
Introduction:
GLP-1 Peptide Impurity Sequencing Analysis plays a critical role in ensuring the safety, efficacy, and regulatory compliance of peptide-based therapeutics. GLP-1 analogs, widely used in metabolic disorders such as diabetes and obesity, are structurally complex and prone to sequence variants and impurities during synthesis and storage.
Advanced approaches such as peptide sequencing of GLP-1 peptide and LC-MS characterization of GLP-1 peptides are essential to accurately detect and characterize these impurities.
Even minor sequence deviations can significantly alter biological activity or immunogenicity. Therefore, accurate identification of impurities is not just a quality requirement—it is a regulatory necessity. This case study highlights how advanced analytical strategies can successfully identify sequence variants and impurities in GLP-1 peptides.
Share via:
Summary:
- GLP-1 peptide drugs are highly sensitive to sequence variations and impurities that can impact safety and efficacy.
- GLP-1 Peptide Impurity Sequencing Analysis is essential for detecting even low-level variants.
- Advanced techniques like LC-MS/MS and High-Resolution Mass Spectrometry (HRMS) enable accurate impurity identification.
- A structured workflow combining sample preparation, fragmentation analysis, and expert validation ensures reliable results.
- This case study demonstrates how ResolveMass Laboratories Inc. identifies and characterizes unknown impurities with high confidence.
1: What Are Sequence Variants and Impurities in GLP-1 Peptides?
Sequence variants are unintended changes in the amino acid sequence of a GLP-1 peptide, while impurities refer to any undesired chemical or structural components present alongside the target peptide. These can arise during synthesis, purification, storage, or handling and must be precisely identified using GLP-1 Peptide Impurity Sequencing Analysis.
For deeper understanding, refer to GLP-1 peptide impurity characterization and analytical characterization of GLP-1 peptide drugs.
Common Types of Impurities
- Truncated peptides (N- or C-terminal deletions)
Result from incomplete peptide synthesis or degradation, leading to shorter peptide chains with reduced biological activity. - Amino acid substitutions
Occur due to coupling errors during solid-phase synthesis, where one amino acid is incorrectly incorporated. - Oxidation and deamidation products
- Oxidation typically affects methionine or tryptophan residues
- Deamidation converts asparagine (Asn) to aspartic acid (Asp), altering charge and structure
- Isomerization (Asp → isoAsp)
Structural rearrangement that does not change mass significantly but affects peptide conformation and function. - Synthesis-related byproducts
Include protecting group remnants, incomplete cleavage products, or side reactions during peptide assembly.
Why They Matter
- Reduced therapeutic potency
Structural changes can directly affect receptor binding and biological activity of GLP-1 analogs. - Potential immunogenicity
Even minor sequence variations can trigger unwanted immune responses in patients. - Stability and shelf-life concerns (see GLP-1 peptide stability analytical methods)
Impurities can accelerate degradation pathways, impacting product consistency over time. - Regulatory compliance risks
Health authorities (FDA, EMA, etc.) require detailed identification and quantification of impurities. Failure to characterize them can delay or block drug approval.
2: Why Is GLP-1 Peptide Impurity Sequencing Analysis Challenging?
GLP-1 impurity analysis is challenging due to structural complexity, low-level impurities, and similar mass profiles of variants.
For detailed insights, see GLP-1 peptide sequencing challenges and GLP-1 peptide sequencing analytical technique.
Key Challenges
| Challenge | Description |
|---|---|
| Structural Similarity | Variants often differ by a single amino acid |
| Low Abundance | Impurities may exist at trace levels (<1%) |
| Complex Fragmentation | Modified peptides produce unpredictable spectra |
| PTMs | Post-translational modifications complicate interpretation |
These challenges demand high-resolution instrumentation and expert data interpretation.
3: Case Study Overview
This case study demonstrates how a GLP-1 peptide sample containing unknown impurities was systematically analyzed and structurally characterized using advanced LC-MS/MS and high-resolution mass spectrometry as part of GLP-1 Peptide Impurity Sequencing Analysis.
This study used advanced methods including peptide sequencing of GLP-1 drugs and GLP-1 analog peptide sequencing workflow to identify unknown impurities.
Objective
The primary goal of this study was to generate accurate, regulatory-compliant impurity data by addressing the following:
- Identify unknown impurities detected in a GLP-1 peptide batch during quality control
- Determine exact sequence variations using de novo sequencing and fragmentation analysis
- Deliver regulatory-ready analytical data with high confidence and reproducibility
Sample Details
- Sample Type: Synthetic GLP-1 analog (therapeutic peptide)
- Purity Concern: Unexpected impurity peaks observed during routine QC (HPLC analysis)
- Impurity Level: Trace-level components (~0.2%–0.8%)
- Initial Limitation: No matching reference standards or database entries available
- Analytical Challenge: Unknown structural identity with closely related mass profiles
Key Analytical Problem
The presence of low-abundance, structurally similar impurities made conventional identification methods insufficient. Therefore, GLP-1 Peptide Impurity Sequencing Analysis using high-resolution MS/MS and de novo sequencing was required to:
- Differentiate between near-isobaric variants
- Localize sequence modifications
- Accurately reconstruct unknown peptide structures
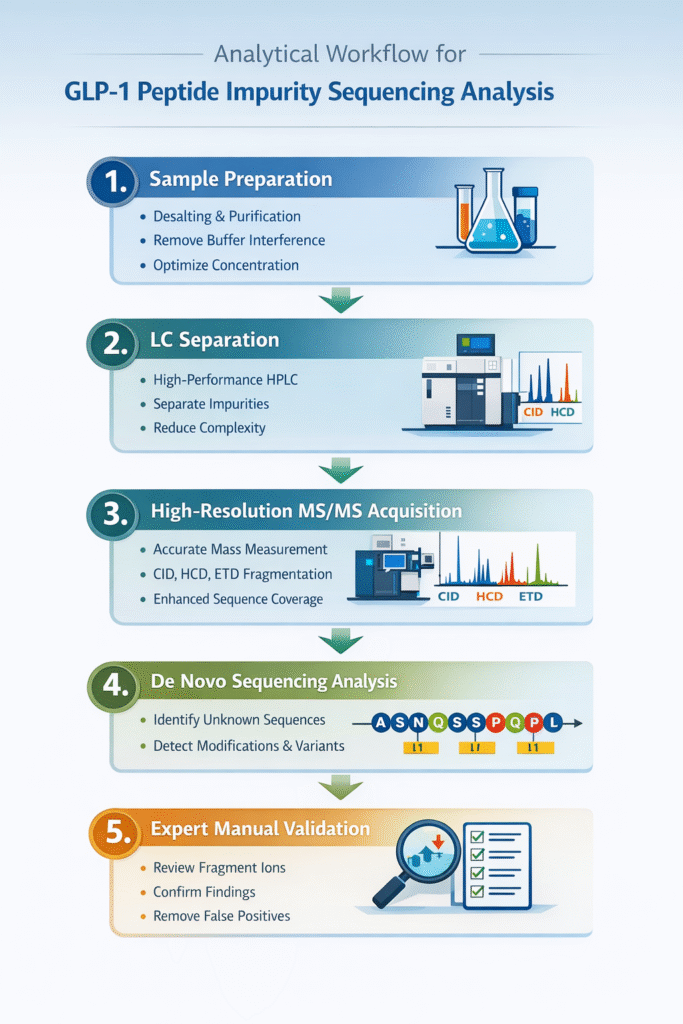
4: Analytical Workflow for GLP-1 Peptide Impurity Sequencing Analysis
A structured, multi-step analytical workflow is essential for accurately identifying sequence variants and low-level impurities in GLP-1 peptides. This approach integrates high-resolution instrumentation with expert data interpretation to ensure reliable, regulatory-grade results.
A structured workflow supported by GLP-1 peptide sequencing CRO services ensures accurate results.
Step 1: Sample Preparation
Proper sample preparation ensures clean, interference-free data and improves detection sensitivity.
- Desalting to remove salts and buffer components that suppress ionization
- Peptide purification to eliminate unrelated contaminants
- Concentration optimization to ensure detectable signal intensity, especially for trace impurities
Outcome: Improved signal quality and reduced background noise for mass spectrometry analysis.
Step 2: LC Separation
High-performance liquid chromatography (HPLC) separates closely related peptide species before MS analysis.
- Resolves co-eluting impurities with similar physicochemical properties
- Enhances detection of low-abundance variants
- Reduces spectral overlap and complexity
Outcome: Individual impurity peaks are isolated, enabling more accurate downstream identification.
Step 3: High-Resolution MS/MS Acquisition
This is the core analytical step where precise mass and structural information is generated.
- Accurate mass measurement (typically <5 ppm error)
- Use of multiple fragmentation techniques:
- CID (Collision-Induced Dissociation) – backbone fragmentation
- HCD (Higher-energy Collisional Dissociation) – high-resolution fragment ions
- ETD (Electron Transfer Dissociation) – preserves labile modifications
- Provides comprehensive fragment ion coverage
Outcome: High-confidence data for identifying subtle sequence differences and modifications.
Step 4: De Novo Sequencing Analysis
De novo sequencing reconstructs peptide sequences directly from MS/MS data without relying on databases.
Supported by high-accuracy methods like de novo GLP-1 peptide sequencing accuracy
- Enables identification of unknown or novel impurities
- Detects amino acid substitutions, truncations, and PTMs
- Essential when reference standards are unavailable
Outcome: Accurate prediction of impurity structures at the amino acid level.
Step 5: Expert Manual Validation
Manual review is critical to ensure accuracy beyond automated software predictions.
- Cross-verification of fragment ion series (b/y ions)
- Confirmation of modification sites and sequence assignments
- Elimination of false positives and ambiguous results
Outcome: Regulatory-grade confidence with validated, reproducible results.
Key Takeaway
GLP-1 Peptide Impurity Sequencing Analysis requires not just advanced instrumentation, but a carefully integrated workflow combining separation, high-resolution detection, computational analysis, and expert validation. This ensures precise identification of even trace-level impurities critical for pharmaceutical quality and compliance.
5: Key Findings from the Case Study
The GLP-1 Peptide Impurity Sequencing Analysis successfully identified and structurally characterized multiple low-level impurities and sequence variants with high analytical confidence. Even trace-level impurities (<0.5%) were accurately detected and validated.
Identified Impurities
The analysis revealed the following specific and structurally confirmed impurities:
- Single Amino Acid Substitution (Ala → Gly)
- Caused by synthesis-related misincorporation
- Detected through precise mass difference and fragment ion shifts
- Challenging due to minimal structural variation
- Oxidized Methionine Variant
- +16 Da mass shift due to oxidation
- Confirmed via MS/MS fragmentation pattern
- Common during storage and handling
- N-terminal Truncated Peptide
- Loss of one or more amino acids from the N-terminus
- Indicates incomplete synthesis or degradation
- Clearly identified through missing b-ion series
- Deamidated Asparagine Residue (Asn → Asp)
- +0.984 Da mass shift
- Alters peptide charge and structure
- Detected using high-resolution mass accuracy and fragmentation mapping
This level of insight is critical for analytical characterization of GLP-1 peptide drugs.
Analytical Confidence
The reliability of the results was ensured through a combination of high-resolution instrumentation and expert validation:
| Parameter | Outcome |
|---|---|
| Sequence Coverage | >95% |
| Mass Accuracy | < 5 ppm |
| Impurity Detection Limit | < 0.5% level |
| Validation Approach | Manual + software confirmation |
Key Insight
This case study confirms that GLP-1 Peptide Impurity Sequencing Analysis can accurately resolve structurally similar, low-abundance impurities using advanced LC-MS/MS workflows combined with expert interpretation. This level of precision is essential for ensuring product quality, safety, and regulatory compliance in peptide therapeutics.
6: How Advanced Mass Spectrometry Enabled Accurate Results
High-resolution mass spectrometry (HRMS) and LC-MS/MS were critical in resolving closely related impurities and confirming sequence variations.
Advantages of HRMS
Techniques like LC-MS characterization of GLP-1 peptides enable:
- High mass accuracy for precise identification
- Ability to distinguish near-isobaric species
- Improved signal-to-noise ratio
Role of Fragmentation Techniques
- CID/HCD: Backbone fragmentation
- ETD: Preserves labile modifications
- Combined approach improves sequence confidence
7: Best Practices for GLP-1 Peptide Impurity Sequencing Analysis
Combining optimized workflows with expert interpretation significantly improves accuracy and reliability.
Best practices align with services offered by a specialized CRO for GLP-1 peptide characterization.
Recommended Strategies
- Use high-resolution instruments for accurate mass detection
- Apply multiple fragmentation modes
- Perform replicate analyses for consistency
- Incorporate manual expert review
- Validate findings with orthogonal techniques if needed
8: Regulatory Importance of Impurity Identification
Regulatory agencies require comprehensive impurity profiling in GLP-1 peptides to ensure patient safety, product efficacy, and consistent manufacturing quality. As part of GLP-1 Peptide Impurity Sequencing Analysis, identifying and characterizing impurities is a mandatory requirement for regulatory approval.
Regulatory compliance requires adherence to guidelines outlined in:
- Regulatory requirements for GLP-1 peptide characterization
- GLP-1 peptide characterization regulatory requirements
Failure to comply can delay approvals and increase costs.
Key Regulatory Expectations
- Identification of impurities above threshold levels
Impurities exceeding regulatory thresholds (typically ≥0.1% depending on guidelines) must be detected and reported with high sensitivity and accuracy. - Structural characterization of unknown variants
Unknown impurities must be fully characterized at the molecular level, including amino acid sequence, modification type, and exact location within the peptide. - Documentation of analytical methods
Detailed reporting of analytical workflows (LC-MS/MS conditions, sample prep, data analysis) is required to ensure transparency and reproducibility. - Reproducibility and validation of results
Methods must be validated and capable of producing consistent results across multiple runs, analysts, and instruments.
Why This Matters for GLP-1 Peptides
- GLP-1 analogs are complex and modification-prone, increasing impurity risks
- Even trace-level impurities can affect biological activity or immunogenicity
- Regulatory bodies such as FDA and EMA demand high-confidence impurity data before approval
Consequences of Non-Compliance
- Delayed regulatory approval due to incomplete impurity profiling
- Additional studies required, increasing cost and timelines
- Batch rejection or product recall in severe cases
- Potential risk to patient safety and brand reputation
Key Takeaway
Robust GLP-1 Peptide Impurity Sequencing Analysis is not just a scientific requirement—it is a regulatory necessity. Accurate identification, thorough characterization, and validated analytical workflows are essential to meet global regulatory standards and ensure successful drug development.
9: Why Choose ResolveMass Laboratories Inc.?
ResolveMass Laboratories Inc. provides industry-leading expertise in peptide sequencing and impurity analysis, ensuring accurate and regulatory-compliant results.
Core Strengths
- Advanced LC-MS/MS and HRMS platforms
- Expertise in de novo peptide sequencing
- Proven track record in GLP-1 analysis
- Regulatory-ready reporting
For companies looking to scale efficiently, consider outsource GLP-1 peptide sequencing services.
Conclusion:
GLP-1 Peptide Impurity Sequencing Analysis is essential for ensuring the safety, efficacy, and regulatory compliance of peptide therapeutics. This case study demonstrates how a structured analytical workflow, combined with high-resolution instrumentation and expert validation, can successfully identify even low-level impurities and sequence variants.
As GLP-1-based therapies continue to grow in importance, robust impurity characterization will remain a cornerstone of pharmaceutical development. Partnering with experienced analytical laboratories ensures confidence in data and faster regulatory approval.
Frequently Asked Questions:
GLP-1 Peptide Impurity Sequencing Analysis is an advanced analytical process used to identify and characterize unknown impurities and sequence variants in GLP-1 peptides.
It combines LC-MS/MS, high-resolution mass spectrometry, and de novo sequencing to deliver precise structural information.
Impurity identification is essential because even small changes can affect drug performance. These impurities may reduce potency or alter biological activity. Some variants can trigger immune responses in patients. Regulatory agencies also require detailed impurity profiling. Without proper identification, drug approval can be delayed or rejected.
Common impurities include:
-Truncated peptides (N- or C-terminal deletions)
-Amino acid substitutions
-Oxidation (e.g., methionine oxidation)
-Deamidation (Asn → Asp)
-Isomerization (Asp → isoAsp)
-Synthesis-related byproducts
Impurities are detected using advanced analytical techniques such as LC-MS/MS and High-Resolution Mass Spectrometry (HRMS).
These methods allow precise mass measurement and fragmentation analysis to identify structural differences.
De novo sequencing determines peptide sequences directly from MS/MS data. It does not rely on existing databases or reference standards. This makes it ideal for identifying unknown impurities. It can detect substitutions, truncations, and modifications. It is a critical tool in GLP-1 Peptide Impurity Sequencing Analysis.
Reference
- Staby A, Steensgaard DB, Haselmann KF, Marino JS, Bartholdy C, Videbæk N, Schelde O, Bosch-Traberg H, Spang LT, Asgreen DJ. Influence of Production Process and Scale on Quality of Polypeptide Drugs: a Case Study on GLP-1 Analogs: Staby et al. Pharmaceutical Research. 2020 Jul;37(7):120.https://link.springer.com/article/10.1007/s11095-020-02817-9
- Hach M, Engelund DK, Mysling S, Mogensen JE, Schelde O, Haselmann KF, Lamberth K, Vilhelmsen TK, Malmstrøm J, Højlys-Larsen KB, Rasmussen TS. Impact of manufacturing process and compounding on properties and quality of follow-On GLP-1 polypeptide drugs. Pharmaceutical Research. 2024 Oct;41(10):1991-2014.https://link.springer.com/article/10.1007/s11095-024-03771-6
- Zhang B, Xu W, Yin C, Tang Y. Characterization of low-level D-amino acid isomeric impurities of Semaglutide using liquid chromatography-high resolution tandem mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis. 2023 Feb 5;224:115164.https://www.sciencedirect.com/science/article/pii/S0731708522005854
- De Groot AS, Mattei A, Gabriel B, Calderini J, Roberts BJ, Lelias S, McAllister M, Boyle C, Martin W, Richard G. Immunogenicity of generic peptide impurities: current orthogonal approaches. Pharmaceutical research. 2025 May;42(5):805-18.https://link.springer.com/article/10.1007/s11095-025-03843-1
- Konduru N, Badam SK, Thamira R, Battula SR, Gundla R. Development and Validation of a Robust Reversed-Phase LC Method for Liraglutide (GLP-1 analogue) and Its Impurities in Injectable Formulations Using a Quality by Design Approach. Available at SSRN 5511143.https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5511143


