Introduction
Protein biomarker identification stands at the forefront of modern biomedical innovation, enabling researchers to decode complex biological systems with unprecedented precision. As diseases become more multifactorial and patient-specific, the demand for highly sensitive and reliable biomarkers continues to grow. Proteins, due to their dynamic nature and direct involvement in cellular processes, provide a powerful window into both normal physiology and disease pathology.
At Resolvemass Laboratories, we focus on bridging technological advancements with real-world clinical applications by offering comprehensive protein biomarker discovery solutions. Through a combination of advanced analytical platforms and data-driven insights, we empower researchers to uncover meaningful biological signals that drive innovation in diagnostics, therapeutics, and personalized medicine.
Share via:
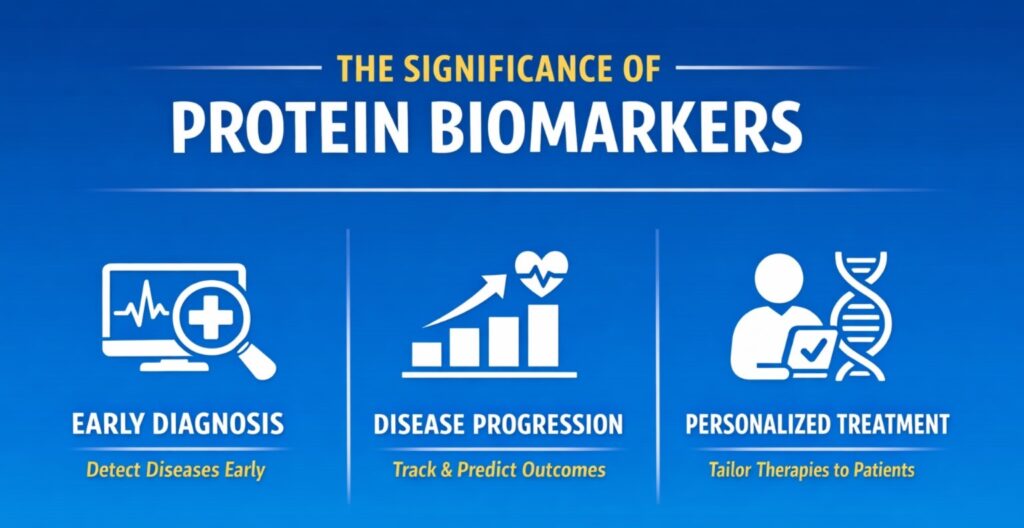
The Significance of Protein Biomarkers
Protein biomarkers serve as measurable indicators of disease presence, progression, and treatment response. Their significance spans various areas of clinical research and therapeutic development:
Disease Diagnosis
- Early Detection: Protein biomarkers can identify diseases before clinical symptoms arise. For instance, elevated levels of certain proteins in blood, such as prostate-specific antigen (PSA) for prostate cancer, can signal the presence of cancer well before it becomes symptomatic.
- Diagnostic Precision: Specific biomarkers enhance diagnostic accuracy by distinguishing between diseases with overlapping symptoms. For example, biomarkers like amyloid-beta and tau are crucial for differentiating Alzheimer’s disease from other neurodegenerative disorders.
Disease Prognosis
- Disease Progression: Monitoring biomarkers allows for tracking disease progression and predicting outcomes. In chronic diseases like diabetes, biomarkers such as HbA1c levels reflect long-term glucose control and predict disease progression and complications.
- Personalized Treatment: Biomarker profiles enable the development of personalized treatment plans. By assessing biomarkers like HER2 in breast cancer, clinicians can tailor treatment strategies to the specific characteristics of the tumor, improving efficacy and reducing side effects.
Therapeutic Development
- Target Identification: Identifying proteins involved in disease pathways provides potential drug targets. For instance, targeting specific proteins like VEGF (vascular endothelial growth factor) has led to the development of targeted therapies for cancer and other diseases.
- Treatment Monitoring: Biomarkers help evaluate the effectiveness of new treatments. Monitoring biomarkers such as C-reactive protein (CRP) can indicate the success of anti-inflammatory treatments in conditions like rheumatoid arthritis.
Biomarker Validation
- Clinical Validation: Ensuring that biomarkers are reliable and reproducible in clinical settings is essential for their successful application in diagnostics and therapeutics. Rigorous validation processes are required to confirm that biomarkers are both clinically relevant and consistently measurable.
Challenges in Protein Biomarker Discovery
Despite significant advancements, protein biomarker discovery faces several technical and biological challenges. One major hurdle is the vast dynamic range of protein concentrations in biological samples, where low-abundance proteins—often the most clinically relevant—are difficult to detect amidst highly abundant ones. This necessitates the use of enrichment techniques and highly sensitive instrumentation.
Another critical challenge lies in biological variability among patients. Factors such as age, lifestyle, genetic background, and environmental exposure can influence protein expression levels, making it difficult to establish universal biomarkers. Ensuring reproducibility across diverse populations requires rigorous validation protocols and large-scale studies, which can be both time-consuming and resource-intensive.
Explore more – Bioanalytical services
Emerging Trends in Protein Biomarker Research
The field of protein biomarker research is rapidly evolving with the integration of advanced technologies and interdisciplinary approaches. One of the most notable trends is the shift toward multi-omics integration, where proteomics data is combined with genomics, transcriptomics, and metabolomics to provide a more holistic understanding of disease biology. This integrative approach enhances the discovery of robust biomarkers by capturing multiple layers of biological regulation.
Additionally, the adoption of artificial intelligence and machine learning is transforming how biomarker data is analyzed. These technologies enable the identification of hidden patterns within complex datasets, significantly improving the accuracy of biomarker discovery. As computational tools become more sophisticated, researchers can predict disease outcomes and treatment responses with greater confidence, paving the way for smarter and faster clinical decisions.
Methodologies for Protein Biomarker Identification
Resolvemass Laboratories utilizes a suite of advanced methodologies to identify and validate protein biomarkers. These techniques are essential for obtaining accurate and reliable biomarker data.
Mass spectrometry is a pivotal technique for protein biomarker identification due to its sensitivity, resolution, and ability to provide detailed structural information.
- Shotgun Proteomics
- Principle: Proteins are enzymatically digested into peptides, which are then analyzed using liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). The resulting peptide fragmentation patterns are used to infer the presence and quantity of proteins.
- Applications: This technique is ideal for comprehensive protein profiling and the discovery of novel biomarkers. It allows for the analysis of complex biological samples, such as blood or tissue extracts, providing a broad view of protein expression and modification.
- Targeted Proteomics
- Principle: Focuses on specific proteins or peptides of interest using techniques like Selected Reaction Monitoring (SRM) or Multiple Reaction Monitoring (MRM) in LC-MS/MS. These techniques provide quantitative data by monitoring specific ion transitions associated with the target proteins.
- Applications: Targeted proteomics is used for the precise quantification of known biomarkers and is essential for validating candidate biomarkers identified through shotgun proteomics. It offers high sensitivity and specificity, making it suitable for clinical applications.
Liquid chromatography separates proteins or peptides based on their physicochemical properties before mass spectrometric analysis, enhancing resolution and sensitivity.
- High-Performance Liquid Chromatography (HPLC)
- Principle: HPLC separates proteins based on their interactions with a stationary phase in a column and a mobile phase that passes through it under high pressure.
- Applications: This technique provides high resolution and is commonly used in conjunction with mass spectrometry to separate and analyze complex protein mixtures. It is essential for achieving clean, well-resolved peptide peaks, which improves the accuracy of downstream analyses.
- Reversed-Phase Liquid Chromatography (RP-LC)
- Principle: RP-LC separates proteins based on their hydrophobicity, using a non-polar stationary phase and a polar mobile phase.
- Applications: Effective for separating and analyzing peptides and proteins based on their hydrophobic interactions. RP-LC enhances the resolution and sensitivity of peptide identification, especially in complex biological samples.
3. Immunoassays
Immunoassays use specific antibodies to detect and quantify proteins in samples, providing high specificity and sensitivity.
- Enzyme-Linked Immunosorbent Assay (ELISA)
- Principle: ELISA involves the use of antigen-antibody interactions to quantify proteins. An enzyme conjugated to the antibody produces a detectable signal upon binding to the target protein.
- Applications: ELISA is widely used for quantifying biomarkers in clinical research and diagnostics. It is suitable for analyzing proteins in various sample types, including blood, urine, and tissue extracts.
- Western Blotting
- Principle: Proteins are separated by gel electrophoresis, transferred to a membrane, and detected using specific antibodies that bind to the target proteins.
- Applications: Western blotting allows for the detection and quantification of specific proteins and their isoforms. It is often used to confirm the presence of biomarkers and analyze protein expression levels.
4. Bioinformatics Tools
Bioinformatics tools are crucial for analyzing and interpreting complex data from proteomics and other analytical techniques.
- Protein Database Searching
- Principle: Compares experimental mass spectrometry data with theoretical protein sequences in databases (e.g., NCBI, UniProt) to identify proteins.
- Applications: Facilitates the identification of known proteins and biomarker candidates by matching experimental data with reference databases. This approach helps in validating identified proteins and understanding their biological roles.
- Data Analysis and Visualization
- Principle: Uses software tools to analyze quantitative data, identify significant biomarkers, and visualize results through graphical representations and statistical analysis.
- Applications: Assists in interpreting complex datasets, identifying trends, and understanding the biological significance of biomarkers. Visualization tools help in communicating findings effectively and supporting decision-making processes.
Role of Sample Quality in Biomarker Identification
The accuracy of protein biomarker identification is heavily dependent on the quality of the biological samples used. Improper sample collection, storage, or handling can lead to protein degradation or modification, ultimately affecting the reliability of analytical results. Standardized protocols for sample processing are essential to minimize variability and maintain protein integrity.
Moreover, pre-analytical factors such as temperature fluctuations, freeze-thaw cycles, and contamination can introduce significant biases in proteomic data. Implementing strict quality control measures and using appropriate preservatives can help mitigate these issues. High-quality samples ensure that the identified biomarkers truly reflect the biological condition being studied rather than experimental artifacts.
Explore more – Proteomics
The Resolvemass Laboratories Advantage
At Resolvemass Laboratories, we offer a comprehensive suite of services for protein biomarker identification, leveraging our expertise and advanced technologies to deliver high-quality results.
1. Customized Experimental Design
We collaborate closely with clients to design experimental protocols tailored to their specific research objectives:
- Sample Collection and Preparation: We optimize protocols for protein extraction and preparation to ensure high-quality samples suitable for analysis. This includes techniques for protein solubilization, concentration, and removal of contaminants.
- Biomarker Discovery and Validation: Our approach involves a combination of mass spectrometry, immunoassays, and bioinformatics to discover and validate potential biomarkers. We ensure that biomarkers are robust, reproducible, and relevant to the research goals.
2. Advanced Analytical Technologies
We utilize state-of-the-art technologies to provide precise and comprehensive protein biomarker data:
- High-Resolution Mass Spectrometers: Instruments such as orbitrap and time-of-flight (TOF) mass spectrometers offer exceptional mass accuracy and resolution, enabling detailed protein analysis and biomarker identification.
- High-Resolution Liquid Chromatography: Techniques like HPLC and reversed-phase LC enhance the separation and resolution of proteins and peptides, improving the sensitivity and specificity of biomarker analysis.
Quantitative vs Qualitative Proteomics in Biomarker Discovery
Proteomics approaches can broadly be categorized into qualitative and quantitative analyses, both of which play vital roles in biomarker research. Qualitative proteomics focuses on identifying proteins present in a sample, providing insights into protein composition and potential biomarker candidates. This approach is particularly useful during the initial discovery phase.
In contrast, quantitative proteomics measures the abundance of proteins across different conditions or sample groups. Techniques such as label-free quantification and isotope labeling enable precise comparisons, helping researchers identify proteins that are significantly upregulated or downregulated in disease states. Combining both approaches offers a comprehensive understanding of protein dynamics and enhances biomarker validation.
3. Expert Data Analysis
Our bioinformatics team employs advanced tools for analyzing and interpreting complex data:
- Comprehensive Data Interpretation: We provide detailed analysis of protein data, including biomarker identification, functional analysis, and correlation with disease states.
- Customized Reporting: Our reports are tailored to client needs, including graphical representations, statistical analysis, and actionable insights. We ensure that our findings are clear, relevant, and useful for decision-making.
Regulatory Considerations in Biomarker Development
The translation of protein biomarkers from research to clinical application requires compliance with stringent regulatory standards. Regulatory agencies emphasize the need for robust validation, reproducibility, and clinical relevance before approving biomarkers for diagnostic or therapeutic use. This ensures patient safety and reliability of test results.
Furthermore, documentation and standardization of analytical methods are critical components of regulatory approval. Laboratories must demonstrate consistency in their processes and maintain transparency in data reporting. Adhering to regulatory guidelines not only facilitates clinical adoption but also enhances the credibility of biomarker-based solutions in the healthcare industry.
Future Perspectives in Protein Biomarker Identification
The future of protein biomarker identification is poised for remarkable growth, driven by innovations in analytical technologies and computational biology. Emerging tools such as single-cell proteomics are enabling researchers to study protein expression at an unprecedented resolution, uncovering cellular heterogeneity that was previously undetectable. This has significant implications for understanding complex diseases like cancer.
In addition, advancements in microfluidics and automation are streamlining workflows, reducing sample requirements, and increasing throughput. These developments make biomarker research more efficient and accessible. As technology continues to evolve, the integration of real-time data analysis and personalized healthcare solutions will further revolutionize the field, bringing precision medicine closer to everyday clinical practice.
Case Study: Identifying Cardiovascular Disease Biomarkers
A research institute engaged Resolvemass Laboratories to identify protein biomarkers associated with cardiovascular disease.
Challenges
- Complex Biological Samples: The study involved analyzing blood plasma samples with a wide range of protein concentrations and potential biomarkers.
- Biomarker Discovery: The objective was to identify biomarkers indicative of early-stage cardiovascular disease and predict patient risk.
Solution
- Sample Preparation and Analysis: We utilized advanced LC-MS/MS techniques to profile proteins and identify potential biomarkers. Enrichment strategies were employed to focus on low-abundance proteins.
- Immunoassays for Validation: Followed up with ELISA and Western blotting to validate identified biomarkers and confirm their relevance.
- Bioinformatics Analysis: Used database searches and data analysis tools to interpret results, identify significant biomarkers, and understand their roles in disease mechanisms.
Outcome
Our approach led to the identification of several novel biomarkers associated with cardiovascular disease. These biomarkers are now being used to develop diagnostic tests and therapeutic strategies aimed at early detection and personalized treatment.
Conclusion
Protein biomarker identification continues to reshape the landscape of biomedical research by enabling deeper insights into disease mechanisms and patient-specific variations. As technologies advance and analytical methods become more refined, the potential for discovering highly specific and clinically relevant biomarkers grows exponentially. These developments are crucial for improving early diagnosis, monitoring disease progression, and designing targeted therapeutic strategies.
At Resolvemass Laboratories, our commitment lies in delivering innovative, reliable, and high-quality biomarker solutions that support cutting-edge research and clinical advancements. By combining expertise, technology, and a forward-looking approach, we aim to empower scientists and healthcare professionals in their pursuit of precision medicine and improved patient outcomes.
Contact Us
For more information about our protein biomarker identification services and how we can assist with your research and development needs, please Resolvemass laboratories & contact us
Frequently Asked Questions
Common techniques include mass spectrometry, liquid chromatography, immunoassays like ELISA, and bioinformatics analysis. These methods allow researchers to detect, separate, and quantify proteins with high precision. Combining multiple techniques often provides more comprehensive and reliable results.
One major challenge is detecting low-abundance proteins in complex biological samples. Variability between individuals and sample handling issues can also affect results. Additionally, validating biomarkers for clinical use requires extensive testing and large datasets, making the process time-consuming.
Protein biomarkers help tailor treatments based on an individual’s unique biological profile. By analyzing specific protein expressions, doctors can select therapies that are more effective and have fewer side effects. This approach improves treatment success rates and enhances patient care.
Diagnostic biomarkers are used to detect or confirm the presence of a disease, while prognostic biomarkers provide information about disease progression and likely outcomes. Both types are essential in clinical decision-making, helping guide treatment strategies and patient management.
Biomarker validation can take several months to years, depending on the complexity of the study. It involves multiple stages, including experimental testing, clinical trials, and regulatory approval. Thorough validation ensures that the biomarker is accurate, reliable, and suitable for clinical use.
Reference:
- National Institutes of Health. (n.d.). Protein biomarker identification and analysis. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC12806446/
- Liu, T., Chen, H., Song, Y., Ye, X., Wang, D., Zhang, Q., Liu, H., Sha, J., Du, L., Qi, S., Geng, Z., Hu, Q., Wang, Y., Cai, M., Chen, D., Song, H., Pan, J., Chen, Y., Ling, T., Chang, C., & Bai, B. (2025). Plasma fractionation and mixture improves coverage in proteomic analysis. Journal of Proteome Research, 24(12), 6215–6225. https://doi.org/10.1021/acs.jproteome.5c00814
- Zhou, Z., Liu, C., Feng, S., Chen, J., Chen, T., Zhu, J., Wu, S., Zhou, C., Huang, C., Xue, J., Qin, X., & Zhan, X. (2024). Identification of novel protein biomarkers and therapeutic targets for ankylosing spondylitis using human circulating plasma proteomics and genome analysis. Analytical and Bioanalytical Chemistry, 416(28), 6357–6366. https://doi.org/10.1007/s00216-024-05521-4


