
Introduction:
GLP-1 Peptide Analytical Characterization plays a crucial role in ensuring the safety, efficacy, and regulatory compliance of peptide-based therapeutics. With the rapid growth of GLP-1 receptor agonists in treating diabetes and obesity, pharmaceutical companies are increasingly facing analytical challenges that demand high expertise and advanced instrumentation.
Leveraging specialized services like peptide analytical characterization services and advanced mass spectrometry experts in drug development is becoming essential.
In this blog, we explore the key analytical challenges in GLP-1 peptide drugs and how CDMOs (Contract Development and Manufacturing Organizations) effectively address them using state-of-the-art analytical strategies.
Share via:
Summary:
- GLP-1 peptide drugs face major analytical challenges due to their structural complexity and instability.
- GLP-1 Peptide Analytical Characterization is critical to ensure purity, potency, and safety.
- Common issues include degradation, aggregation, impurity profiling, and bioanalytical variability.
- CDMOs solve these challenges using advanced techniques like LC-MS, peptide mapping, and QbD approaches.
- Strategic outsourcing through peptide CDMO services improves regulatory compliance, scalability, and time-to-market.
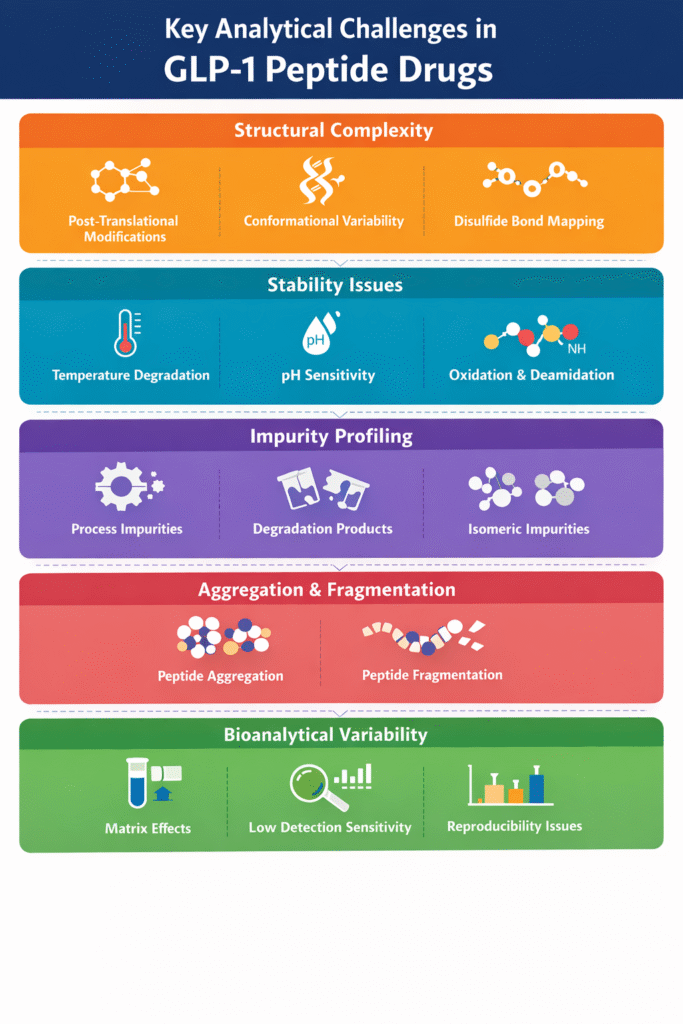
1: What Are the Key Analytical Challenges in GLP-1 Peptide Drugs?
GLP-1 peptides are complex, sensitive molecules that are highly prone to degradation, making accurate analysis difficult and critical for drug quality. Advanced support such as peptide characterization in drug development is often required.
1. Structural Complexity
GLP-1 peptides possess intricate amino acid sequences and higher-order structures that demand advanced analytical techniques.
- Presence of post-translational modifications (PTMs)
- Conformational variability affecting biological activity
- Challenges in disulfide bond mapping and sequence confirmation
2. Stability Issues
GLP-1 peptides are highly sensitive to environmental conditions, leading to degradation.
- Temperature-induced degradation during storage and handling
- pH sensitivity affecting peptide integrity
- Chemical degradation such as oxidation and deamidation
3. Impurity Profiling
Impurities can originate from synthesis, processing, or storage and must be tightly controlled.
- Process-related impurities from peptide synthesis
- Degradation products formed over time
- Isomeric impurities that are difficult to separate and detect
4. Aggregation and Fragmentation
GLP-1 peptides may aggregate or fragment, impacting safety and efficacy.
- Aggregation reduces solubility and bioavailability
- Fragmentation leads to loss or alteration of biological activity
5. Bioanalytical Variability
Accurate quantification in biological systems remains a major challenge.
- Matrix effects interfering with detection
- Low sensitivity at therapeutic concentrations
- Reproducibility issues across assays and laboratories
2: Why Is GLP-1 Peptide Analytical Characterization Important?
GLP-1 Peptide Analytical Characterization ensures safety, efficacy, and regulatory approval. Strong bioanalytical strategy in drug development is critical here. It provides a comprehensive understanding of the peptide’s structure, purity, and performance throughout its lifecycle.
Key Objectives
- Confirm peptide identity and structure
Ensures the correct amino acid sequence and structural integrity of the GLP-1 peptide. - Detect and quantify impurities
Identifies process-related impurities and degradation products that may impact safety. - Ensure batch-to-batch consistency
Maintains uniform quality across different manufacturing batches. - Support regulatory submissions via regulatory support for generic drugs
Provides validated analytical data required for global regulatory approvals.
Regulatory Importance Table
| Parameter | Importance |
|---|---|
| Identity Testing | Confirms correct peptide sequence |
| Purity Analysis | Ensures absence of harmful impurities |
| Stability Studies | Determines shelf life and storage conditions |
| Potency Testing | Confirms biological activity and therapeutic effect |
3: How CDMOs Solve GLP-1 Peptide Analytical Challenges
CDMOs solve GLP-1 peptide analytical challenges by combining advanced technologies, scientific expertise, and regulatory experience to ensure accurate, reliable, and compliant results.
CDMOs solve these challenges using expertise and infrastructure such as peptide drug development CDMO and pharmaceutical CDMO US and Canada.
1. Advanced LC-MS Techniques
CDMOs utilize high-resolution LC-MS systems for precise and sensitive peptide characterization.
- Accurate mass determination for peptide confirmation
- Identification and quantification of impurities
- Detailed peptide sequencing for structural validation
2. Peptide Mapping
Peptide mapping is a critical tool for confirming structure and detecting modifications.
- Enzymatic digestion to break peptides into smaller fragments
- Sequence verification for identity confirmation
- Detection of post-translational modifications (PTMs)
3. Quality by Design (QbD) Approach
A systematic and science-based approach to method development ensures robust analytical performance.
- Design of Experiments (DoE) for method optimization
- Improved method robustness and reproducibility
- Risk-based analysis to identify critical parameters
4. Stability-Indicating Methods
CDMOs develop specialized methods to monitor peptide stability and degradation.
- Forced degradation studies under stress conditions
- Real-time and accelerated stability testing
- Evaluation of degradation pathways
5. Bioanalytical Expertise
Advanced bioanalytical capabilities enhance accuracy in complex biological matrices.
- LC-MS/MS for sensitive and selective quantification
- Immunoassays for targeted analysis
- Pharmacokinetic studies for drug behavior assessment
Key Solutions
- Advanced LC-MS → supported by mass spectrometry experts
- Peptide Mapping → via peptide characterization services
- QbD Approach → linked with analytical method development
- Stability Studies → supported by bioanalysis in Canada
- Bioanalytical Testing → enabled by peptide testing service

4: Role of GLP-1 Peptide Analytical Characterization in Drug Development
GLP-1 Peptide Analytical Characterization supports every stage from discovery to commercialization, often alongside outsourced chemistry in drug discovery.
Development Stages and Analytical Role
| Stage | Role of Analytical Characterization |
|---|---|
| Discovery | Structural confirmation of peptide sequence and identity |
| Preclinical | Stability studies and impurity profiling |
| Clinical | Bioanalytical quantification and pharmacokinetic evaluation |
| Commercial | Quality control, batch release, and regulatory compliance |
Key Takeaway
At every stage, GLP-1 Peptide Analytical Characterization ensures that the drug remains safe, effective, and compliant with regulatory standards, ultimately accelerating successful product development and market approval.
5: Key Techniques Used in GLP-1 Peptide Analytical Characterization
GLP-1 Peptide Analytical Characterization relies on multiple complementary analytical techniques to ensure accurate identification, purity, and structural integrity of peptide drugs.
Advanced tools like peptide sameness study services and case studies such as generic peptide analytical characterization highlight these techniques.
Common Techniques
- LC-MS/MS (Liquid Chromatography–Mass Spectrometry)
Used for precise identification and quantification of peptides, including impurity profiling and sequence confirmation. - HPLC (High-Performance Liquid Chromatography)
Essential for purity analysis, separation of components, and detection of degradation products. - Circular Dichroism (CD)
Helps determine the secondary structure of peptides, such as alpha-helix and beta-sheet conformations. - NMR Spectroscopy (Nuclear Magnetic Resonance)
Provides detailed structural elucidation and insight into molecular conformation. - Capillary Electrophoresis (CE)
Used to analyze charge variants and assess peptide heterogeneity.
Key Takeaway
A combination of these advanced techniques ensures comprehensive GLP-1 Peptide Analytical Characterization, enabling accurate assessment of peptide quality, stability, and functionality.
6: Benefits of Partnering with a CDMO for GLP-1 Peptide Analytical Characterization
Partnering with a CDMO for GLP-1 Peptide Analytical Characterization improves efficiency, ensures regulatory compliance, and delivers high-quality analytical results.
Outsourcing to a best peptide CDMO or peptide CDMO in United States offers major advantages
Key Benefits
- Access to Advanced Instrumentation
CDMOs are equipped with state-of-the-art technologies like LC-MS/MS, HPLC, and advanced spectroscopy tools, ensuring precise and reliable analysis. - Regulatory Expertise
Experienced teams understand global regulatory requirements (FDA, EMA), helping streamline approvals and ensure compliance. - Faster Turnaround Time
Established workflows and dedicated analytical teams accelerate method development and testing timelines. - Cost-Effective Solutions
Outsourcing eliminates the need for expensive in-house infrastructure and reduces operational costs. - Scalability via outsourcing peptide manufacturing
CDMOs offer flexible services that can scale from early-stage research to commercial manufacturing needs.
Key Takeaway
Collaborating with a CDMO enables pharmaceutical companies to achieve robust and reliable GLP-1 Peptide Analytical Characterization while focusing on core drug development activities.
7: How ResolveMass Laboratories Excels in GLP-1 Peptide Analytical Characterization
ResolveMass Laboratories delivers industry-leading expertise in GLP-1 Peptide Analytical Characterization through advanced technologies, scientific excellence, and regulatory-focused solutions.
ResolveMass stands out among generic pharmaceutical CDMO Canada and CDMO for generic drug development in Canada.
Core Strengths
- Specialized LC-MS Platforms
Advanced high-resolution LC-MS systems for precise peptide identification, sequencing, and impurity analysis. - Expertise in Peptide Impurity Profiling
Deep understanding of process-related impurities, degradation pathways, and isomeric variants. - Customized Analytical Method Development
Tailored analytical strategies designed to meet specific project and regulatory requirements. - Regulatory-Compliant Workflows
Fully aligned with global guidelines (FDA, EMA) ensuring smooth submission and approval processes. - Proven peptide experience including ganirelix peptide characterization
Extensive experience supporting GLP-1 and other peptide-based drug development programs.
Why Choose ResolveMass?
- Deep Scientific Expertise
Highly skilled scientists with specialized knowledge in peptide analysis and characterization. - Strong Regulatory Alignment
Ensures all analytical data meets stringent global compliance standards. - High-Quality Data Integrity
Robust systems and validated methods to ensure accurate, reproducible results. - End-to-End Analytical Support
Comprehensive services from early-stage development to commercial batch release.
Key Takeaway
ResolveMass Laboratories stands out as a trusted partner for GLP-1 Peptide Analytical Characterization, offering precision, reliability, and regulatory excellence to accelerate successful peptide drug development.
8: Future Trends in GLP-1 Peptide Analytical Characterization
GLP-1 Peptide Analytical Characterization is rapidly evolving with emerging technologies that enhance accuracy, speed, and data insights in peptide analysis.
The future is shaped by innovation and outsourcing models like CDMO vs CRO for generic drug development and CDMO acceleration strategies.
Trends to Watch
- AI-Driven Analytical Data Interpretation
Artificial intelligence and machine learning are improving data analysis by enabling faster interpretation of complex LC-MS datasets and predicting impurity profiles. - Automation in Peptide Mapping
Automated workflows reduce manual errors, improve reproducibility, and accelerate peptide sequencing and PTM identification. - High-Throughput Screening Methods
Advanced platforms allow simultaneous analysis of multiple samples, significantly increasing efficiency in drug development pipelines. - Advanced Bioanalytical Platforms
Next-generation LC-MS/MS systems and hybrid technologies provide enhanced sensitivity and precision for analyzing peptides in complex biological matrices.
Conclusion:
GLP-1 Peptide Analytical Characterization is essential for overcoming the inherent challenges of peptide drug development. From structural complexity to stability and impurity profiling, these challenges require advanced analytical solutions.
CDMOs like ResolveMass Laboratories play a critical role by offering expertise, cutting-edge technologies, and regulatory support to ensure successful drug development. By leveraging specialized analytical strategies, companies can accelerate time-to-market while maintaining the highest quality standards.
By partnering with experts in outsourcing generic drug development Canada and analytical development for generic drugs Canada, companies can accelerate development timelines while ensuring regulatory compliance and product quality.
Frequently Asked Questions:
GLP-1 Peptide Analytical Characterization is a comprehensive analytical process used to evaluate peptide-based drugs. It involves confirming the peptide’s identity, structure, purity, and stability using advanced techniques like LC-MS and HPLC. This process helps detect impurities, degradation products, and structural variations. It ensures that the drug meets quality standards throughout development. Ultimately, it plays a key role in ensuring safety and therapeutic effectiveness.
GLP-1 Peptide Analytical Characterization is essential to ensure that peptide drugs are safe, effective, and consistent. It helps identify impurities and degradation products that may impact patient safety. The process ensures batch-to-batch consistency during manufacturing. It also provides critical data required for regulatory approvals from agencies like FDA and EMA. Without proper characterization, drug quality and performance cannot be guaranteed.
GLP-1 peptide analysis faces several challenges due to the complex nature of peptides. These include structural complexity, instability under environmental conditions, and difficulty in impurity profiling. Peptides are prone to aggregation and fragmentation, which can affect efficacy. Additionally, bioanalytical variability in biological samples complicates accurate quantification. These challenges require advanced analytical techniques and expertise to overcome.
Multiple analytical techniques are used to fully characterize GLP-1 peptides. LC-MS/MS is widely used for identification, sequencing, and impurity analysis. HPLC helps in purity assessment and separation of components. NMR spectroscopy provides detailed structural information. Circular dichroism is used to study secondary structure, while capillary electrophoresis analyzes charge variants. Together, these techniques ensure comprehensive peptide characterization.
CDMOs provide specialized expertise and advanced infrastructure for peptide analysis. They use high-end instruments like LC-MS/MS and implement robust analytical methods. CDMOs also follow regulatory-compliant workflows, ensuring smooth approvals. Their experience helps in handling complex challenges like impurity profiling and stability studies. Additionally, outsourcing to CDMOs improves efficiency, reduces costs, and accelerates drug development timelines.
The future of GLP-1 Peptide Analytical Characterization is driven by technological advancements. AI and machine learning are being used for faster and more accurate data interpretation. Automation is improving reproducibility and reducing human errors in peptide mapping. High-throughput screening enables faster analysis of multiple samples. Advanced bioanalytical platforms are enhancing sensitivity and precision. These innovations are making peptide analysis more efficient and scalable.
Reference
- Yaseen Z, Nandave M, Sharma L. Anti-diabetic biologicals: Exploring the role of different analytical techniques. Critical Reviews in Analytical Chemistry. 2025 Mar 6:1-22.https://www.tandfonline.com/doi/abs/10.1080/10408347.2025.2472793
- Flier JS. Drug development failure: how GLP-1 development was abandoned in 1990. Perspectives in Biology and Medicine. 2024;67(3):325-36.https://muse.jhu.edu/pub/1/article/936213/summary
- Vinukonda A, Rapolu K, Jadi RK, Devadasu VR. Complex peptide injectables: development and challenges. International Journal of Peptide Research and Therapeutics. 2025 Mar 13;31(3):51.https://link.springer.com/article/10.1007/s10989-025-10710-5
- Hach M, Engelund DK, Mysling S, Mogensen JE, Schelde O, Haselmann KF, Lamberth K, Vilhelmsen TK, Malmstrøm J, Højlys-Larsen KB, Rasmussen TS. Impact of manufacturing process and compounding on properties and quality of follow-On GLP-1 polypeptide drugs. Pharmaceutical Research. 2024 Oct;41(10):1991-2014.https://link.springer.com/article/10.1007/s11095-024-03771-6
- Pereira AJ, de Campos LJ, Xing H, Conda-Sheridan M. Peptide-based therapeutics: challenges and solutions. Medicinal Chemistry Research. 2024 Aug;33(8):1275-80.https://link.springer.com/article/10.1007/s00044-024-03269-1


