.
.
.
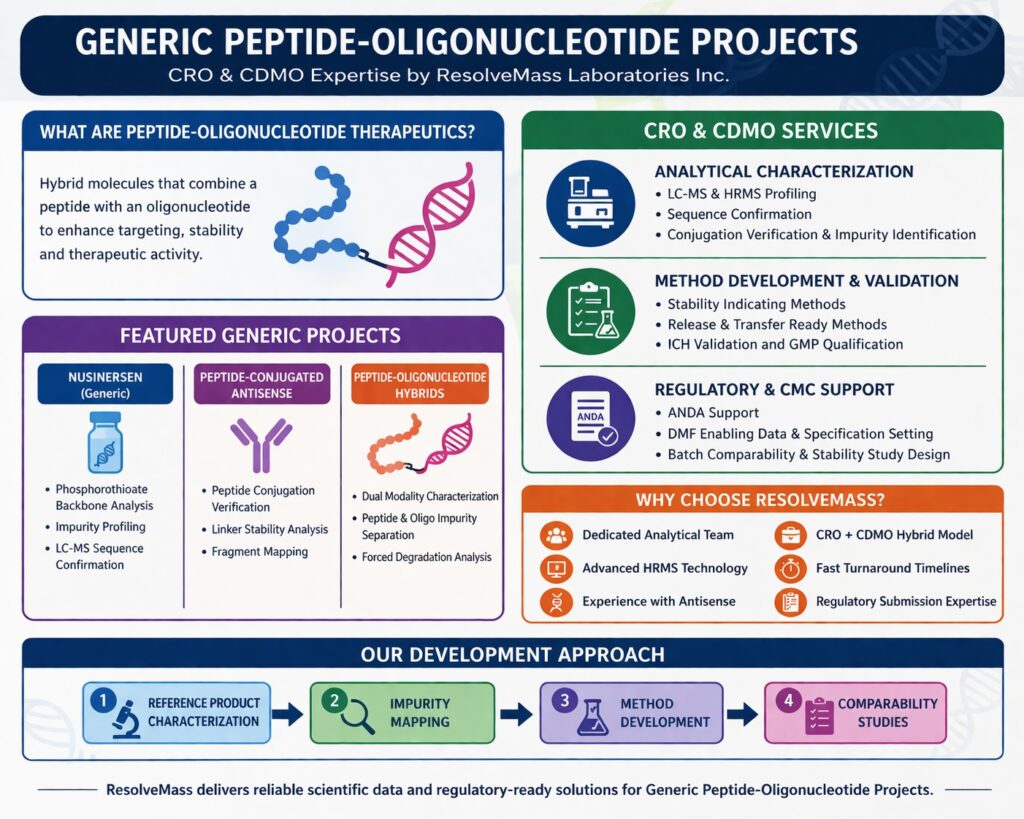
Generic Peptide-Oligonucleotide Projects

ResolveMass Laboratories Inc. supports pharmaceutical and biotech companies working on Generic Peptide-Oligonucleotide Projects with specialized CRO and CDMO services spanning analytical characterization, method development, impurity profiling, and regulatory-ready documentation. Our team has hands-on experience supporting complex peptide-oligonucleotide hybrid therapeutics including phosphorothioate oligonucleotides, conjugated peptides, and modified backbone chemistries.
Explore our Peptide-Oligonucleotide Conjugation Services
Share via;
CRO & CDMO Services for Generic Peptide-Oligonucleotide Development
Analytical Characterization
- LC-MS and HRMS profiling
- Sequence confirmation
- Conjugation verification
- Impurity identification
- Degradation pathway analysis
Method Development & Validation
- Stability indicating methods
- Release method development
- Transfer ready methods
- ICH validation support
- GMP method qualification
Regulatory & CMC Support
- ANDA support
- DMF enabling data Specification setting
- Batch comparability
- Stability study design
View our CMC Services for Peptide-Oligonucleotide Conjugates
Featured Generic Peptide-Oligonucleotide Projects
ResolveMass Laboratories Inc. has expertise supporting complex peptide-oligonucleotide therapeutics. Individual project pages will provide deeper technical scope and development approaches.
Nusinersen (Generic Development Support)
- Phosphorothioate backbone characterization
- Oligonucleotide impurity profiling
- LC-MS sequence confirmation
- Stability indicating method development
- Comparative analytical studies
Peptide-Conjugated Antisense Oligonucleotides
- Peptide conjugation verification
- Linker stability analysis
- Fragment mapping
- Conjugate purity assessment
Peptide-Oligonucleotide Hybrids
- Dual modality characterization
- Peptide and oligo impurity separation
- Mass balance studies
- Forced degradation analysis
Learn more about POC Synthesis and Characterization
Why Choose ResolveMass Laboratories Inc.
- Dedicated peptide-oligonucleotide analytical team
- Advanced HRMS instrumentation
- Experience with antisense therapeutics
- CRO + CDMO hybrid support model
- Fast turnaround timelines
- Regulatory submission experience
- Confidential generic development programs
- Custom method development
- Stability and degradation expertise Scalable analytical support
Technical Capabilities for Peptide-Oligonucleotide Generics
Generic peptide-oligonucleotide therapeutics require specialized analytical workflows. ResolveMass Laboratories Inc. applies orthogonal techniques to ensure structural confirmation, impurity identification, and comparability assessment. Our workflows are designed to support ANDA submissions and global regulatory filings while ensuring robustness and reproducibility.
- High resolution LC-MS characterization
- Ion pairing LC method development
- Oligonucleotide mapping
- Peptide mapping
- Conjugate analysis
- Impurity structural elucidation
- Forced degradation studies
- Stability indicating methods
- Counterion analysis
- Comparative analytical studies
Review our QC Testing for Peptide-Oligonucleotide Conjugates
Our Development Approach for Generic Peptide-Oligonucleotide Projects
ResolveMass follows a structured development model starting with reference product characterization, followed by impurity mapping, method development, and comparability studies. This systematic approach ensures regulatory alignment and reduces development risk. Our team collaborates closely with clients to tailor analytical strategies for each molecule, whether standalone oligonucleotide, peptide conjugate, or hybrid therapeutic.
Discuss Your Generic Peptide-Oligonucleotide Project
ResolveMass Laboratories Inc. partners with pharmaceutical and biotech companies working on Generic Peptide-Oligonucleotide Projects, including Nusinersen and other peptide-conjugated antisense therapeutics. Our CRO and CDMO services help accelerate development while maintaining regulatory compliance and analytical rigor.
Frequently Asked Questions
These molecules contain both peptide and oligonucleotide components, often with modified backbones, linkers, and conjugates. This increases complexity in separation, detection, and impurity identification. Advanced LC-MS workflows and orthogonal techniques are required to ensure accurate characterization and comparability.
Nusinersen generics require sequence confirmation, phosphorothioate impurity profiling, stability indicating methods, degradation studies, and comparative analytics. CROs provide LC-MS characterization, method validation, and regulatory documentation to support ANDA submissions.
Comparability is demonstrated by analyzing the generic candidate alongside the reference product using orthogonal analytical techniques. Parameters such as impurity profile, sequence integrity, and stability behavior are evaluated. Matching these attributes helps confirm similarity and supports regulatory expectations.
Common impurities include truncated sequences, deletion variants, oxidation products, and conjugation-related byproducts. Degradation during storage may also create additional variants. Sensitive analytical methods are needed to detect and identify these low-level species accurately.
Regulatory submissions typically require comprehensive analytical comparability with the reference product. Agencies expect detailed impurity profiling, validated methods, and structural confirmation. Proper documentation and scientifically justified specifications are also important.
Early CRO involvement helps identify analytical challenges before development progresses too far. Initial reference product characterization can guide method development and risk assessment. This approach reduces delays and improves overall development efficiency.
Reference:
- Klabenkova, K., Fokina, A., & Stetsenko, D. (2021). Chemistry of peptide-oligonucleotide conjugates: A review. Molecules, 26(17), 5420. https://doi.org/10.3390/molecules26175420
- Venkatesan, N., & Kim, B. H. (2006). Peptide conjugates of oligonucleotides: Synthesis and applications. Chemical Reviews, 106(9), 3712–3761. https://doi.org/10.1021/cr0502448
- Malinowska, A. L., Huynh, H. L., & Bose, S. (2024). Peptide-oligonucleotide conjugation: Chemistry and therapeutic applications. Current Issues in Molecular Biology, 46(10), 11031–11047. https://doi.org/10.3390/cimb46100655
