This Leuprolide Depot Case Study Sustained Release highlights major technical improvements in PLGA microsphere engineering that help reduce the unwanted initial drug burst and maintain steady hormone suppression for 1 to 6 months. By optimizing the lactide:glycolide ratio, controlling primary emulsion cooling, and refining particle Span to ≤ 0.5, consistent near zero-order release can be achieved. These improvements are especially important for therapies used in prostate cancer and central precocious puberty. The approach improves dosing predictability, reduces plasma level fluctuations, and supports better patient adherence. Tighter control of polymer architecture also improves batch-to-batch reproducibility during scale-up.
Learn more about the fundamental stages of creating complex formulations: Explore the Generic Drug Development Process for ANDA
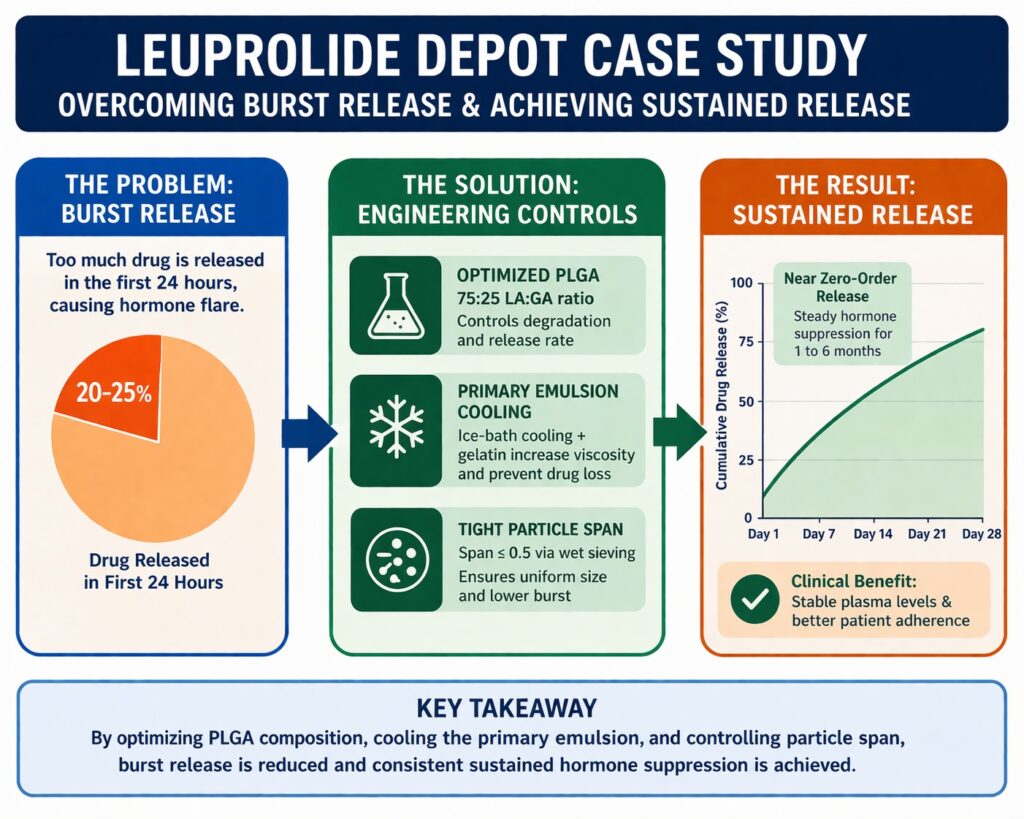
The pharmaceutical landscape for long-acting injectables (LAIs) increasingly requires precise delivery systems capable of maintaining therapeutic levels over extended periods. This Leuprolide Depot Case Study Sustained Release from ResolveMass Laboratories Inc. explains how advanced microencapsulation strategies help stabilize leuprolide acetate, a water-soluble GnRH agonist, inside biodegradable polymer matrices. One major challenge is “burst release,” where a large portion of the drug releases within the first 24 hours after injection. This early spike can cause hormonal flare or reduced levels later in the cycle. To address this, a multi-factor strategy is applied, including polymer end-group selection, viscosity control, and detailed analytical characterization. These measures help maintain microstructural equivalence to reference products such as Lupron Depot while ensuring stable and controlled drug release.
Share via:
Key Takeaways: Leuprolide Depot Case Study Sustained Release
- Burst release (20–25% in 24 hours) is minimized using optimized PLGA composition, acid end-groups, and controlled microsphere porosity.
- Polymer parameters such as LA:GA ratio (75:25), molecular weight, and Tg govern degradation rate and sustained release duration.
- Controlling particle size distribution (Span ≤ 0.5) significantly reduces early drug release and improves dosing predictability.
- Optimized W1/O/W2 double-emulsion process, viscosity enhancement, and ice-bath cooling improve encapsulation efficiency (>70%).
- Dense microsphere structure with low porosity (~11%) supports near zero-order release for 1–6 months.
- Microstructural equivalence, analytical characterization, and QbD strategies enable reproducible scale-up and bioequivalent generic development.
Technical Foundations of Leuprolide Depot Case Study Sustained Release
Sustained release in leuprolide depots is achieved by encapsulating the peptide within a PLGA matrix that regulates drug liberation through diffusion across water-filled pores and gradual hydrolytic erosion of the polymer network. The polymer grade selection is the most critical determinant of release duration, with the lactide:glycolide (LA:GA) ratio and molecular weight serving as primary tuning parameters. These factors directly influence polymer hydration, pore formation, and degradation kinetics. Controlled selection of these attributes allows formulators to tailor release profiles for monthly or multi-month dosing. Careful adjustment of polymer composition also improves peptide stability during storage and after injection.
Partner with experts to navigate the complexities of peptide formulation: Work with a Specialized CRO for Leuprolide Depot Development
Polymer Chemistry and Degradation Kinetics
PLGA is a linear copolymer that degrades through hydrolysis into lactic acid and glycolic acid, which are naturally metabolized in the body. Degradation speed depends on monomer hydrophobicity. Lactic acid slows water penetration, while glycolic acid increases hydrophilicity and accelerates breakdown. For a one-month depot, a 75:25 LA:GA ratio is commonly used to balance stability and erosion. This ratio supports predictable degradation while maintaining structural integrity. Polymer composition also affects microenvironment pH and peptide stability.
The molecular weight and polydispersity index further influence release behavior. High molecular weight polymers create dense microspheres with slower release, while low molecular weight polymers form porous structures with faster release. Glass transition temperature must also be controlled so the polymer remains stable at body temperature. Maintaining a glassy state prevents premature relaxation and limits rapid drug diffusion. These parameters together define release consistency and stability.
| Polymer Attribute | Specification (1-Month LD) | Analytical Technique | Impact on Release |
|---|---|---|---|
| LA:GA Ratio | 75:25 (74.3:25.7 observed) | 1H NMR Spectroscopy | Controls degradation rate |
| Weight-Average Mw | ~13.0 kDa | GPC | Determines matrix strength |
| Number-Average Mn | ~8.7 kDa | GPC | Influences polymer chain mobility |
| Polydispersity (PDI) | ~1.5 | GPC | Affects batch consistency |
| Acid Number | ~12.9 mg KOH/g | Titration | Indicates carboxylic acid end-groups |
| Glass Transition (Tg) | ~48.6°C | DSC | Ensures stability at 37°C |
The Critical Role of End-Group Chemistry in Leuprolide Depot Case Study Sustained Release
The interaction between the positively charged leuprolide peptide and PLGA polymer is enhanced by carboxylic acid end-groups in acid-capped polymers. Ester-capped polymers are more hydrophobic and provide fewer binding sites. Acid-capped PLGA allows ion exchange between leuprolide acetate and polymer carboxyl groups. This interaction anchors the peptide within the matrix during hydration. As a result, early drug diffusion is minimized and sustained release is maintained. End-group chemistry therefore plays an important role in reducing premature leakage.
Deep dive into the specific hurdles of analyzing long-acting injectables: Understand Leuprolide Depot Analytical Challenges
Mechanisms of Initial Burst Release in Leuprolide Depot Case Study Sustained Release
Burst release refers to the rapid liberation of drug, often 20–25% of the total dose, within the first 24 hours after exposure to aqueous media. This effect is driven by surface-associated drug and interconnected pore networks. In this Leuprolide Depot Case Study Sustained Release, the burst is described as a two-phase kinetic process consisting of an initial rapid phase followed by a secondary constant-rate phase. Understanding these phases is essential for designing formulations with reduced variability. Control of surface drug loading and pore connectivity is particularly important. Manufacturing conditions strongly influence these structural characteristics.
Primary and Secondary Burst Kinetics
Continuous monitoring of microspheres shows that the 24-hour burst includes two stages:
Primary (1°) Phase:
About 80% of the burst occurs in this stage. Drug on the surface or near the outer layer quickly dissolves upon contact with fluid. This phase is highly sensitive to surface porosity and drug distribution. Reducing surface drug lowers this component. Proper washing and process optimization help control this stage.
Secondary (2°) Phase:
The remaining 20% follows a slower diffusion-controlled pattern. Water penetration creates channels that allow gradual release. Polymer composition and internal morphology affect this phase. Controlling polymer density stabilizes release. Balanced hydration prevents excessive diffusion.
| Release Phase | Contribution (%) | Dominant Mechanism | Factors Influencing Phase |
|---|---|---|---|
| Initial Burst (Day 1) | 20%–25% | Surface desorption & pore diffusion | Particle size, surface porosity, loading |
| Zero-Order (Day 3-21) | 50%–60% | Matrix diffusion & polymer swelling | Polymer Mw, LA:GA ratio, end-groups |
| Final Phase (Day 21+) | 15%–20% | Bulk erosion & matrix collapse | Hydrolytic degradation rate, micro-pH |
Structural Determinants: Porosity and Span
The internal and external morphology of microspheres determines the magnitude of burst release. High-burst formulations often display hollow or wrinkled structures with substantial surface porosity, while successful depots such as Lupron Depot exhibit dense polymer cores with uniformly distributed pores. These structural differences often arise from solvent removal rates during manufacturing. Rapid evaporation of dichloromethane (DCM) can create channels that remain as permanent pores. Controlling solvent extraction therefore directly reduces burst release and improves release uniformity.
The “Span,” representing particle size distribution width, is also a critical quality attribute. A high Span indicates a larger fraction of fine particles that increase burst release due to higher surface area. Reducing the Span to ≤ 0.5 through wet sieving lowers initial burst from approximately 24% to about 14%. This adjustment also increases mean residence time (MRT) in vivo. Uniform particle size improves injectability and enhances reproducibility across batches.
Engineering Precision in the Manufacturing of Leuprolide Depot Case Study Sustained Release
Achieving sustained release requires a tightly controlled double-emulsion solvent evaporation process (W1/O/W2). Each parameter must be optimized to ensure high encapsulation efficiency and dense microsphere formation. Control of emulsification energy, viscosity, and solvent removal rate is essential. Minor deviations in process parameters can significantly affect drug distribution. Robust process control therefore improves batch consistency and release predictability.
Get a step-by-step guide on manufacturing this complex therapeutic: How to Develop Generic Leuprolide Depot
Primary Emulsion Optimization and the Cooling Mechanism
Formation of the primary emulsion involves dispersing aqueous leuprolide solution into the polymer phase. Because leuprolide is water-soluble, it may diffuse into the external phase. This reduces encapsulation efficiency. Viscosity control and cooling help stabilize droplets. Increased viscosity prevents droplet rupture. Cooling improves size stability.
ResolveMass applies two strategies:
Viscosity Enhancement via Gelatin
Adding ~1.5% gelatin increases internal phase viscosity. This stabilizes droplets during mixing. Drug migration is reduced. Emulsion stability improves. Encapsulation efficiency increases.
Ice-Bath Cooling
Cooling the primary emulsion increases polymer viscosity. This limits aqueous phase extraction. Encapsulation efficiency rises from ~50% to >70%. Cooling also reduces droplet coalescence.
Secondary Emulsification and Fluid Dynamics
The secondary emulsification step defines final particle size. Typically particles must be below 50 μm for injection. Fluid dynamics control droplet formation. Proper mixing prevents droplet rupture. Controlled turbulence produces uniform microspheres. These conditions support consistent sustained release.
Key parameters include Reynolds number, Weber number, and Kolmogorov length scale. Maintaining higher viscosity reduces droplet breakage. Controlled turbulence ensures uniform particles. These principles help scale laboratory processes to manufacturing.
Solvent Removal and “Annealing” Strategies
After emulsification, solvent removal hardens microspheres. The removal rate influences porosity. Slow removal allows drug diffusion. Rapid removal increases pore formation. Optimized conditions balance these effects. Proper drying produces dense microspheres.
Annealing involves heating near the glass transition temperature. This collapses internal voids and reduces residual solvent. Dense matrices reduce burst release. Residual solvent levels should remain below 1 ppm. Annealing also improves mechanical stability.
Analytical Validation of Microstructural Equivalence
Microstructural analysis confirms equivalence to reference products. ResolveMass Laboratories uses advanced analytical tools to characterize microspheres. Structural parameters correlate with release kinetics. Accurate characterization ensures reproducibility. Analytical validation supports regulatory requirements.
Ensure your generic submission meets all necessary testing standards: Review Analytical Requirements for ANDA Generic Drugs
Comprehensive Reverse Engineering Protocol
A systematic workflow is used:
- Mannitol and excipient removal using cold water
- Polymer extraction using THF or DCM
- Drug and gelatin separation via solvent extraction
- Molecular analysis using GPC, 1H NMR, and titration
These steps allow accurate comparison with reference formulations. Matching composition ensures consistent release. Strict tolerances are required.
| Component | Labeled Value (Lupron Depot) | Observed (Case Study) | Significance |
|---|---|---|---|
| Leuprolide Acetate | 8.5 wt% | 8.5 ± 0.1 wt% | Potency match |
| PLGA Content | 88.3 wt% | 88.3 wt% | Structural match |
| Gelatin | 1.5 wt% | 1.5 wt% | Viscosity match |
| D-Mannitol | 15.0 wt% | 15.0 wt% | Tonicity match |
| Residual DCM | < 600 ppm | < 1 ppm | Safety match |
Visualization and Porosity Assessment
SEM and CLSM imaging reveal internal microsphere structure. Successful depots show uniform honeycomb pores. High-burst formulations display large vacuoles. These structural differences affect release behavior. Controlled porosity reduces early diffusion. Imaging provides formulation insight.
Overall porosity of Lupron Depot is about 11.6%, compared with up to 47% in standard formulations. Lower porosity supports sustained release. Uniform pore distribution improves diffusion control. These characteristics enable predictable pharmacokinetics.
Clinical Significance: Maintaining the Therapeutic Window
Controlling burst release is clinically important. Leuprolide depots suppress LH and FSH levels. Continuous suppression maintains therapeutic hormone levels. Inconsistent release can affect treatment outcomes. Stable release improves safety and efficacy. Patients benefit from predictable dosing.
Mitigate safety concerns by identifying impurities early in development: Nitrosamine Risk Assessment in Generic Drugs
Pharmacokinetic and Pharmacodynamic Profiles
A successful depot maintains plasma concentration near 0.1 ng/mL. After injection, a temporary testosterone flare occurs. Excessive burst release may worsen symptoms. Controlled release minimizes this effect. Stable release improves tolerability.
Testosterone should drop to ≤ 50 ng/dL by Day 28. Suppression must continue until the next dose. Pediatric CPP formulations maintain estradiol < 20 pg/mL. Optimized depots show strong clinical performance. Consistent release supports long-term therapy.
Comparison of Long-Acting Formulations
Different durations require different polymer designs. One-month depots use 75:25 PLGA. Longer depots use PLA or high-lactide PLGA. Polymer selection controls degradation speed. Injectability must also be considered. Each duration presents unique formulation challenges.
| Product Type | Duration | Active Agent | Polymer Matrix | Key Challenge |
|---|---|---|---|---|
| 1-Month Depot | 4 Weeks | Leuprolide Acetate | 75:25 PLGA | Managing 20% burst |
| 3-Month Depot | 12 Weeks | Leuprolide Acetate | PLA or High LA PLGA | Avoiding late-stage lag |
| 4-Month Depot | 16 Weeks | Leuprolide Acetate | High Mw PLA | Injectability via gauge |
| 6-Month Depot | 24 Weeks | Leuprolide Acetate | Bio-erodible PLA | Long-term stability |
Overcoming Generic Development Challenges in Leuprolide Depot Case Study Sustained Release
Generic development is difficult due to complex PLGA microsphere behavior. Small variations can change release kinetics. Bioequivalence requires matching burst, plateau, and erosion phases. This creates a significant barrier. Tight formulation control is necessary. Advanced analytics improve success.
Decide the best path for your project: Outsourcing vs. Internal development: CRO vs. In-House ANDA Development
The Bioequivalence Hurdle
Regulatory agencies require pharmacokinetic similarity across all release phases. Establishing IVIVC is challenging. PLGA degradation creates acidic microenvironments. This may cause peptide modification. Modified peptide becomes inactive. Both total and active drug release must be measured.
Strategic Solutions for Generic Developers
ResolveMass recommends a Quality by Design approach. Microstructural equivalence should be prioritized. Critical parameters must be tightly controlled. Advanced analytical tools improve monitoring. These strategies increase bioequivalence success. Consistent processing ensures reliable release.
Future Directions in Sustained Release Peptide Delivery
Next-generation leuprolide depots focus on improved control and materials. Microfluidics enables uniform particle production. Reduced size variation lowers burst release. Precision engineering improves linear release. These advances support long-acting formulations. Predictability improves clinical outcomes.
Accelerate your time-to-market with specialized North American manufacturing: CDMO to Accelerate Generic Drug Development in US and Canada
Microfluidics and Precision Engineering
Microfluidic systems produce monodisperse microspheres. Particle size distribution becomes narrow. Reduced variability improves pharmacokinetics. Manufacturing reproducibility increases. These systems support precise sustained release. Controlled structure enhances performance.
AI and Predictive Modeling in Formulation Development
AI tools help optimize formulations. Machine learning predicts burst release behavior. Digital twins reduce trial-and-error experiments. Development timelines can be shortened. Predictive modeling improves design decisions. Bioequivalence success rates increase.
Conclusion of the Leuprolide Depot Case Study Sustained Release
This Leuprolide Depot Case Study Sustained Release shows that stable peptide delivery using PLGA microspheres requires careful chemical and process control. Ionic interactions, optimized double-emulsion processing, and detailed analytical validation help minimize burst release. These strategies maintain consistent hormone suppression and improve treatment outcomes. The same principles support next-generation long-acting injectables. Improved sustained release technology ultimately enhances patient compliance and therapeutic reliability.
For specialized expertise in overcoming complex depot development challenges and achieving predictable sustained-release profiles, partner with ResolveMass Laboratories:
https://resolvemass.ca/contact/
Frequently Asked Questions:
The initial burst release causes a sudden increase in leuprolide levels immediately after injection. This spike can trigger a temporary hormone surge, which may worsen symptoms in prostate cancer patients or cause rapid hormonal changes in children with CPP. Patients may experience bone pain, urinary complications, or temporary disease flare. Controlled sustained release reduces these risks and supports smoother therapeutic suppression.
ResolveMass achieves a narrow Span by carefully controlling secondary homogenization and maintaining stable fluid dynamics during emulsification. Balanced processing conditions help create uniform droplet sizes before microsphere hardening. Post-processing steps such as wet sieving remove very small and oversized particles. This results in consistent particle size distribution and more predictable release kinetics.
Gelatin increases the viscosity of the internal aqueous phase during double emulsion preparation. This thicker internal phase helps stabilize drug-loaded droplets and prevents them from breaking during mixing. Improved droplet stability leads to higher encapsulation efficiency. It also promotes uniform drug distribution within the PLGA matrix and reduces the risk of dose dumping.
Cooling the primary emulsion increases the viscosity of the polymer solution before secondary emulsification. The thicker polymer phase slows down diffusion of the water-soluble peptide into the external phase. As a result, the polymer solidifies faster and traps the drug inside the microspheres. This method improves drug loading and reduces peptide loss during processing.
The acid number reflects the amount of carboxylic acid end groups present on the PLGA polymer. These acidic groups interact with the positively charged leuprolide peptide through ionic bonding. Stronger interaction keeps the drug embedded within the matrix during early hydration. This reduces rapid diffusion and supports controlled sustained release as the polymer degrades.
Autocatalysis occurs when acidic degradation products from PLGA accumulate inside the microsphere. These acids lower the internal pH and accelerate further polymer breakdown. If not properly controlled, this can lead to faster-than-expected degradation. This may cause premature release or a sudden increase in drug release near the end of the dosing cycle.
One-month formulations typically use 75:25 PLGA, which degrades relatively quickly to allow shorter release duration. Six-month depots use PLA or high-lactide PLGA with higher molecular weight. These polymers are more hydrophobic and degrade more slowly. The slower hydration and erosion allow extended drug release over several months.
Glass transition temperature determines whether the polymer remains rigid at body temperature. A Tg above 37°C keeps the matrix in a stable glassy state. If Tg drops too low, the polymer becomes soft and drug diffusion increases. Maintaining a proper Tg ensures controlled release and prevents premature drug leakage.
Reference:
- Zhou, J., Walker, J., Ackermann, R., Olsen, K., Hong, J. K. Y., Wang, Y., & Schwendeman, S. P. (2020). Effect of manufacturing variables and raw materials on the composition-equivalent PLGA microspheres for 1-month controlled release of leuprolide. Molecular Pharmaceutics, 17(5), 1502–1515. https://doi.org/10.1021/acs.molpharmaceut.9b01188
- Kim, T. H., Park, T. G., Klose, D., Siepmann, F., Siepmann, J., & others. (2025). Release mechanisms of PLGA-based drug delivery systems: A review. Pharmaceutics. National Institutes of Health (PubMed Central). https://pmc.ncbi.nlm.nih.gov/articles/PMC12663523/
- Butreddy, A., Gaddam, R. P., Kommineni, N., Dudhipala, N., & Voshavar, C. (2021). PLGA/PLA-based long-acting injectable depot microspheres in clinical use: Production and characterization overview for protein/peptide delivery. International Journal of Molecular Sciences, 22(16), 8884. https://doi.org/10.3390/ijms22168884
- Butreddy, A., Gaddam, R. P., Kommineni, N., Dudhipala, N., & Voshavar, C. (2021). PLGA/PLA-based long-acting injectable depot microspheres in clinical use: Production and characterization overview for protein/peptide delivery. International Journal of Molecular Sciences, 22(16), 8884. https://doi.org/10.3390/ijms22168884
- Shore, N. D., Guerrero, S., Sanahuja, R. M., Gambús, G., & Parente, A. (2019). A new sustained-release, 3-month leuprolide acetate formulation achieves and maintains castrate concentrations of testosterone in patients with prostate cancer. Clinical Therapeutics, 41(3), 412–425. https://doi.org/10.1016/j.clinthera.2019.01.004
- Chen, C., Zhao, Y., Tang, K., Ning, H., Yu, X., Zhu, Y., & Shi, Q. (2025). Sustainable release artifact in PLGA microspheres for prolonged local anesthesia in postoperative pain management. Journal of Biomaterials Applications, 39(7), 714–722. https://pmc.ncbi.nlm.nih.gov/articles/PMC12856208/
- Marberger, M., Kaisary, A. V., Shore, N. D., Karlin, G. S., Savulsky, C., Mis, R., Leuratti, C., & Germa, J. R. (2010). Effectiveness, pharmacokinetics, and safety of a new sustained-release leuprolide acetate 3.75-mg depot formulation for testosterone suppression in patients with prostate cancer: A phase III, open-label, international multicenter study. Clinical Therapeutics, 32(4), 744–757. https://doi.org/10.1016/j.clinthera.2010.04.013



