Introduction
GLP-1 Peptide Stability Analytical Methods are essential for identifying, measuring, and understanding how GLP-1 peptides degrade under different conditions. These methods are not only used for routine testing but also for detailed impurity profiling and structural analysis. This level of insight is critical for both regulatory approval and drug development.
These analytical approaches also help researchers understand how environmental factors like temperature, light, and pH affect peptide stability. With this knowledge, scientists can improve storage conditions and extend product shelf life. As peptide drugs become more advanced, the need for precise and reliable analysis continues to grow.
Access professional support: CRO for GLP-1 Peptide Characterization
Modern GLP-1 analogs such as semaglutide, liraglutide, and exenatide present unique analytical challenges. Their peptide structure makes them sensitive to degradation and prone to multiple breakdown pathways. This complexity requires highly sensitive and specialized analytical techniques.
In addition, these peptides can undergo chemical changes such as oxidation or deamidation, which may impact their biological activity. Detecting these changes at very low levels is important for patient safety. Therefore, advanced analytical tools are necessary for complete characterization.
Explore our full suite of services: Peptide Characterization Service
Share via:
🔍 Summary
- GLP-1 peptide stability analytical methods rely primarily on LC-MS, RP-HPLC, and orthogonal techniques to identify degradation pathways with high sensitivity.
- LC-MS/MS and HRMS are the gold standards for structural elucidation of degradation products and impurity profiling.
- Forced degradation studies (thermal, pH, oxidative) are essential to develop stability-indicating methods.
- Chromatographic separation (RP-HPLC, UPLC) enables quantification of intact peptide vs impurities with regulatory compliance.
- Size-exclusion and aggregation analysis detect higher-order degradation (dimers, oligomers).
- Orthogonal analytical approaches improve reliability, especially for regulatory submissions.
- Method validation (ICH Q2/Q14) ensures robustness, specificity, and reproducibility.
- Emerging tools include high-resolution MS, multi-dimensional LC, and hybrid workflows for deeper degradation profiling.
GLP-1 Peptide Stability Analytical Methods: Core Analytical Platforms
The most effective GLP-1 Peptide Stability Analytical Methods combine chromatographic separation with high-resolution mass spectrometry. This combination allows detailed identification and accurate measurement of degradation products. It also enables the study of multiple degradation pathways within a single workflow.
These platforms are designed to handle complex peptide molecules while maintaining high sensitivity and specificity. By integrating separation and detection, they provide both qualitative and quantitative data. This approach is now widely accepted as a standard in peptide analysis.
1. LC-MS/MS and LC-HRMS for Degradation Product Identification
LC-MS/MS is one of the most important techniques for identifying degradation fragments and modifications in GLP-1 peptides. It provides detailed fragmentation patterns that help determine molecular structures. This makes it highly effective for studying complex degradation mechanisms.
High-resolution mass spectrometry (HRMS) further improves accuracy by enabling precise mass measurements. It can distinguish between closely related impurities that other methods may miss. This level of detail is essential for regulatory compliance and product safety.
Key capabilities:
- Detection of oxidation, deamidation, and peptide bond cleavage
- Identification of very low-level impurities (<0.1%)
- Structural analysis through fragmentation patterns
These techniques also support metabolite identification and pathway mapping during drug development. Their high sensitivity ensures that even minor degradation products are detected. As a result, LC-MS-based methods are central to modern peptide analysis.
Learn more about mass spec applications: LC-MS Characterization of GLP-1 Peptides
2. RP-HPLC and UPLC Stability-Indicating Methods
RP-HPLC is a key technique in GLP-1 Peptide Stability Analytical Methods for separating intact peptides from their degradation products. It is widely used because it is reliable and easy to validate. This makes it suitable for routine laboratory workflows.
UPLC builds on this by offering faster analysis and better resolution. It reduces run times while maintaining high performance. This is especially useful in high-throughput environments such as quality control labs.
Applications:
- Routine stability testing
- Batch release and quality control
- Impurity profiling
Key considerations:
- Proper gradient optimization
- Use of MS-compatible buffers
- Effective separation of similar impurities
These methods support regulatory submissions by providing consistent and validated data. Their reliability makes them essential tools in pharmaceutical analysis.
Discover our testing capabilities: Peptide Sameness Testing Methods
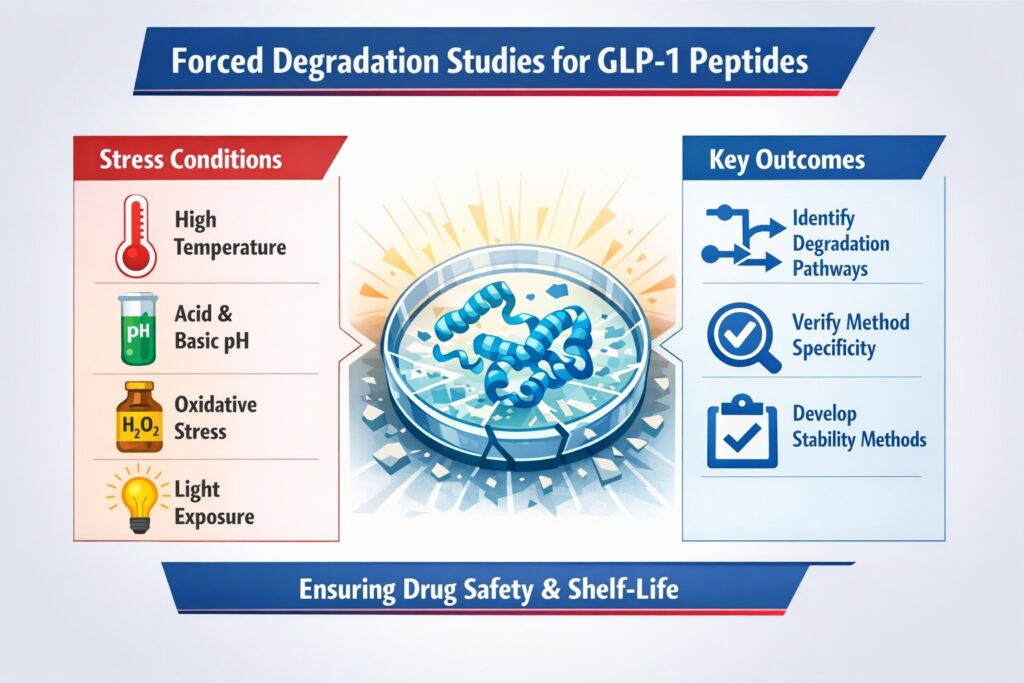
Forced Degradation Studies in GLP-1 Peptide Stability Analytical Methods
Forced degradation studies expose GLP-1 peptides to extreme conditions to generate degradation products. These studies are important for developing and validating analytical methods. They ensure that all possible degradation pathways can be detected.
By simulating harsh environments, researchers can better understand how peptides behave over time. This helps predict shelf life and improve formulation design. It also supports risk assessment during development.
Stress Conditions Commonly Applied:
- High temperature (thermal degradation)
- Acidic and basic pH conditions
- Oxidative stress (e.g., H₂O₂ exposure)
- Light exposure (photolytic degradation)
Outcome:
- Identification of degradation pathways
- Verification of method specificity
- Development of stability-indicating methods
These studies also guide packaging and storage decisions. They provide scientific support for stability claims in regulatory submissions. Overall, they are essential for product quality assurance.
Deep dive into stability testing: Forced Degradation Studies
GLP-1 Peptide Stability Analytical Methods for Aggregation and Higher-Order Degradation
Aggregation is a major degradation pathway in GLP-1 peptides and requires specialized analysis. These aggregates can affect both safety and drug performance. Detecting them is therefore very important.
Higher-order structures like dimers and oligomers may form under stress conditions. These are often not visible using standard techniques. That is why additional analytical methods are required.
Key Techniques:
- Size-Exclusion Chromatography (SEC-LC)
- SEC combined with HRMS
- Dynamic Light Scattering (DLS)
Why it matters:
- Detects dimers, trimers, and oligomers
- Helps assess formulation stability
- Ensures product safety and effectiveness
These methods also help study long-term storage effects. They provide insights into how peptides interact and aggregate. This information supports better formulation design.
Understand structural requirements: Analytical Characterization of GLP-1 Peptide Drugs
Comparative Table: GLP-1 Peptide Stability Analytical Methods
| Method | Purpose | Strengths | Limitations |
|---|---|---|---|
| LC-MS/MS | Degradation identification | High sensitivity, structural info | Complex data analysis |
| LC-HRMS | Accurate mass measurement | High resolution | Expensive instrumentation |
| RP-HPLC | Quantification & separation | Robust, validated | Limited structural insight |
| UPLC | Faster separation | High throughput | Method development complexity |
| SEC-LC | Aggregation analysis | Detects oligomers | Lower resolution for impurities |
Each method plays a specific role in analysis. No single technique can provide complete information. Therefore, combining methods is often necessary.
Orthogonal Analytical Strategies in GLP-1 Peptide Stability Analytical Methods
A single method cannot fully analyze GLP-1 peptide degradation. Orthogonal strategies combine multiple techniques to improve accuracy and reliability. This approach reduces the chance of missing important degradation products.
Typical Workflow:
- RP-HPLC/UPLC → Separation
- LC-MS/MS → Identification
- SEC-LC → Aggregation analysis
- HRMS → Confirmation
This combined approach provides a complete understanding of peptide stability. It also supports regulatory expectations for detailed analytical data.
Optimize your development process: Analytical Method Development and Validation Service
Method Validation and Regulatory Considerations
GLP-1 Peptide Stability Analytical Methods must be validated according to ICH guidelines to ensure consistent performance. Validation confirms that a method is suitable for its intended use. It also ensures reliability across different laboratories.
Key Validation Parameters:
- Specificity
- Linearity
- LOD/LOQ
- Robustness
Validated methods support regulatory approval and ensure consistent product quality. They also reduce variability and improve data integrity over time.
Ensure regulatory compliance: FDA Peptide Sameness Study Requirements
Emerging Trends in GLP-1 Peptide Stability Analytical Methods
New technologies are improving how peptide degradation is studied. These tools offer higher sensitivity and better resolution. They also allow deeper structural analysis.
Innovations:
- Multi-dimensional LC (2D-LC)
- Native mass spectrometry
- AI-based spectral analysis
- Hydrogen-deuterium exchange MS (HDX-MS)
These advancements help detect degradation pathways that were previously difficult to observe. They also improve efficiency and reduce analysis time. As technology evolves, these methods will become more widely used.
Stay updated on impurity analysis: GLP-1 Peptide Impurity Characterization
Conclusion
GLP-1 Peptide Stability Analytical Methods are critical for ensuring the safety, quality, and effectiveness of peptide-based drugs. They provide detailed insights into how peptides degrade and help maintain product integrity throughout their lifecycle.
From LC-MS/MS and RP-HPLC to advanced orthogonal approaches, these tools create a strong analytical framework. They support regulatory compliance and enable accurate impurity analysis. Their combined use ensures reliable and reproducible results.
As GLP-1 therapies continue to grow, the demand for advanced analytical techniques will increase. Future developments will focus on improving sensitivity, speed, and automation. These improvements will further strengthen peptide stability studies.
📞 Contact Us
For advanced GLP-1 peptide stability analytical methods and degradation analysis support:
📌 Frequently Asked Questions (FAQs)
LC-MS/MS and LC-HRMS are among the most sensitive techniques used in GLP-1 Peptide Stability Analytical Methods. They can detect very small amounts of degradation products with high precision. These methods also provide structural details, making them highly useful for advanced analysis and research.
RP-HPLC is widely used for routine quality control because it is reliable and easy to validate. It provides consistent separation and quantification of peptides and impurities. This makes it a preferred choice in many pharmaceutical laboratories.
Aggregation is usually detected using techniques like Size-Exclusion Chromatography (SEC) and Dynamic Light Scattering (DLS). These methods identify larger molecular structures such as dimers or oligomers. Monitoring aggregation is important to ensure product safety and stability.
GLP-1 peptides can degrade through processes such as oxidation, deamidation, and aggregation. These changes may affect the drug’s performance and stability. Identifying these pathways is important for maintaining product quality.
GLP-1 Peptide Stability Analytical Methods are typically validated following ICH Q2(R2) and Q14 guidelines. These ensure that the methods are accurate, reliable, and consistent. Following these standards is necessary for regulatory approval.
GLP-1 peptide analysis can be difficult due to their instability and complex structure. They can degrade in multiple ways, which requires sensitive and advanced methods. Proper method selection is important to overcome these challenges.
Reference:
- Jiang, N., Su, D., Chen, D., Huang, S., Tang, C., Jing, L., Yang, C., Zhou, Z., Yan, Z., & Han, J. (2024). Discovery of a novel glucagon-like peptide-1 (GLP-1) analogue from bullfrog and investigation of its potential for designing GLP-1-based multiagonists. Journal of Medicinal Chemistry, 67(1), 180–198. https://doi.org/10.1021/acs.jmedchem.3c01049
- Müller, T. D., Finan, B., Bloom, S. R., D’Alessio, D., Drucker, D. J., Flatt, P. R., Fritsche, A., Gribble, F., Grill, H. J., Habener, J. F., Holst, J. J., Langhans, W., Meier, J. J., Nauck, M. A., Perez-Tilve, D., Pocai, A., Reimann, F., Sandoval, D. A., Schwartz, T. W., … Tschöp, M. H. (2019). Glucagon-like peptide 1 (GLP-1). Molecular Metabolism, 30, 72–130. https://doi.org/10.1016/j.molmet.2019.09.010
- Manandhar, B., & Ahn, J.-M. (2015). Glucagon-like peptide-1 (GLP-1) analogs: Recent advances, new possibilities, and therapeutic implications. Journal of Medicinal Chemistry, 58(3), 1020–1037. https://doi.org/10.1021/jm500810s
- Drucker, D. J. (2018). Discovery, characterization, and clinical development of the glucagon-like peptides. Molecular Metabolism, 14, 80–99. https://doi.org/10.1016/j.molmet.2018.01.001


