Introduction
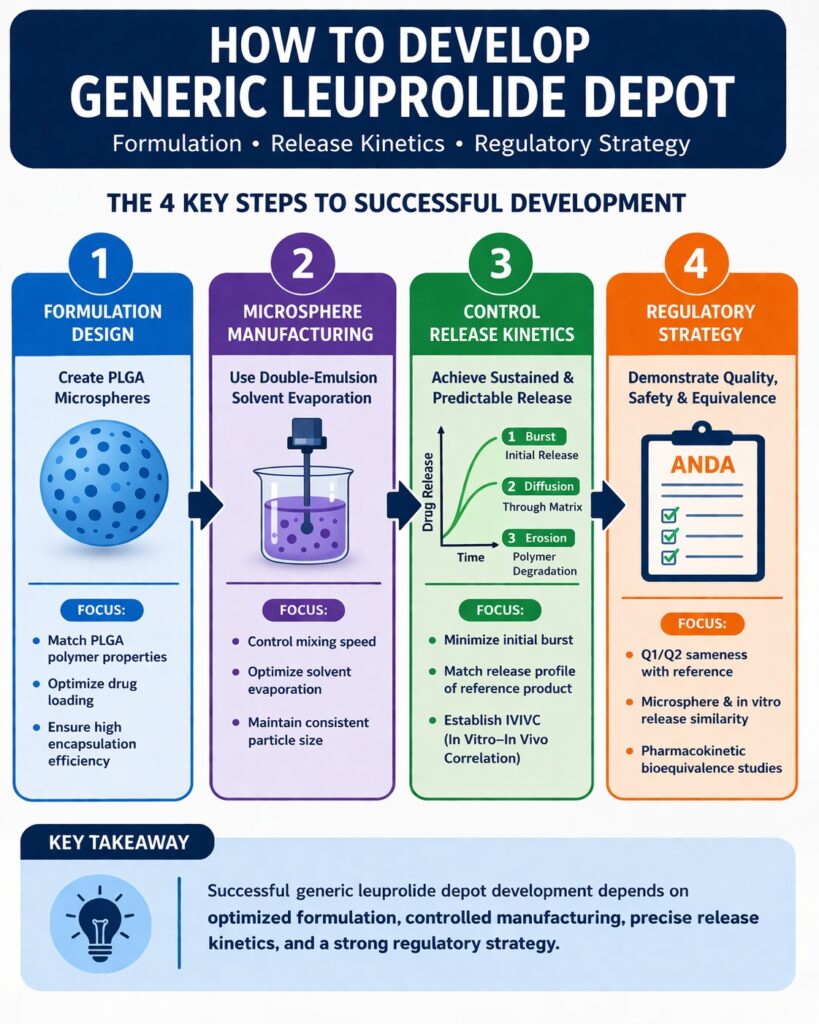
Understanding How to Develop Generic Leuprolide Depot products requires expertise in polymer science, injectable drug formulation, and regulatory strategy. Unlike conventional generics that only need to match the active ingredient, depot injections must also reproduce the controlled drug release behavior of the reference product. This requirement makes development more complex and technically demanding.
Leuprolide depot formulations, such as Lupron Depot®, use biodegradable poly(lactic-co-glycolic acid) (PLGA) microspheres to deliver the drug slowly after intramuscular injection. These microspheres gradually degrade in the body and release the peptide over several weeks or months. Because this delivery mechanism is highly specialized, creating a generic version requires precise control of formulation and manufacturing parameters.
Explore Our Services: Comprehensive Leuprolide Depot Generic Drug Development Services
Another challenge is the sensitivity of peptide drugs like leuprolide. Factors such as temperature, pH, and processing conditions can affect drug stability during production and storage. Therefore, maintaining peptide stability and consistent drug release is critical for successful product development.
This article explains How to Develop Generic Leuprolide Depot products by focusing on three important areas:
- PLGA microsphere formulation design
- Control of drug release kinetics and IVIVC
- Regulatory strategies for complex injectable generics
Understanding these elements helps developers design formulations that closely match the performance of the reference product.
Share via:
Summary (Key Insights)
- Developing a generic leuprolide depot requires precise control of PLGA microsphere formulation, drug loading, and polymer degradation to replicate the reference listed drug (RLD) pharmacokinetics.
- PLGA microsphere engineering determines drug release behavior through polymer composition, molecular weight, and particle size.
- Release kinetics must mimic the RLD profile, including initial burst release and sustained release phases over 1–6 months.
- In vitro–in vivo correlation (IVIVC) is critical for predicting clinical performance and supporting bioequivalence studies.
- Regulatory approval for generic leuprolide depot products involves complex injectable guidance, Q1/Q2 sameness evaluation, and pharmacokinetic bioequivalence.
- Analytical characterization and manufacturing control are essential to ensure consistent microsphere morphology and drug release performance.
- Strategic regulatory planning helps reduce development timelines and mitigate risks during ANDA or global submissions.
How to Develop Generic Leuprolide Depot: Key Formulation Strategies
A critical step in How to Develop Generic Leuprolide Depot formulations is recreating PLGA microspheres that closely resemble those used in the reference product. Developers must match polymer properties, drug loading levels, and microsphere morphology to achieve similar drug release behavior. Proper formulation design ensures that the generic product delivers the drug at a rate comparable to the original formulation.
Leuprolide acetate depot formulations typically use biodegradable PLGA polymer matrices. After intramuscular injection, the polymer slowly degrades through hydrolysis. As the polymer chains break down, the encapsulated peptide drug is gradually released into the bloodstream. This mechanism allows therapeutic drug levels to be maintained for extended periods.
Learn More: Choosing a Specialized Peptide CDMO for Complex Formulations
For generic developers, achieving Q1/Q2 equivalence is an important regulatory requirement. This means the formulation must contain the same ingredients and similar quantities as the reference drug. However, compositional similarity alone is not enough. Developers must also recreate the internal structure of the microspheres to ensure that drug release kinetics remain comparable.
Another important factor in formulation design is encapsulation efficiency. High encapsulation efficiency ensures that most of the peptide drug is embedded within the polymer matrix rather than located on the microsphere surface. Surface-associated drug can cause excessive burst release, which may disturb the intended pharmacokinetic profile.
Formulation scientists also analyze polymer characteristics such as molecular weight, viscosity, and degradation rate. These properties affect how microspheres form during fabrication and how they degrade after injection. Optimizing these variables helps create stable microspheres with consistent performance and reliable drug delivery.
Core Formulation Components
| Component | Role in Depot Formulation |
|---|---|
| Leuprolide acetate | Active peptide drug |
| PLGA polymer | Controlled-release matrix |
| Organic solvent (e.g., DCM) | Polymer dissolution during encapsulation |
| Stabilizers (PVA) | Emulsion stabilization |
| Buffer salts | Maintain peptide stability |
Key Formulation Parameters
Important variables that influence microsphere performance include:
- PLGA composition (lactide:glycolide ratio)
- Polymer molecular weight
- Polymer end-group chemistry (ester vs acid)
- Drug loading percentage
- Microsphere particle size distribution
Research shows that PLGA degradation and erosion mainly control sustained drug release kinetics. Because of this, polymer selection becomes one of the most critical decisions during formulation development. Polymer characteristics directly determine the duration of drug release and systemic exposure following injection.
How to Develop Generic Leuprolide Depot: Microsphere Manufacturing Techniques
Another essential aspect of How to Develop Generic Leuprolide Depot formulations is selecting an appropriate manufacturing method for PLGA microspheres. One of the most widely used techniques is the double-emulsion solvent evaporation method. This process allows efficient encapsulation of peptide drugs while preserving the structural integrity of the microspheres.
The double-emulsion method is particularly suitable for hydrophilic molecules such as peptides. During encapsulation, the polymer forms a protective barrier around the drug, which helps maintain peptide stability and improve drug loading efficiency. The technique also allows researchers to control particle size and drug distribution within the microsphere matrix.
Related Reading: The Complete Generic Drug Development Process for ANDA
Careful control of process conditions is essential to maintain batch-to-batch consistency. Variables such as mixing speed, temperature, and solvent removal rate can influence microsphere structure and drug loading. Even small variations in these conditions can affect the final product quality and drug release behavior.
Scaling up manufacturing from laboratory scale to commercial production also presents challenges. Parameters that work well in small-scale batches may behave differently at larger production volumes. Developers therefore conduct process optimization studies to maintain consistent microsphere structure during scale-up.
Typical Manufacturing Process
- Primary emulsion formation (W/O)
- Leuprolide dissolved in aqueous phase
- Emulsified in polymer organic solution
- Secondary emulsion (W/O/W)
- Primary emulsion dispersed in stabilizer solution
- Solvent evaporation
- Organic solvent removed under agitation
- Microsphere hardening
- Polymer solidifies around encapsulated drug
- Washing and drying
- Removal of residual solvents and stabilizers
Critical Process Parameters
Manufacturing reproducibility is essential for successful generic development. Key process parameters include:
- Homogenization speed
- Solvent evaporation rate
- Polymer concentration
- Temperature control
- Emulsifier concentration
Small changes in these variables can alter particle size and internal drug distribution, which ultimately affects release kinetics and product performance.
How to Develop Generic Leuprolide Depot: Controlling Release Kinetics
A major requirement in How to Develop Generic Leuprolide Depot products is accurately reproducing the multiphase drug release profile of the reference formulation. Both the rate and duration of drug release must match the original product to ensure comparable pharmacokinetics and therapeutic outcomes.
PLGA microspheres generally show three main phases of drug release. Each phase is governed by different physical and chemical processes occurring within the polymer matrix. Understanding these mechanisms helps developers design formulations that closely replicate the reference drug behavior.
Release Phases
| Release Phase | Mechanism |
|---|---|
| Initial burst | Surface drug diffusion |
| Diffusion phase | Drug transport through polymer matrix |
| Erosion phase | Polymer degradation and drug release |
The initial burst release occurs when drug molecules located near the microsphere surface diffuse rapidly into surrounding fluids. A small burst is normal, but excessive burst release may cause higher-than-expected drug levels immediately after injection.
During the diffusion phase, the drug slowly moves through the polymer matrix while the microsphere structure remains relatively intact. Drug molecules pass through small pores inside the microsphere and gradually enter the bloodstream.
The erosion phase begins when PLGA polymer chains start degrading through hydrolysis. As the polymer matrix breaks down, larger channels form inside the microsphere. This accelerates drug release and sustains delivery over extended periods.
Key Factors Affecting Release Kinetics
- Polymer molecular weight
- Microsphere porosity
- Polymer crystallinity
- Particle size
- Drug distribution inside polymer matrix
Many long-acting injectable formulations aim to approach near zero-order release kinetics, where the drug is released at a nearly constant rate over time. Achieving this type of controlled release helps maintain stable therapeutic drug levels in the body.
In Vitro–In Vivo Correlation (IVIVC) in Generic Leuprolide Depot Development
Establishing in vitro–in vivo correlation (IVIVC) is a critical step when learning How to Develop Generic Leuprolide Depot formulations. IVIVC links laboratory drug release results with pharmacokinetic outcomes observed in clinical studies. This relationship allows scientists to predict how a formulation will behave inside the human body.
A Level A IVIVC represents the highest level of correlation. It creates a direct point-to-point relationship between in vitro release data and in vivo drug absorption. When successfully developed, this model becomes a powerful tool during formulation optimization.
IVIVC models also allow developers to evaluate formulation changes without immediately conducting new clinical trials. This significantly reduces development costs and speeds up the optimization process. Regulatory agencies often consider strong IVIVC data supportive evidence for complex injectable products.
Another advantage of IVIVC is improved scientific understanding of formulation behavior. By comparing laboratory release results with clinical pharmacokinetic data, researchers can identify which formulation variables most strongly influence drug release.
Common In Vitro Release Methods
- Dialysis-based release testing
- Sample-and-separate methods
- Continuous flow systems
These testing approaches attempt to mimic physiological conditions as closely as possible. Accurate simulation of biological environments helps ensure that laboratory results reflect real drug release behavior.
Analytical Characterization for Generic Leuprolide Depot Products
Extensive analytical characterization is required to demonstrate similarity between a generic product and the reference leuprolide depot formulation. Analytical studies provide detailed information about microsphere structure, polymer properties, and drug distribution.
Physicochemical characterization confirms that the generic microspheres have similar particle size, morphology, and internal structure as the reference product. These attributes directly influence drug release performance, so they must be thoroughly evaluated during development.
Technical Insight: Analytical Requirements for Successful ANDA Submissions
Drug content analysis is also essential. Analytical testing verifies that the correct amount of leuprolide is encapsulated within the microspheres. Consistent drug loading ensures predictable release kinetics and reliable therapeutic performance.
Another critical component of analytical evaluation is stability testing. Developers must confirm that the formulation maintains its structure, potency, and performance throughout its shelf life. Accelerated stability studies help predict long-term storage behavior.
Key Analytical Methods
Physicochemical Characterization
- Particle size analysis (laser diffraction)
- Surface morphology (SEM)
- Polymer molecular weight (GPC)
- Differential scanning calorimetry (DSC)
Drug Content and Distribution
- HPLC peptide quantification
- Encapsulation efficiency measurement
- Drug distribution mapping
Release Testing
- In vitro drug release profiling
- Stability testing under accelerated conditions
Advanced analytical techniques help demonstrate product similarity and support regulatory documentation required for approval.
Regulatory Strategy for Generic Leuprolide Depot Development
A well-planned regulatory strategy is essential when determining How to Develop Generic Leuprolide Depot products for market approval. Because depot injections are classified as complex generics, regulatory agencies require extensive scientific evidence to demonstrate equivalence.
Developers must show that the generic formulation matches the reference drug in composition, microsphere structure, and drug release performance. This includes demonstrating Q1/Q2 sameness, similar microsphere characteristics, and comparable pharmacokinetic behavior in clinical studies.
Expert Guidance: Nitrosamine Risk Assessment in Generic Drug Development
Early communication with regulatory agencies can help clarify expectations and reduce development risks. Regulatory guidance documents often describe recommended testing strategies for complex injectable drugs, including analytical characterization and bioequivalence assessment.
In addition to scientific data, regulatory submissions must include detailed manufacturing documentation. Process validation, quality specifications, and manufacturing controls are required to confirm consistent product quality.
Regulatory Requirements
Typical expectations include:
- Q1/Q2 sameness evaluation
- Microsphere characterization
- In vitro release similarity
- Pharmacokinetic bioequivalence studies
Bioequivalence Study Design
Clinical studies typically evaluate:
- Plasma leuprolide concentrations
- Suppression of testosterone levels
- Pharmacokinetic parameters such as Cmax and AUC
Because depot formulations release drug slowly over time, bioequivalence studies may extend for several months.
Common Challenges in Generic Leuprolide Depot Development
Developing a generic leuprolide depot involves multiple technical and regulatory challenges. The complexity of PLGA microsphere systems means that many formulation and manufacturing parameters must be carefully controlled.
One of the most difficult challenges is replicating the internal structure of the reference microspheres. Small differences in porosity, polymer distribution, or drug localization can significantly affect drug release kinetics.
Another challenge is managing initial burst release. If too much drug is released immediately after injection, plasma drug concentrations may rise above the expected level compared with the reference product.
Manufacturing scale-up also adds complexity. Maintaining consistent particle size distribution and encapsulation efficiency across large batches requires precise control of processing conditions and strict quality assurance.
Key Challenges
- Replicating microsphere structure and porosity
- Controlling initial burst release
- Establishing IVIVC
- Scaling up manufacturing processes
- Meeting complex regulatory requirements
Strategic Comparison: CRO vs. In-House ANDA Development: Making the Right Choice
Because PLGA polymer variability can strongly influence drug release behavior, careful selection and characterization of polymer materials are essential.
Future Trends in Generic Leuprolide Depot Development
Advances in pharmaceutical technology are improving how scientists approach How to Develop Generic Leuprolide Depot products. New analytical tools and computational models allow researchers to better understand drug release mechanisms from polymer microspheres.
Mechanistic modeling is increasingly used to simulate drug release based on polymer degradation, diffusion behavior, and microsphere structure. These models allow scientists to evaluate formulation strategies before conducting physical experiments.
Artificial intelligence and machine learning are also being explored for formulation optimization. These tools can analyze large experimental datasets and help identify optimal formulation parameters more efficiently.
Partner with Us: Pharmaceutical CDMO Services in the US and Canada
Improved polymer characterization techniques are also providing deeper insights into PLGA degradation behavior. With better understanding of polymer properties, developers can more accurately predict how formulation changes may influence drug release kinetics.
Innovations in Development
- Mechanistic modeling of drug release
- AI-based formulation optimization
- Advanced polymer characterization tools
- Continuous manufacturing techniques
These innovations are helping reduce development risks and accelerate regulatory approval timelines.
Conclusion
Understanding How to Develop Generic Leuprolide Depot formulations requires a combination of advanced formulation science, detailed analytical characterization, and strategic regulatory planning. Because PLGA microsphere systems are complex, developing depot generics is significantly more challenging than conventional small-molecule generics.
Successful development depends on precise control of microsphere composition, polymer characteristics, and manufacturing conditions. These factors collectively determine how the drug is released after injection and how closely the generic formulation matches the reference product.
Accelerate Your Project: How a CDMO Can Accelerate Generic Drug Development
Key development elements include designing robust PLGA microsphere formulations, controlling burst release and sustained release kinetics, establishing predictive IVIVC models, and implementing comprehensive analytical characterization. Each of these components contributes to demonstrating product similarity and ensuring consistent therapeutic performance.
Developers must also create regulatory and bioequivalence strategies that align with requirements for complex injectable drugs. Early planning and strong analytical data packages can significantly increase the likelihood of successful approval.
As regulatory frameworks for complex generics continue to evolve, companies with strong expertise in polymer science, injectable formulation development, and regulatory strategy will be best positioned to develop high-quality generic leuprolide depot products.
If you are exploring generic leuprolide depot development or analytical characterization support, you can connect with experts here:
Frequently Asked Questions (FAQs)
The generic version of LUPRON DEPOT® is leuprolide acetate for depot suspension. It contains the same active ingredient and works in a similar way by suppressing hormone production in the body. Generic versions are designed to provide comparable therapeutic effects while typically being more affordable. However, developing these generics can be complex because they use controlled-release microsphere technology.
Leuprolide depot is a long-acting injectable formulation that slowly releases the drug leuprolide acetate over time. It is commonly used to treat conditions such as prostate cancer, endometriosis, uterine fibroids, and precocious puberty. The medicine works by reducing certain hormone levels in the body. Because it is a depot injection, one dose can work for several weeks or months.
Several medications can be used as alternatives to LUPRON DEPOT, depending on the medical condition being treated. Drugs such as goserelin (Zoladex®), triptorelin, and degarelix are commonly prescribed hormone-suppressing therapies. These medications work through similar hormonal pathways but may differ in dosing schedule and formulation. A healthcare provider usually selects the best option based on the patient’s specific treatment needs.
Lupron Depot can be expensive, but there are ways patients may reduce the cost. Some options include using manufacturer savings programs, patient assistance programs, or insurance coverage plans. Pharmacies and healthcare providers may also help identify discount programs or alternative formulations. In some cases, using approved generic versions may significantly lower treatment costs.
LUPRON DEPOT provides long-acting hormone suppression, which means patients do not need frequent dosing. The controlled-release formulation maintains consistent drug levels in the body over several weeks or months. It is widely used for conditions such as prostate cancer, endometriosis, and uterine fibroids. This long-acting delivery system can improve treatment convenience and patient adherence.
LUPRON DEPOT is manufactured by AbbVie, a global pharmaceutical company. The product is produced in regulated pharmaceutical manufacturing facilities that follow strict quality and safety standards. These facilities comply with international regulatory guidelines to ensure consistent product quality. Distribution then occurs globally through approved healthcare supply chains.
Reference:
- Sethi, R., & Sanfilippo, N. (2009). Six-month depot formulation of leuprorelin acetate in the treatment of prostate cancer. Clinical Interventions in Aging, 4, 259–267. https://doi.org/10.2147/CIA.S4885
- U.S. Food and Drug Administration. (2012). Lupron (leuprolide acetate) injection label (NDA 020011, S-040). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020011s040lbl.pdf



