Introduction:
The development of generic leuprolide depot formulations is highly complex because it requires matching the exact drug release pattern of the reference product. These Leuprolide Depot Analytical Challenges mainly come from the need to control the PLGA polymer matrix and the structure of microspheres. Even very small changes in polymer type or particle design can affect how the drug is released over time. Because of this, companies must follow a well-controlled and repeatable formulation process to achieve consistent results.
To successfully manage these Leuprolide Depot Analytical Challenges, a combination of advanced analytical techniques is required. These include polymer testing, peptide stability studies, and sensitive in vitro release testing. When used together, these tools help ensure bioequivalence, meet regulatory requirements, and maintain product quality. Early use of analytical data also reduces development risks and improves overall formulation performance.
Explore our full suite of services: Learn more about the Generic Drug Development Process for ANDA
Share via:
Summary:
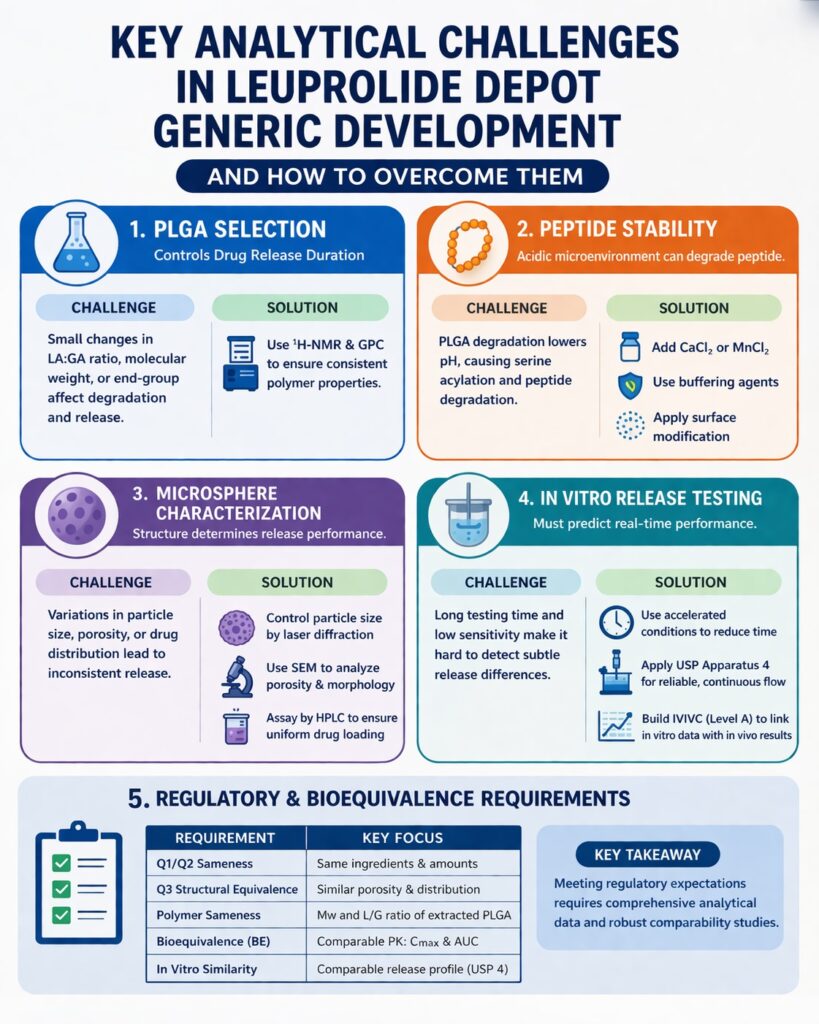
- Complex formulation requirements: Developing generic leuprolide depot products requires precise control of PLGA polymer properties and microsphere structure to match the reference drug’s release profile.
- Critical role of advanced analytics: Techniques like NMR, GPC, HPLC-MS/MS, and IVR testing are essential to ensure bioequivalence, stability, and product quality.
- PLGA selection drives performance: Factors such as lactide:glycolide ratio, molecular weight, and end-group chemistry directly impact degradation rate and drug release behavior.
- Peptide stability is a major risk: Acidic microenvironments within microspheres can lead to degradation pathways like acylation, requiring careful formulation strategies and monitoring.
- Microsphere design influences release: Particle size, porosity, and drug distribution must be tightly controlled to ensure consistent dosing and predictable release profiles.
- Regulatory success depends on total equivalence: Achieving Q1, Q2, Q3 sameness, robust IVIVC, and compliance with FDA guidelines is critical for approval of complex generics.
How to Overcome Leuprolide Depot Analytical Challenges in PLGA Selection
Choosing the right PLGA polymer is one of the most important steps in overcoming Leuprolide Depot Analytical Challenges. This polymer directly controls how the drug is released by breaking down over time through hydrolysis. Since this process depends on polymer properties and environmental conditions, even small differences can lead to unexpected release behavior.
The degradation rate of PLGA depends on factors such as lactide-to-glycolide ratio, molecular weight, and end-group chemistry. Each of these must be carefully selected and tested to ensure consistency. In addition, storage conditions like temperature and moisture can impact polymer stability, making continuous monitoring essential.
Copolymer Composition and Sequence Analysis
The ratio of lactic acid (LA) to glycolic acid (GA) affects important properties like hydrophobicity and glass transition temperature (Tg). These properties influence how water enters the microspheres and how fast the polymer degrades. For example, a 75/25 L/G ratio is commonly used in 1-month depot formulations to achieve steady drug release over 30–35 days.
Proton Nuclear Magnetic Resonance (^1H-NMR) is used to confirm this ratio and verify polymer identity. Advanced NMR methods can also study how monomers are arranged in the polymer chain. This is important because uneven structures may lead to inconsistent degradation and drug release.
Molecular Weight Distribution and Polydispersity Index
The molecular weight (Mw) and polydispersity index (PDI) affect the strength and degradation behavior of PLGA microspheres. A higher molecular weight provides better stability, while a narrow PDI ensures uniform breakdown of the polymer. Variations in these values can lead to inconsistent drug release.
During manufacturing, stress from heat or mixing can change molecular weight. Techniques like Gel Permeation Chromatography (GPC) are used to monitor these changes. Keeping a consistent molecular weight distribution helps avoid issues like sudden drug release or uneven dosing.
Deep Dive into Technical Guidance: How to Develop Generic Leuprolide Depot: A Step-by-Step Guide
End-Group Chemistry and Hydrophilicity
PLGA polymers can have different end groups, such as acid-capped or ester-capped forms. These groups affect how quickly the polymer absorbs water and degrades. Acid-capped PLGA degrades faster, which may be useful for short-term formulations but can increase the risk of peptide instability.
Analytical tools like titration and NMR help identify these end groups and ensure consistency. Understanding how these groups interact with the drug helps improve both stability and release performance.
| Polymer Attribute | Analytical Technique | Impact on Depot Performance |
| LA:GA Molar Ratio | ^1H-NMR | Sets the rate of hydrolytic degradation and Tg. |
| Molecular Weight (Mw, Mn) | GPC/SEC | Controls the timing of bulk erosion and mechanical stability. |
| End-Group Type | NMR / Titration | Influences initial water uptake and peptide stability. |
| Glass Transition (Tg) | DSC | Determines storage stability and “self-healing” temperature. |
| Residual Monomers | GC-MS / HPLC | Affects local pH during early stages of degradation. |
Addressing Leuprolide Depot Analytical Challenges in Peptide Stability
Peptide stability is another major concern in Leuprolide Depot Analytical Challenges. As PLGA degrades, it creates an acidic environment inside the microspheres. This drop in pH can damage the peptide and reduce its effectiveness.
In some cases, the internal pH can fall as low as 2.0. Under these harsh conditions, the peptide may undergo chemical changes such as hydrolysis or oxidation. Continuous monitoring is necessary to ensure the drug remains stable throughout its shelf life.
Expert Insights: Understand Analytical Requirements for ANDA Generic Drugs
The Mechanism and Identification of Serine Acylation
Acylation is a key degradation pathway where the peptide reacts with the polymer. Although leuprolide acetate does not have common reactive sites, studies show that serine can still undergo acylation.
Advanced methods like HPLC-MS/MS are used to detect these changes. These techniques can identify even small modifications that may affect product quality. This highlights the importance of sensitive analytical tools in peptide analysis.
Stability-indicating RP-HPLC methods are also used to separate and measure impurities. Mass spectrometry confirms their structure, helping ensure compliance with regulatory standards.
Mitigation Strategies: Additives and Formulation Controls
To reduce acylation, formulators use strategies that limit interaction between the peptide and polymer. One method involves adding salts like Calcium Chloride (CaCl2) or Manganese Chloride (MnCl2), which compete with the peptide for binding sites.
Other approaches include using buffering agents, modifying polymer surfaces, or temporarily protecting reactive groups. Combining these methods often provides better stability and longer shelf life.
| Stabilization Strategy | Material Examples | Mechanism of Action |
| Divalent Cation Competition | CaCl2, MnCl2 | Blocks peptide-PLGA ionic interactions. |
| pH Neutralization | MgCO3, ZnO | Buffers the internal μpH to minimize acid-catalyzed reactions. |
| Surface Passivation | PEG-PLA Copolymers | Reduces peptide adsorption to the hydrophobic polymer surface. |
| Amine Masking | Self-immolative Groups | Temporarily blocks nucleophilic sites until physiological pH is reached. |
Overcoming Leuprolide Depot Analytical Challenges in Microsphere Characterization
Understanding microsphere structure is essential for controlling drug release and solving Leuprolide Depot Analytical Challenges. Features like porosity and drug distribution directly affect how the drug is released.
The double emulsion (W1/O/W2) method is commonly used to produce microspheres. Process conditions such as temperature and mixing speed must be carefully controlled to ensure consistent results.
Strategic Development: Choosing the Right Partner: Generic Drug Development CRO for ANDA
Particle Size Distribution and Syringeability
Particle size affects both drug release and ease of injection. Larger particles release the drug slowly, while smaller ones may cause a rapid initial release. A balanced size distribution is important for consistent performance.
Laser diffraction is used to measure particle size. A narrow size range ensures uniform dosing and smooth injection, which is important for patient safety and regulatory approval.
Internal Porosity and Surface Morphology
Porosity controls how quickly water enters the microspheres. High porosity leads to faster drug release, while low porosity supports slower release. Controlling pore structure is therefore critical.
Scanning Electron Microscopy (SEM) helps visualize the surface and internal structure. Additional tests measure pore volume and distribution, helping optimize formulation design.
Drug Loading and Distribution Homogeneity
Uniform drug distribution ensures consistent release and dosing. Uneven distribution can cause variability in treatment outcomes, which is not acceptable for regulated products.
HPLC is used to measure drug content after dissolving microspheres. Imaging techniques can further confirm uniform distribution, helping improve formulation quality.
Solving Leuprolide Depot Analytical Challenges in In Vitro Release Testing
In vitro release (IVR) testing is essential for evaluating how the formulation performs. These tests must detect small differences between products and predict real-life behavior, making them a key part of Leuprolide Depot Analytical Challenges.
Since real-time testing takes a long time, accelerated methods are often used. These methods provide faster results while still maintaining accuracy.
Risk Mitigation: The Importance of Nitrosamine Risk Assessment in Generic Drugs
Real-Time vs. Accelerated Release Methods
Accelerated testing involves modifying environmental conditions to speed up polymer degradation. Common approaches include increasing temperature, adjusting pH, and adding solvents. These changes help reduce testing time without compromising data quality.
For leuprolide acetate, specific accelerated conditions can compress a 30-day release into just a few days. This significantly improves development efficiency. However, careful validation is required to ensure these methods accurately reflect real-world behavior.
The Advantage of USP Apparatus 4
USP Apparatus 4 is widely considered the gold standard for IVR testing of long-acting injectables. It provides continuous flow conditions that better mimic physiological environments. This results in more reliable and reproducible data.
The use of glass beads within the system helps maintain uniform flow and prevents aggregation. This modification improves consistency, especially for sensitive formulations. Overall, this method enhances the predictive power of in vitro testing.
Establishing In Vitro-In Vivo Correlation (IVIVC)
A Level A IVIVC represents the highest standard of correlation between in vitro and in vivo data. Achieving this requires advanced mathematical modeling and a deep understanding of drug release mechanisms. It allows developers to predict clinical outcomes based on laboratory data.
Such correlations can significantly reduce the need for clinical studies. This not only lowers development costs but also accelerates time to market. Establishing a robust IVIVC is therefore a major objective in generic drug development.
Regulatory Pathways for Navigating Leuprolide Depot Analytical Challenges
Regulatory approval for generic leuprolide depot formulations requires extensive analytical and clinical evidence. These products are classified as complex generics, meaning that simple equivalence is not sufficient. Developers must demonstrate Q1, Q2, and Q3 equivalence to the reference product.
This includes matching formulation composition, structural characteristics, and performance attributes. Regulatory agencies closely evaluate all aspects of the product, from raw materials to final dosage form. Comprehensive documentation is essential for successful approval.
Compliance Checklist: Essential Requirements for ANDA Submission of Generic Drugs
FDA Product-Specific Guidance and PK Metrics
The FDA provides detailed guidance for different leuprolide depot formulations. These guidelines outline the requirements for demonstrating bioequivalence. Key pharmacokinetic parameters must fall within predefined acceptance ranges.
Parameters such as C{max} and AUC are particularly important. They reflect both initial and sustained drug release. Meeting these criteria ensures that the generic product performs similarly to the reference drug in clinical settings.
Analytical Characterization of the Finished Product
Regulators require detailed characterization of the final product, not just the raw materials. This includes analysis of the extracted PLGA to confirm its properties after processing. Such testing ensures that manufacturing does not alter critical attributes.
Techniques like GPC and ^1H-NMR are used for this purpose. Comparative studies with the reference product are also required. These analyses provide strong evidence of equivalence and support regulatory approval.
| Regulatory Requirement | Documentation Required | Analytical Method |
| Q1/Q2 Sameness | Same ingredients in same amounts | Formulation audit and assay. |
| Q3 Structural Equivalence | Equivalent porosity and distribution | SEM, BET, and Raman. |
| Polymer Sameness | Mw and L/G ratio of extracted PLGA | GPC and ^1H-NMR. |
| Bioequivalence (BE) | Comparative PK study in patients | LC-MS/MS of plasma samples. |
| In Vitro Similarity | Comparable release profiles | USP 4 (Real-time and Accelerated). |
Leveraging Advanced Analytical Expertise at ResolveMass Laboratories Inc.
Overcoming Leuprolide Depot Analytical Challenges demands specialized expertise and advanced instrumentation. ResolveMass Laboratories Inc. offers comprehensive analytical solutions tailored for complex injectable formulations. Their capabilities support every stage of development, from early characterization to final validation.
Accelerate Your Pipeline: How a CDMO can Accelerate Generic Drug Development in the US and Canada
Specialized Polymer and Peptide Characterization
The team provides detailed polymer fingerprinting using advanced techniques such as NMR and high-resolution mass spectrometry. This enables precise identification of polymer composition and structure. Such insights are critical for ensuring product equivalence.
Their expertise also extends to peptide analysis, including impurity profiling and degradation studies. This ensures that even minor modifications are detected and controlled. As a result, product quality and safety are maintained at the highest level.
Specialized Solutions: Explore Leuprolide Depot Generic Drug Development Services
Integrated Method Development and Regulatory Support
ResolveMass offers end-to-end support for analytical method development and validation. Their workflows are aligned with ICH and FDA requirements, ensuring regulatory compliance. This integrated approach streamlines development and reduces delays.
Their experience with IVR methods and IVIVC modeling further enhances their value. By bridging the gap between laboratory data and clinical outcomes, they help accelerate product approval. This makes them a trusted partner in complex generic development.
Conclusion
The development of generic leuprolide depot products requires careful handling of multiple analytical factors. Addressing Leuprolide Depot Analytical Challenges involves detailed work in polymer selection, peptide stability, microsphere design, and release testing.
With the right analytical strategies and expert support, these challenges can be successfully managed. ResolveMass Laboratories Inc. plays a key role by offering advanced testing and reliable data. Their expertise helps ensure that products meet high standards of quality, safety, and performance.
FAQs: Leuprolide Depot Development
Leuprolide acetate does not have the usual reactive amine groups seen in many peptides, which limits common acylation pathways. However, the serine residue contains a reactive hydroxyl group that can interact with the PLGA polymer, especially in acidic conditions inside the microsphere. This interaction leads to chemical modification of the peptide over time. Advanced tools like HPLC-MS/MS confirm this behavior, making serine an important focus area in Leuprolide Depot Analytical Challenges.
Even if two PLGA materials appear similar in composition and molecular weight, small differences can exist based on the manufacturing source. These differences may include polymer chain arrangement, impurities, or residual materials. Such variations can influence how the polymer absorbs water and degrades, which directly affects drug release. For this reason, detailed polymer comparison is essential to overcome Leuprolide Depot Analytical Challenges and ensure consistent product performance.
As PLGA breaks down, it produces acidic byproducts that can collect inside the microsphere and lower the internal pH. This acidic environment can damage the peptide by speeding up degradation reactions such as hydrolysis and acylation. Monitoring this internal pH helps scientists understand stability issues. Managing this factor is critical in addressing Leuprolide Depot Analytical Challenges effectively.
Leuprolide acetate is highly soluble in water, which makes it prone to escaping from the internal phase during processing. Cooling the primary emulsion increases the thickness of the polymer solution, helping to trap the drug within the droplets. This step reduces drug loss and improves encapsulation efficiency. It is an important process control strategy in managing Leuprolide Depot Analytical Challenges.
Microfluidic systems allow better control over particle formation, producing microspheres with uniform size and structure. This leads to more predictable drug release and improved dose consistency. Unlike traditional methods, microfluidics reduces stress on sensitive peptides by avoiding high shear forces. This technology is becoming valuable in solving complex Leuprolide Depot Analytical Challenges.
Residual solvents such as DCM can remain trapped in the microspheres after manufacturing if not properly removed. These solvents may soften the polymer and change its release behavior, especially in the early stages. They also raise safety concerns, so strict limits are set by regulatory guidelines. Proper drying techniques are required to control this aspect of Leuprolide Depot Analytical Challenges.
When microspheres are injected into the body, the immune system reacts to them as foreign materials. This response can change the local environment, including enzyme activity and tissue structure. These factors may influence how the polymer degrades compared to lab conditions. Understanding this helps improve in vitro models and strengthens IVIVC in Leuprolide Depot Analytical Challenges.
Reference:
- Zhou, Y., & Park, K. (2020). Effect of manufacturing variables and raw materials on the composition-equivalent PLGA microspheres. Molecular Pharmaceutics, 17(5), 1502–1515. https://doi.org/10.1021/acs.molpharmaceut.9b01188
- Makadia, H. K., & Siegel, S. J. (2011). Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers, 3(3), 1377–1397. https://doi.org/10.3390/polym3031377
- Zhang, Y., & Schwendeman, S. P. (2012). Minimizing acylation of peptides in PLGA microspheres. Journal of Controlled Release, 162(1), 119–126. https://doi.org/10.1016/j.jconrel.2012.04.022
- Guo, N., Zhang, Q., Sun, Y., & Yang, H. (2019). Separation and identification of acylated leuprorelin inside PLGA microspheres. International Journal of Pharmaceutics, 560, 273–281. https://doi.org/10.1016/j.ijpharm.2019.01.061
- Ding, A. G., & Schwendeman, S. P. (2008). Acidic microclimate pH distribution in PLGA microspheres monitored by confocal laser scanning microscopy. Pharmaceutical Research, 25(9), 2041–2052. https://doi.org/10.1007/s11095-008-9594-3
- Hirota, K., Doty, A. C., Ackermann, R., Zhou, J., Olsen, K. F., Feng, M. R., Wang, Y., Choi, S., Qu, W., Schwendeman, A. S., & Schwendeman, S. P. (2016). Characterizing release mechanisms of leuprolide acetate-loaded PLGA microspheres for IVIVC development I: In vitro evaluation. Journal of Controlled Release, 244(Pt B), 302–313. https://doi.org/10.1016/j.jconrel.2016.08.023



