.
.
.
Semaglutide Generic Drug Development Services

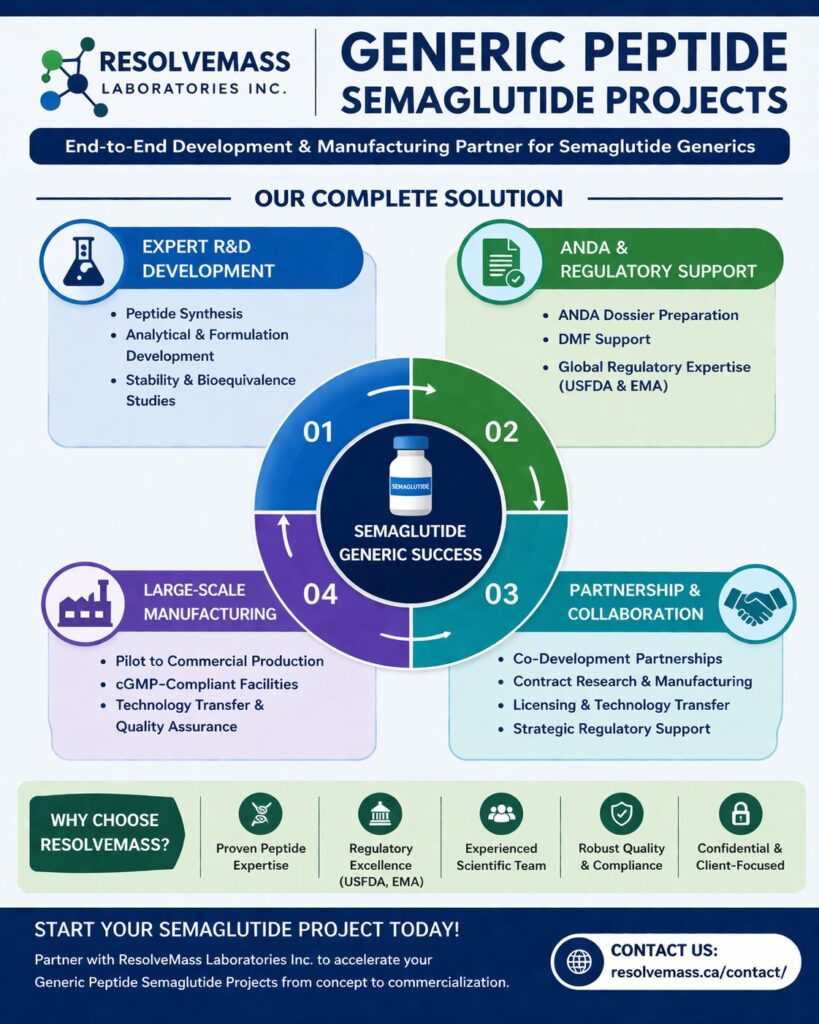
ResolveMass Laboratories Inc. specializes in Generic Peptide Semaglutide Projects, delivering end-to-end solutions from early-stage research to ANDA submission and commercial-scale manufacturing. With deep expertise in peptide chemistry, analytical development, and regulatory compliance, we support pharmaceutical companies in accelerating their semaglutide generic programs efficiently and reliably. Our integrated capabilities ensure seamless execution, reduced timelines, and regulatory success in highly competitive markets.
Our Expertise in Generic Semaglutide Development
We bring extensive experience in developing complex peptide-based generics like semaglutide. Our scientific team combines advanced analytical techniques, formulation knowledge, and regulatory insights to deliver high-quality outcomes.
- Peptide synthesis and impurity profiling
- Analytical method development and validation
- Formulation development (injectables and oral delivery)
- Stability studies as per ICH guidelines
- Bioequivalence strategy support
- Regulatory documentation for ANDA submission
Explore our specialized approach to peptide drug development: Learn more about our Specialized Peptide CDMO Services
End-to-End ANDA Support for Semaglutide Projects
ResolveMass Laboratories Inc. offers complete R&D services required for ANDA submission for semaglutide generics. Our structured approach ensures compliance with global regulatory standards while minimizing development risks.
Development
- API characterization
- Analytical development
- Formulation optimization
Regulatory
- ANDA dossier preparation
- DMF support
- Regulatory gap assessment
Validation
- Process validation
- Analytical validation
- Stability studies
Understand the roadmap for successful generic filings: Discover the Generic Drug Development Process for ANDA
Large-Scale Manufacturing Capabilities
We provide scalable and compliant manufacturing solutions for semaglutide projects. Our facilities are designed to meet global regulatory expectations, ensuring consistent product quality and supply chain reliability.
- Pilot to commercial-scale manufacturing
- cGMP-compliant production facilities
- Technology transfer support
- Batch consistency and quality assurance
- Supply chain and distribution readiness
See how we support generic launches across North America: Learn about Pharmaceutical CDMO services in the US and Canada
Partnership & Collaboration Opportunities
We actively collaborate with pharmaceutical companies, biotech firms, and investors looking to enter or expand in Generic Peptide Semaglutide Projects. Whether you require full development support or strategic partnership, ResolveMass Laboratories Inc. provides flexible collaboration models tailored to your business goals.
- Co-development partnerships
- Contract research and manufacturing (CRDMO)
- Licensing and technology transfer
- Strategic regulatory support
Decide on the best model for your project infrastructure: Compare CRO vs. In-House ANDA Development
Why Choose ResolveMass Laboratories Inc.?
Our reputation is built on scientific rigor, regulatory expertise, and consistent delivery. Clients trust us for our transparency, technical depth, and ability to navigate complex peptide drug development challenges.
- Proven expertise in peptide-based generics
- Strong regulatory understanding (USFDA, EMA)
- Experienced scientific and leadership team
- Robust quality systems and compliance
- Confidential and client-focused approach
Mitigate risks during the development phase: Read about Nitrosamine Risk Assessment in Generic Drugs
Start Your Semaglutide Project Today
If you are planning or exploring Generic Peptide Semaglutide Projects, ResolveMass Laboratories Inc. offers the scientific expertise, regulatory knowledge, and manufacturing capabilities needed for success. From early-stage development to commercialization, we are your reliable partner in bringing semaglutide generics to market efficiently and compliantly.
👉 Contact Us: https://resolvemass.ca/contact/
Frequently Asked Questions
Semaglutide is structurally complex due to its long peptide chain and sensitivity to environmental conditions. Small variations during synthesis or formulation can impact its performance. This makes its development more demanding compared to conventional small-molecule generics.
Research and development is critical for establishing formulation stability, analytical methods, and process consistency. It ensures the product matches the reference drug in quality and performance. Strong R&D also reduces regulatory risks and improves approval timelines.
ANDA submission requires a complete set of development data including analytical validation, formulation development, and stability studies. It also involves preparing regulatory documentation aligned with authority requirements. Each step must demonstrate equivalence and compliance.
ResolveMass offers scalable manufacturing solutions designed to meet global quality standards. From pilot batches to commercial production, all processes follow strict compliance protocols. This ensures consistent product quality and reliable supply.
Regulatory compliance is maintained through adherence to international guidelines and validated processes. All development activities are documented carefully to support submissions. Continuous quality checks ensure readiness for regulatory review.
The main challenges include maintaining peptide stability, ensuring accurate impurity profiling, and achieving bioequivalence. These require advanced technologies and skilled expertise. Addressing these challenges early improves overall project success.
Reference:
- Kommu, S., & Whitfield, P. (2024). Semaglutide. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK603723/
- Dagli, N., Kumar, S., Ahmad, R., Narwaria, M., & Haque, M. (2023). An update on semaglutide research: A bibliometric analysis and literature review. Cureus, 15(10), e46510. https://pmc.ncbi.nlm.nih.gov/articles/PMC12159091/
