feasibility assessment, companies may face analytical challenges, formulation difficulties, or regulatory delays later in the Drug Development Process ANDA.
A well-planned product selection stage significantly improves the success rate, reduces development risk, and helps companies save both time and investment.
Step 2: Reference Listed Drug (RLD) Characterization in the Generic Drug Development Process ANDA
The second step in the Drug Development Process ANDA is detailed analytical characterization of the reference listed drug (RLD). This stage creates the scientific foundation required to develop a generic drug that matches the quality, performance, and stability of the reference product.
This stage typically involves advanced analytical testing and support from peptide characterization in drug development or peptide analytical characterization services when the product involves complex molecules.
Strong RLD characterization significantly reduces regulatory risk and improves formulation success.
What happens during RLD characterization?
In this stage, the reference drug is thoroughly analyzed using advanced analytical techniques to understand its composition and behavior. The key analytical activities include:
- API identification and confirmation
- Impurity profiling
- Dissolution behavior analysis
- Stability testing
- Excipients identification
- Evaluation of physical and chemical properties
Why RLD characterization is important in the Generic Drug Development Process ANDA
Without a clear understanding of the reference product, it is not possible to develop a generic drug that performs the same way in the human body. Weak characterization often leads to formulation failure, bioequivalence issues, or regulatory delays later in the Drug Development Process ANDA.
Strong analytical characterization at this stage reduces regulatory risk, improves formulation success, and significantly increases the chances of ANDA approval.
Step 3: API Development in the Generic Drug Development Process ANDA
The third step in the Drug Development Process ANDA is the development and full characterization of the active pharmaceutical ingredient (API). This stage ensures that the API used in the generic product meets regulatory quality requirements and matches the performance of the reference listed drug.
Many pharmaceutical companies outsource early API characterization to outsourced chemistry in drug discovery or work with a generic pharmaceutical CDMO Canada to improve efficiency and reduce development timelines.
What are the main objectives of API development?
During this stage, pharmaceutical companies focus on understanding the chemical and physical properties of the API and ensuring impurity control. The key objectives include:
- Identification and control of impurities
- Understanding degradation pathways
- Development of analytical testing methods
- Confirmation of physical and chemical properties
- Evaluation of stability and compatibility with excipients
Why API development matters in the Generic Drug Development Process ANDA
Regulatory authorities expect a well-defined impurity profile and complete scientific understanding of the API. Weak API characterization can lead to regulatory queries, delays, or even rejection of the ANDA submission.
A strong API development strategy not only improves product quality but also increases the overall success rate of the Drug Development Process ANDA.
Step 4: Formulation Development in the Generic Drug Development Process ANDA
The fourth step in the Drug Development Process ANDA is formulation development, where the generic product is designed to match the performance of the reference listed drug (RLD). This stage is critical because even small formulation differences can lead to bioequivalence failure.
Companies often accelerate formulation programs by outsourcing generic drug development Canada or working with a CDMO for generic drug development in Canada.
What happens during formulation development?
In this stage, pharmaceutical scientists develop and optimize the formulation so that it behaves the same way as the reference drug. The key activities include:
- Selection of suitable excipients
- Optimization of the dissolution profile
- Prototype formulation development
- Evaluation of physical stability
- Compatibility studies between API and excipients
What is the goal of formulation development in the Generic Drug Development Process ANDA?
The main goal is to match the dissolution profile, stability, and performance of the reference drug as closely as possible. If the formulation is not optimized properly, the generic drug may fail in bioequivalence studies, which can significantly delay the Drug Development Process ANDA.
A well-optimized formulation not only improves the chances of regulatory approval but also reduces development timelines and overall project risk.
Step 5: Analytical Method Development in the Generic Drug Development Process ANDA
The fifth step in the Drug Development Process ANDA is analytical method development, which ensures that the quality, safety, and equivalence of the generic drug can be accurately demonstrated. Without reliable analytical methods, it is not possible to generate regulatory-acceptable data.
Many companies rely on analytical method development for generic drugs or mass spectrometry experts in drug development to generate regulatory-ready data.
What methods are typically developed?
During this stage, pharmaceutical companies develop multiple analytical methods to evaluate the drug product and API. The most important methods include:
- Assay methods for determining drug strength
- Impurity testing methods for identifying and quantifying impurities
- Dissolution methods to compare performance with the reference drug
- Stability-indicating methods to monitor degradation over time
- Methods for content uniformity and related substances
Why this step is critical in the Generic Drug Development Process ANDA
Analytical data forms the foundation of the ANDA submission. If the analytical methods are not robust, sensitive, and well-optimized, the regulatory agency may question the accuracy of the results, which can lead to delays or additional studies.
Strong analytical method development improves data quality, reduces regulatory risk, and significantly increases the success rate of the Drug Development Process ANDA.
Step 6: Method Validation in the Generic Drug Development Process ANDA
The sixth step in the Drug Development Process ANDA is method validation, which ensures that all analytical methods used in development and submission are reliable, accurate, and acceptable to regulatory authorities. Without proper validation, the data generated during development cannot be used in an ANDA submission.
Regulatory-ready validation strategies are often supported by regulatory support for generic drugs US and Canada CDMO to reduce submission risk.
What validation parameters are evaluated?
During this stage, analytical methods are validated according to regulatory guidelines to confirm their reliability and performance. The key validation parameters include:
- Accuracy
- Precision
- Specificity
- Linearity
- Robustness
- Limit of detection (LOD) and limit of quantification (LOQ)
Why method validation is important in the Generic Drug Development Process ANDA
Regulatory agencies only accept data generated using validated analytical methods. If the methods are not properly validated, the entire analytical data package may be questioned, which can lead to delays, additional studies, or even rejection of the submission.
Validated methods ensure that the analytical data is reliable, reproducible, and scientifically sound, which significantly increases the success rate of the Drug Development Process ANDA.
Step 7: Bioequivalence Studies in the Generic Drug Development Process ANDA
The seventh step in the Drug Development Process ANDA is conducting bioequivalence studies to prove that the generic drug performs the same way in the human body as the reference listed drug (RLD). This is one of the most critical stages because regulatory approval depends heavily on bioequivalence results.
Companies planning complex molecules such as peptides often rely on peptide testing service or peptide sameness study services in Canada to ensure regulatory success.
What is the objective of bioequivalence studies?
The primary objective is to demonstrate that the rate and extent of absorption of the generic drug are similar to the reference drug. In simple terms, both products must deliver the same amount of drug into the bloodstream at a similar speed.
What are the key components of bioequivalence studies?
Bioequivalence studies involve multiple scientific and regulatory steps, including:
- Study design and protocol development
- Sample analysis using validated analytical methods
- Pharmacokinetic evaluation
- Statistical analysis of bioequivalence parameters
- Preparation of regulatory-ready study reports
Why bioequivalence studies are critical in the Generic Drug Development Process ANDA
A successful bioequivalence study proves therapeutic equivalence and significantly increases the chances of ANDA approval. However, if the study fails, companies may need to reformulate the product, which can delay the entire Drug Development Process ANDA.
Step 8: Stability Studies in the Generic Drug Development Process ANDA
The eighth step in the Drug Development Process ANDA is conducting stability studies to prove that the generic drug maintains its quality, safety, and effectiveness throughout its shelf life. Regulatory authorities require stability data before approving any ANDA submission.
For complex molecules such as GLP-1 drugs, stability and analytical characterization are often supported by GLP-1 peptide analytical characterization and peptide characterization case study of semaglutide.
What does stability testing include?
During this stage, the finished drug product is tested under different storage conditions to evaluate its long-term performance. Stability testing typically includes:
- Long-term stability studies
- Accelerated stability studies
- Degradation analysis
- Shelf-life determination
- Evaluation of physical, chemical, and microbiological stability
Why stability studies are essential in the Generic Drug Development Process ANDA
Regulatory agencies must be confident that the generic drug will remain safe and effective until the end of its shelf life. Weak or incomplete stability data can delay approval or result in additional regulatory queries during the Drug Development Process ANDA.
Strong stability studies not only support regulatory approval but also improve product reliability and market success.
Step 9: ANDA Submission in the Generic Drug Development Process ANDA
The final step in the Drug Development Process ANDA is preparing and submitting the complete ANDA dossier to the U.S. FDA for regulatory approval. This stage brings together all scientific, analytical, and regulatory data generated during development.
Many pharmaceutical companies speed up approval timelines by working with a generic drug development CRO for ANDA or a pharmaceutical CDMO US and Canada.
What is included in the ANDA submission?
The ANDA submission must contain a complete set of regulatory documents and scientific data that demonstrate the quality, safety, and equivalence of the generic drug. The key components include:
- Analytical data
- Method validation reports
- Stability data
- Bioequivalence study reports
- Manufacturing and quality information
- Regulatory documentation and technical reports
Why ANDA submission is critical in the Generic Drug Development Process ANDA
A well-prepared submission significantly increases the chances of faster approval and reduces the risk of regulatory queries. Incomplete or poorly organized data can lead to delays, additional questions from the FDA, or even rejection of the application during the Drug Development Process ANDA.
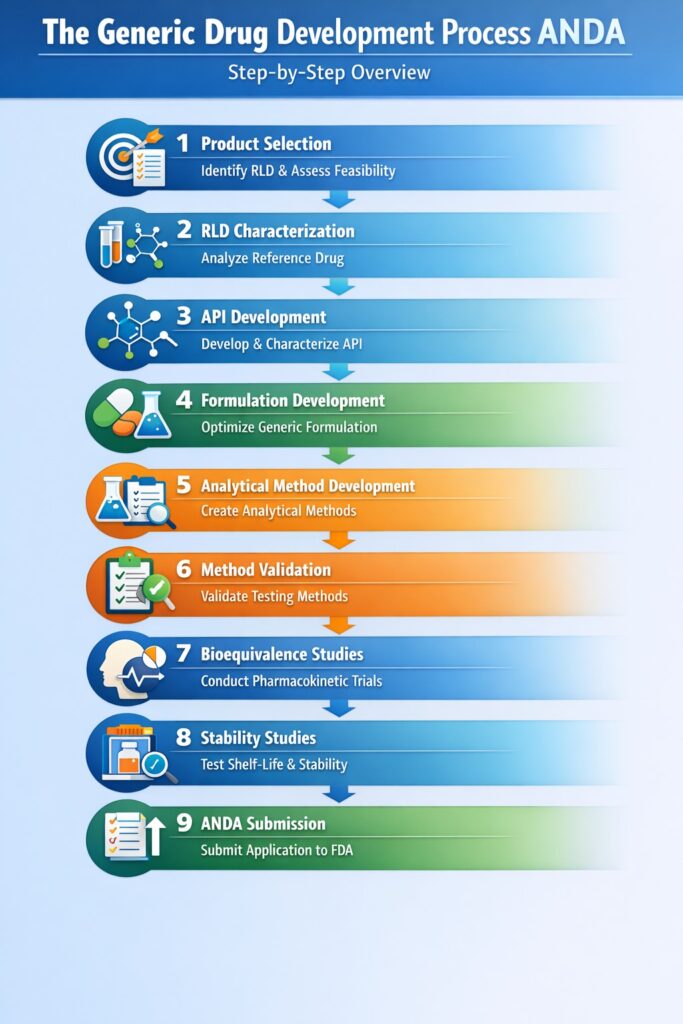
2: Step-by-Step Table of the Generic Drug Development Process ANDA
| Step | Stage | Objective |
|---|---|---|
| 1 | Product Selection | Identify a feasible generic product and evaluate market and regulatory potential |
| 2 | RLD Characterization | Understand the reference listed drug (RLD) through detailed analytical testing |
| 3 | API Development | Control impurities and confirm physical and chemical properties of the API |
| 4 | Formulation Development | Match the performance and dissolution profile of the reference drug |
| 5 | Analytical Method Development | Generate reliable and regulatory-acceptable analytical data |
| 6 | Method Validation | Ensure analytical methods meet regulatory compliance requirements |
| 7 | Bioequivalence Studies | Prove therapeutic equivalence between generic and reference drug |
| 8 | Stability Studies | Confirm product stability and establish shelf life |
| 9 | ANDA Submission | Submit the complete regulatory dossier and obtain FDA approval |
3: Why Expertise Matters in the Generic Drug Development Process ANDA
The Drug Development Process ANDA is not just a regulatory process—it is a highly scientific and analytical pathway. Companies that rely only on formulation development often face delays because they underestimate the importance of analytical characterization and impurity control.
Companies that work with experienced partners such as a CDMO for generic projects in Canada or a CDMO accelerate generic drug development US and Canada significantly improve approval success rates.
Why expert support is important
Generic drug development involves multiple complex steps, and each stage must meet strict regulatory expectations. Working with experienced analytical and regulatory experts helps companies avoid common mistakes and improve the overall success rate of the Drug Development Process ANDA.
Key benefits of working with experienced experts
- Reduced development timelines
- Improved analytical data quality
- Better impurity identification and control
- Minimized regulatory risk
- Higher probability of ANDA approval
- Faster resolution of regulatory queries
A strong scientific and regulatory strategy, supported by experienced experts, can significantly improve the efficiency and success of the Generic Drug Development Process ANDA.
Conclusion:
The Generic Drug Development Process ANDA is a structured, science-driven pathway that requires strong analytical expertise, formulation knowledge, and regulatory understanding. From product selection to final submission, every stage plays a critical role in determining the success of a generic drug.
Companies that follow a step-by-step Generic Drug Development Process ANDA and invest in early analytical strategy can significantly reduce development risk and accelerate approval timelines.
Frequently Asked Questions:
The Generic Drug Development Process ANDA is the step-by-step scientific and regulatory pathway used to develop a generic drug and obtain approval from the U.S. FDA. It focuses on proving that the generic drug is equivalent to the reference listed drug (RLD) in quality, safety, and performance.
Companies can improve success rates by starting with proper product selection, performing strong analytical characterization, developing robust analytical methods, and working with experienced analytical and regulatory experts.
The most common reasons include poor analytical characterization, weak impurity control, failed bioequivalence studies, incomplete stability data, and poorly validated analytical methods.
An ANDA submission typically includes analytical data, method validation reports, stability studies, bioequivalence study reports, manufacturing information, and regulatory documentation.
Bioequivalence studies are clinical studies designed to prove that the generic drug delivers the same amount of drug into the bloodstream at the same rate as the reference drug. These studies are one of the most important requirements for ANDA approval.
Reference
- Emily M, Ioanna N, Scott B, Beat F. Reflections on FDA draft guidance for products containing nanomaterials: is the abbreviated new drug application (ANDA) a suitable pathway for nanomedicines?. The AAPS journal. 2018 Aug 20;20(5):92.https://link.springer.com/article/10.1208/s12248-018-0255-0
- Beers DO, Karst KR. Generic and innovator drugs: A guide to FDA approval requirements. Wolters Kluwer; 2013 May 22.https://books.google.com/books?hl=en&lr=&id=b8WE21vXtoUC&oi=fnd&pg=PR21&dq=Step-by-Step+Process+of+Generic+Drug+Development+for+ANDA+Submission&ots=KS0a2Wk26y&sig=dOo-9UFIq75htCWUoHsONHg6N9A
- MARZO A. Clinical pharmacokinetic registration file for NDA and ANDA procedures. Pharmacological research. 1997 Dec 1;36(6):425-50.https://www.sciencedirect.com/science/article/pii/S1043661897902541
- Kumar, K.A., Bose, P.S.C., Sathyanarayana, M.B., Vasantharaju, S.G., Ligade, V., Amatha, S., Osmani, R.A., Barwa, R., Pawar, S.D. and Ravi, G., 2025. Strategic approaches for selecting regulatory pathways in drug-device combination products: A comprehensive review. Journal of Applied Pharmaceutical Science, 15(10), pp.003-021.https://japsonline.com/abstract.php?article_id=4604&sts=2


