
Introduction:
Peptide Drug Regulatory Requirements are among the most stringent in the pharmaceutical industry due to the complexity, sensitivity, and therapeutic importance of peptide-based drugs. As peptide therapeutics continue to grow in demand, regulatory agencies require comprehensive control over every stage of development and manufacturing.
For companies outsourcing production, Contract Development and Manufacturing Organizations (CDMOs) must not only meet these requirements but exceed them. Leveraging a strong bioanalytical strategy in drug development is essential to ensure regulatory success.
Share via:
Summary:
- Peptide Drug Regulatory Requirements demand strict compliance with global standards like GMP, ICH, and FDA/EMA guidelines.
- CDMOs must ensure quality, traceability, and validated processes throughout peptide manufacturing.
- Critical areas include raw material control, impurity profiling, analytical validation, and documentation.
- Regulatory success depends on robust quality systems, experienced teams, and advanced analytical capabilities.
- Choosing the right CDMO partner directly impacts approval timelines, product safety, and market success.
1: What Are Peptide Drug Regulatory Requirements?
Peptide Drug Regulatory Requirements are the comprehensive guidelines established by global regulatory authorities to ensure the safety, quality, and efficacy of peptide-based therapeutics.
These requirements govern every stage of the peptide lifecycle—from raw material sourcing to manufacturing, analysis, and final product release.
Key Regulatory Components Include:
- Good Manufacturing Practices (GMP) compliance to ensure consistent product quality
- ICH guidelines (Q7, Q8, Q9, Q10) for development, risk management, and quality systems
- FDA and EMA regulatory frameworks for market approval in the US and Europe
- Data integrity and traceability standards to ensure transparency and audit readiness
Peptide drugs are inherently sensitive to environmental conditions, synthesis variations, and degradation pathways, making strict regulatory oversight essential. Even minor deviations in manufacturing or handling can impact product stability, purity, and therapeutic performance.
Peptide drugs require advanced peptide characterization in drug development to ensure compliance, as even minor variations can affect product quality and therapeutic performance.
As a result, adherence to Peptide Drug Regulatory Requirements is critical not only for regulatory approval but also for ensuring patient safety and long-term product success.
2: Why Are Peptide Drug Regulatory Requirements More Stringent?
Peptide Drug Regulatory Requirements are more stringent because peptides are structurally complex and highly sensitive molecules, making them more vulnerable to degradation, impurities, and process variability.
Even minor changes in manufacturing conditions can significantly impact their quality, stability, and biological activity, which is why regulatory authorities impose tighter controls.
Key Reasons for Stringent Requirements:
- Structural complexity vs. small molecules
Peptides consist of amino acid sequences with specific folding and bonding patterns, making them more difficult to synthesize and control. - High sensitivity to environmental conditions
Peptides can degrade due to slight variations in temperature, pH, light, and solvents, affecting their stability and efficacy. - Higher risk of impurities
Common impurities include:- Deletion sequences
- Truncated peptides
- Oxidation products
- Aggregates
- Dependence of biological activity on precise structure
Unlike small molecules, even a small structural modification in a peptide can lead to loss of therapeutic function or altered pharmacological effects. - Complex manufacturing processes
Techniques like solid-phase peptide synthesis (SPPS) require tight control over each step to ensure consistency and purity.
To address these challenges, companies rely on mass spectrometry experts in drug development and advanced peptide analytical characterization services.
What This Means for CDMOs
Due to these challenges, CDMOs must:
- Maintain strict compliance with global Peptide Drug Regulatory Requirements
- Implement advanced process controls and monitoring systems
- Use high-resolution analytical techniques (e.g., LC-MS, HPLC)
- Establish robust validation and quality assurance frameworks
3: Core Peptide Drug Regulatory Requirements in Manufacturing
Peptide Drug Regulatory Requirements in manufacturing focus on ensuring consistent quality, safety, and traceability across the entire production lifecycle. CDMOs must implement robust systems, validated processes, and strict compliance frameworks to meet global regulatory expectations.
1. GMP Compliance for Peptide Drug Manufacturing
GMP compliance ensures that peptide drugs are consistently produced and controlled according to predefined quality standards.
Key Requirements:
- Controlled and classified manufacturing environments
- Qualified equipment and systems (IQ, OQ, PQ)
- SOP-driven and standardized operations
- Skilled and trained personnel
What CDMOs Must Deliver:
- Fully GMP-certified manufacturing facilities
- Routine regulatory audits and inspection readiness
- Well-documented compliance history and quality records
2. Raw Material Control and Traceability
Peptide Drug Regulatory Requirements mandate complete traceability of all raw materials used in peptide synthesis.
Regulatory Requirements:
- Vendor qualification and approval systems
- Certificates of Analysis (CoA) for all materials
- End-to-end batch traceability
CDMO Responsibilities:
- Robust supplier qualification programs
- Incoming material testing and verification
- Comprehensive documentation from sourcing to final product
3. Impurity Profiling and Control
Impurity profiling is a critical component of Peptide Drug Regulatory Requirements to ensure product purity and patient safety.
Critical for safety and regulatory approval. Advanced studies like GLP-1 peptide analytical characterization and peptide characterization case study of semaglutide demonstrate real-world compliance approaches.
Common Types of Impurities:
- Deletion sequences
- Truncated peptides
- Oxidation products
- Aggregates
CDMO Deliverables:
- Advanced analytical techniques (LC-MS, HPLC, UPLC)
- Defined impurity limits and acceptance criteria
- Stability-indicating analytical methods
4. Analytical Method Development and Validation
Validated analytical methods are essential to accurately assess peptide identity, purity, and potency.
Robust analytical methods are essential. Learn more about analytical method development for generic drugs and analytical development for generic drugs in Canada.
Regulatory Expectations:
- Specificity
- Accuracy
- Precision
- Linearity
- Robustness
What CDMOs Must Provide:
- Fully validated methods in accordance with ICH Q2(R2)
- Stability studies under various conditions
- Reliable batch release testing protocols
5. Process Validation and Control Strategy
Process validation ensures that peptide manufacturing consistently produces products meeting quality specifications.
Key Elements:
- Process design based on scientific understanding
- Process qualification through documented studies
- Continued process verification during routine production
CDMO Requirements:
- Scalable and reproducible manufacturing processes
- Identification of critical process parameters (CPPs)
- Risk-based control strategies aligned with regulatory expectations
6. Documentation and Data Integrity
Strong documentation and data integrity practices are central to Peptide Drug Regulatory Requirements and regulatory approvals.
For companies operating in North America, bioanalysis in Canada plays a key role in regulatory alignment.
Key Requirements:
- Compliance with ALCOA+ principles
(Attributable, Legible, Contemporaneous, Original, Accurate) - Secure electronic data management systems
- Complete audit trails for all activities
CDMO Must Ensure:
- Comprehensive and accurate batch manufacturing records
- Secure, validated, and compliant data systems
- Fully regulatory-ready documentation for inspections and submissions
Summary
Meeting Peptide Drug Regulatory Requirements in manufacturing requires a combination of GMP compliance, advanced analytics, validated processes, and robust data integrity systems. CDMOs that excel in these areas enable faster approvals, reduced risk, and consistent product quality.

4: Key Global Regulatory Guidelines for Peptide Drugs
Peptide Drug Regulatory Requirements are governed by internationally recognized guidelines that ensure consistent quality, safety, and efficacy across global markets. These frameworks guide CDMOs in developing compliant manufacturing and quality systems.
Major Regulatory Guidelines and Their Relevance
| Guideline | Focus Area | Relevance to Peptide Drugs |
|---|---|---|
| ICH Q7 | GMP for Active Pharmaceutical Ingredients (APIs) | Establishes core manufacturing compliance for peptide APIs |
| ICH Q8 | Pharmaceutical Development | Supports process understanding and design for robust peptide production |
| ICH Q9 | Quality Risk Management | Enables risk-based control strategies for peptide manufacturing |
| ICH Q10 | Pharmaceutical Quality System | Ensures lifecycle quality management and continuous improvement |
| FDA cGMP | Manufacturing Practices (USA) | Defines regulatory expectations for peptide drug approval in the U.S. |
| EMA Guidelines | European regulatory standards | Governs market authorization and compliance in the EU |
To navigate these frameworks, companies often partner with a pharmaceutical CDMO in the US and Canada or explore CDMO vs CRO for generic drug development.
Why These Guidelines Matter
These global frameworks are central to meeting Peptide Drug Regulatory Requirements, as they:
- Ensure consistent manufacturing quality across batches
- Enable risk-based decision-making during development
- Support regulatory submissions and approvals globally
- Promote lifecycle management and continuous improvement
What This Means for CDMOs
To comply with Peptide Drug Regulatory Requirements, CDMOs must:
- Align operations with ICH, FDA, and EMA expectations
- Implement integrated quality management systems (QMS)
- Maintain audit readiness and regulatory documentation
- Ensure global compliance for multi-region product launches
5: What CDMO Partners Must Deliver to Meet Peptide Drug Regulatory Requirements
To successfully meet Peptide Drug Regulatory Requirements, CDMO partners must deliver a combination of technical expertise, advanced infrastructure, and regulatory excellence. Their capabilities directly influence product quality, approval timelines, and commercial success.
1. Deep Technical Expertise
CDMOs must possess extensive experience in peptide synthesis and characterization to ensure precision and consistency.
Expected Capabilities:
- Solid-phase peptide synthesis (SPPS) expertise
- Handling of complex peptide modifications (cyclization, conjugation, etc.)
- Proven scale-up capabilities from R&D to commercial production
Why it matters:
Technical expertise minimizes process variability and ensures compliance with strict Peptide Drug Regulatory Requirements.
2. Advanced Analytical Infrastructure
High-quality analytical data is essential for demonstrating compliance with Peptide Drug Regulatory Requirements.
Essential for compliance—supported by peptide testing services.
Required Technologies:
- LC-MS/MS for impurity identification and quantification
- High-resolution mass spectrometry for structural confirmation
- HPLC/UPLC systems for purity and stability analysis
Why it matters:
Robust analytical capabilities ensure accurate characterization, impurity profiling, and regulatory acceptance.
3. Quality-by-Design (QbD) Approach
QbD ensures that quality is built into the manufacturing process rather than tested only at the final stage.
Key Benefits:
- Improved process robustness
- Reduced batch-to-batch variability
- Enhanced process understanding
- Faster and smoother regulatory approvals
Why it matters:
Regulatory agencies strongly favor QbD-based approaches as part of modern Peptide Drug Regulatory Requirements.
4. Regulatory Support and Filing Expertise
CDMOs must provide end-to-end regulatory support to meet global Peptide Drug Regulatory Requirements.
Includes DMF, IND, NDA submissions. Strong regulatory support for generic drugs in US and Canada CDMO is critical.
Supported Submissions:
- DMF (Drug Master File)
- IND (Investigational New Drug)
- NDA (New Drug Application)
Key Deliverables:
- Complete and compliant technical documentation
- Regulatory writing and submission support
- Full audit and inspection readiness
Why it matters:
Strong regulatory expertise reduces approval delays and ensures compliance across multiple regions.
5. Scalability and Technology Transfer
Efficient scale-up and technology transfer are critical for transitioning from development to commercialization.
CDMO Must Ensure:
- Consistent and reproducible processes from lab to commercial scale
- Well-defined and documented technology transfer protocols
- Minimal batch-to-batch variation
Why it matters:
Scalability ensures that processes remain compliant with Peptide Drug Regulatory Requirements while meeting commercial demands.
For commercialization success, companies often outsource peptide manufacturing to CDMO or work with a peptide drug development CDMO.
Explore options like:
Summary
Meeting Peptide Drug Regulatory Requirements requires CDMO partners to integrate scientific expertise, advanced analytics, quality-driven processes, and regulatory knowledge. The right partner not only ensures compliance but also accelerates time-to-market and reduces development risks.

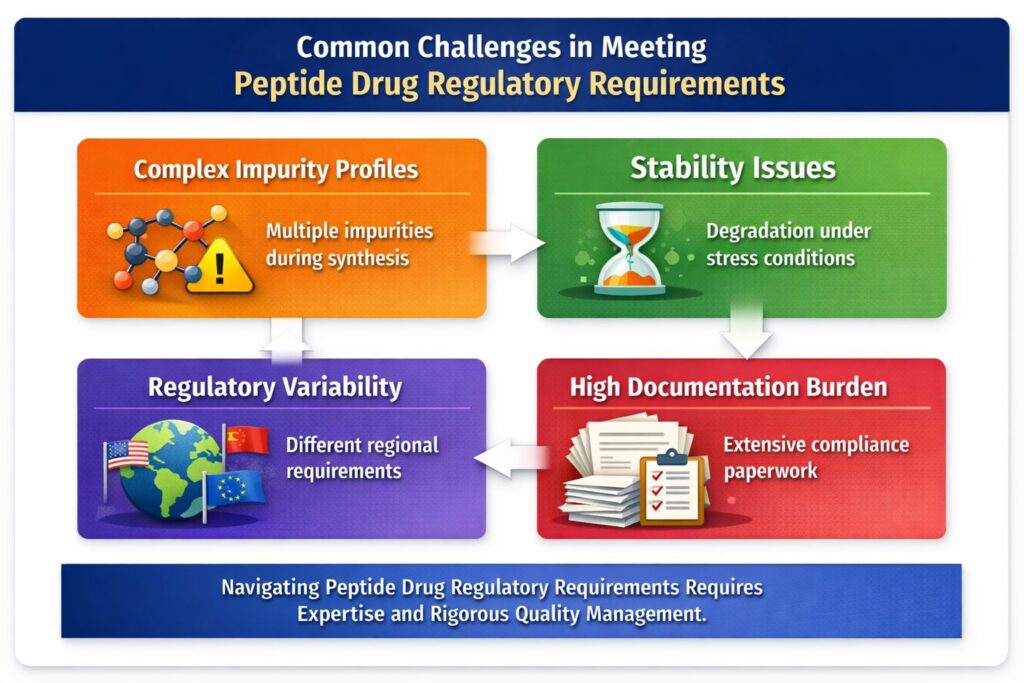
6: Common Challenges in Meeting Peptide Drug Regulatory Requirements
Meeting Peptide Drug Regulatory Requirements is challenging due to the inherent complexity of peptides, stringent global standards, and extensive compliance expectations. These challenges can impact timelines, costs, and regulatory approvals if not properly managed.
1. Complex Impurity Profiles
Peptide synthesis often leads to a wide range of impurities that must be identified, controlled, and quantified.
Requires advanced analysis such as generic peptide drug analytical characterization case study.
Key Challenges:
- Formation of deletion and truncated sequences
- Presence of oxidation products and aggregates
- Difficulty in separating structurally similar impurities
Impact:
Inadequate impurity control can lead to regulatory rejection or safety concerns, making advanced analytical characterization essential.
2. Stability Issues
Peptides are highly sensitive molecules that can degrade under environmental and processing conditions.
Key Factors Affecting Stability:
- Temperature fluctuations
- pH variations
- Light exposure
- Oxidative environments
Impact:
Stability issues can affect shelf life, potency, and efficacy, requiring robust formulation and stability studies to meet regulatory expectations.
3. Regulatory Variability Across Regions
Peptide Drug Regulatory Requirements can differ across regulatory authorities, creating complexity in global approvals.
Key Challenges:
- Differences between FDA, EMA, and other regional guidelines
- Varying documentation and submission formats
- Changing regulatory expectations over time
Impact:
Companies must adopt flexible and globally aligned regulatory strategies to ensure smooth approvals in multiple markets.
4. High Documentation Burden
Regulatory compliance requires extensive documentation at every stage of peptide drug development and manufacturing.
Key Requirements:
- Detailed batch manufacturing records
- Analytical validation reports
- Stability data and study reports
- Audit trails and data integrity documentation
Impact:
Managing large volumes of documentation increases operational complexity and requires strong data management systems to maintain compliance.
Companies also use reverse engineering for development of generic drugs and outsourced chemistry in drug discovery to overcome these challenges.
Summary
Overcoming these challenges in Peptide Drug Regulatory Requirements requires advanced analytical capabilities, robust quality systems, regulatory expertise, and proactive risk management. CDMOs that effectively address these areas can significantly improve compliance outcomes and accelerate product approval.
7: How ResolveMass Laboratories Inc. Supports Compliance
ResolveMass Laboratories Inc. supports compliance with Peptide Drug Regulatory Requirements by delivering high-quality analytical expertise, regulatory-ready data, and robust quality systems. Their integrated scientific approach ensures reliable, audit-ready outcomes for peptide drug development.
Key Capabilities That Support Regulatory Compliance
1. Advanced Analytical Characterization
ResolveMass provides comprehensive analytical solutions to ensure accurate peptide identification and quality assessment.
- High-resolution mass spectrometry
- LC-MS/MS-based characterization
- Structural confirmation and purity analysis
Impact:
Enables precise data generation required for regulatory submissions and approvals.
2. Expertise in Impurity Profiling and Metabolite Analysis
Understanding impurities is critical for meeting Peptide Drug Regulatory Requirements.
- Identification of process-related and degradation impurities
- Advanced impurity profiling strategies
- In-depth metabolite analysis
Impact:
Ensures safety, quality, and compliance with regulatory impurity thresholds.
3. Regulatory-Ready Data Packages
ResolveMass delivers documentation aligned with global regulatory expectations.
- Comprehensive analytical reports
- Method validation data
- Stability and characterization data packages
Impact:
Facilitates smoother DMF, IND, and NDA submissions with reduced regulatory queries.
4. Strong Focus on Data Integrity and Quality Systems
Compliance is built on reliable and transparent data practices.
- Adherence to ALCOA+ principles
- Secure and validated data systems
- Audit-ready documentation and traceability
Impact:
Ensures full alignment with global Peptide Drug Regulatory Requirements and inspection readiness.
Their expertise in peptide sameness study services in Canada and case studies like peptide characterization of ganirelix generic project demonstrate proven regulatory success.
They also support outsourcing generic drug development in Canada through integrated CDMO solutions such as:
- CDMO for generic drug development in Canada
- CDMO for generic projects in Canada
- Generic pharmaceutical CDMO Canada
Integrated Compliance Approach
ResolveMass Laboratories Inc. combines scientific expertise, advanced technology, and regulatory understanding to provide end-to-end support for peptide drug development.
Their integrated approach enables clients to:
- Achieve confident and successful regulatory outcomes
- Meet stringent regulatory expectations
- Reduce compliance risks
- Accelerate development timelines
Conclusion:
Meeting Peptide Drug Regulatory Requirements is essential for ensuring the safety, efficacy, and approval of peptide therapeutics. CDMO partners play a crucial role in achieving compliance by delivering robust quality systems, advanced analytical capabilities, and regulatory expertise.
Choosing the right partner—whether for peptide CDMO vs CRO evaluation or full-scale development—is not just strategic; it is essential for regulatory success and long-term market impact.
Frequently Asked Questions:
Peptide Drug Regulatory Requirements are a set of strict guidelines established by global authorities like the FDA, EMA, and ICH. These regulations ensure that peptide drugs are safe, effective, and of high quality. They cover every stage, including raw material sourcing, manufacturing, testing, and documentation. Due to the sensitive nature of peptides, even minor deviations must be controlled. Compliance is essential for regulatory approval and patient safety.
Regulatory requirements are stricter because peptides are structurally complex and highly sensitive molecules. They can easily degrade under environmental conditions like temperature, pH, and light. Additionally, peptide synthesis often produces multiple impurities that must be carefully controlled. Even small structural changes can affect biological activity. Therefore, tighter controls are required to ensure consistency, safety, and efficacy.
CDMOs play a critical role by providing expertise in manufacturing, analytical testing, and regulatory compliance. They ensure adherence to GMP standards and support process validation and quality control. CDMOs also assist with regulatory submissions such as DMF, IND, and NDA. Their infrastructure and experience help reduce development risks and timelines. Choosing the right CDMO is key to successful regulatory approval.
Analytical techniques such as LC-MS/MS, HPLC, UPLC, and high-resolution mass spectrometry are widely used. These methods help in identifying, quantifying, and characterizing impurities. They also ensure peptide purity, structure, and stability. Advanced analytical tools are essential due to the complexity of peptide molecules. Accurate analytical data is crucial for regulatory submissions and approvals.
Key regulatory guidelines include ICH Q7 (GMP for APIs), Q8 (pharmaceutical development), Q9 (risk management), and Q10 (quality systems). Additionally, FDA cGMP and EMA guidelines govern manufacturing and approvals in the US and Europe. These frameworks ensure consistency, safety, and quality across the product lifecycle. They also promote risk-based approaches and continuous improvement. Compliance with these guidelines is mandatory for global market access.
Common impurities include deletion sequences, truncated peptides, oxidation products, and aggregates. These impurities can form during synthesis, purification, or storage. Some impurities are structurally similar to the desired peptide, making them difficult to detect. If not controlled, they can affect safety and efficacy. Therefore, advanced impurity profiling and strict limits are required for regulatory compliance.
Quality-by-Design (QbD) focuses on building quality into the process rather than testing it at the end. It involves understanding critical process parameters and their impact on product quality. QbD improves process robustness and reduces variability. It also helps in identifying risks early in development. Regulatory agencies encourage QbD as it leads to more efficient and reliable approvals.
Reference
- Koleng J, Owens III DE, Hannon K. How The Right CDMO Partner Can Accelerate Biologics Development.https://www.bioprocessonline.com/doc/how-the-right-cdmo-partner-can-accelerate-biologics-development-0001
- Shaffer C. Making the Most of a CDMO Relationship: CDMOs and their pharma and biopharma clients could benefit from a little relationship counseling—just enough to clarify key issues like scope of services. Genetic Engineering & Biotechnology News. 2020 Apr 1;40(4):54-6.https://journals.sagepub.com/doi/abs/10.1089/gen.40.04.13
- Hotha KK. Fast-to-Clinic, Fast-to-Market in Biotech Innovation: Optimizing Chemistry Manufacturing & Controls (CMC) Excellence for Clinical and Commercial Success. Advances in Chemical Engineering and Science. 2024;14(3):155-71.https://drhothas.com/wp-content/uploads/2024/09/Fast-to-Clinic-Fast-to-Market-in-Biotech-Innovation-Optimizing-Chemistry-Manufacturing-Controls-CMC-Excellence-for-Clinical-and-Commercial-Success.pdf
- Spichiger D. CordenPharma-The CDMO Partner You Need. CHIMIA. 2025 May 28;79(5):378-.https://www.chimia.ch/chimia/article/download/2025_378/2025_378
- Taylor M. An Experienced CDMO Can Be a Differentiator in the Rapidly Growing Biologics Market.https://www.scorpiusbiologics.com/hubfs/SCRP88_Article01.pdf


