Introduction: When the Synthesis Route Itself Becomes the Problem
The pharmaceutical industry’s encounter with nitrosamine contamination has revealed an important reality: nitrosamine formation pathways in API synthesis are rarely accidental or random contamination events. Instead, they arise as a direct outcome of specific chemical environments unintentionally created during the manufacturing of drug substances. A clear understanding of how chemistry-related process decisions generate nitrosamine risk is essential for establishing a long-term and scientifically reliable control strategy.
Regulatory authorities, including the U.S. Food and Drug Administration and European Medicines Agency, have emphasized this expectation within their nitrosamine guidance frameworks. Manufacturers are now expected to perform root-cause-driven risk assessments that critically evaluate the synthetic route itself rather than relying solely on finished-product screening. This regulatory shift transfers responsibility from only the quality control laboratory to the process chemistry function, significantly increasing the importance of every reagent selection, solvent system, process parameter, and reaction sequence decision made during API development and manufacturing.
Need expert help with your API evaluation? Explore our Nitrosamine Analysis Services
Share via:
Article Summary:
- Nitrosamine generation during API manufacturing is typically not a random event; it is a foreseeable outcome driven by specific synthetic route and process chemistry choices.
- The most common mechanism involves reactions between secondary amines and nitrosating species, although solvent selection, quenching strategy, and pH conditions can significantly influence impurity formation.
- Commonly used amide solvents such as DMF, DMA, and NMP may decompose under acidic or elevated-temperature environments, producing dimethylamine, a well-known precursor for nitrosamine impurities.
- Sodium nitrite and related reagents employed in diazotization or downstream quench operations can act as active nitrosating sources within the reaction system.
- Tertiary amines including TEA and DIPEA are also relevant from a risk perspective because they may undergo N-dealkylation under harsh processing conditions, forming secondary amines capable of nitrosamine formation.
- Multi-product manufacturing equipment and inadequate campaign segregation can introduce additional nitrosamine hazards through residual nitrite or amine carryover, even if the intended synthesis pathway appears low risk.
- Effective nitrosamine mitigation begins during process and route development, where chemistry and engineering decisions are made, rather than relying exclusively on downstream quality control testing.
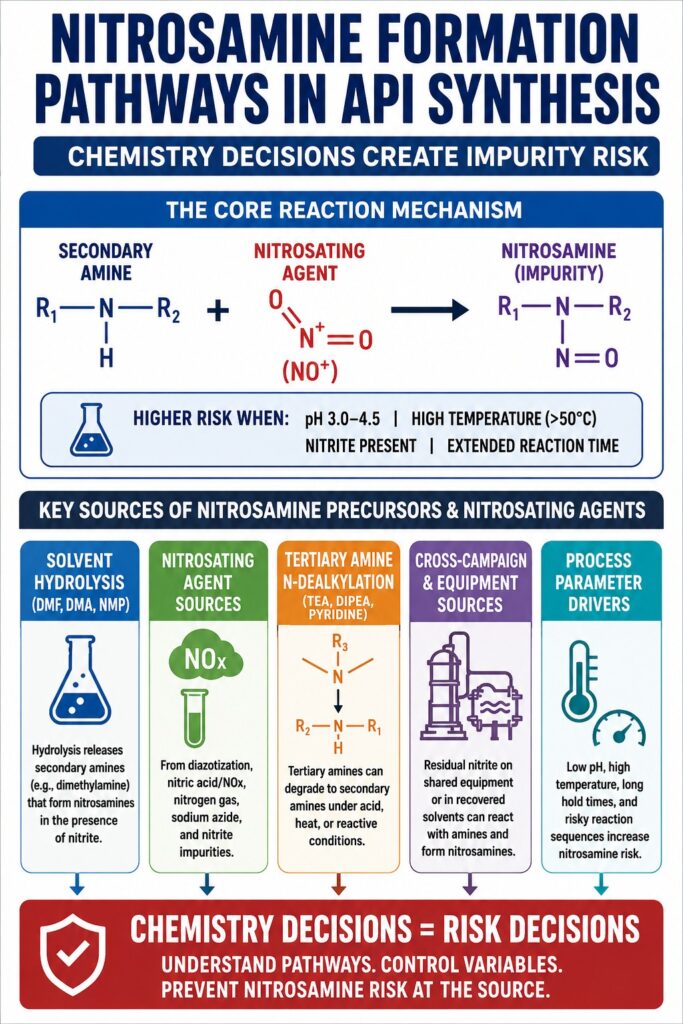
The Core Reaction Mechanism: Secondary Amines and Nitrosating Agents
Nitrosamines are generated when a secondary amine reacts with a nitrosating agent under conditions that facilitate electrophilic attack on the nitrogen atom. The reaction rate and overall extent of nitrosamine formation are strongly influenced by process variables such as pH, temperature, and the concentration of reactive intermediates. Each of these variables is directly controlled by process chemistry decisions made during API synthesis.
The classical nitrosation mechanism proceeds through the formation of a nitrosonium ion (NO⁺) or related nitrogen oxide species such as N₂O₃ or N₂O₄. These reactive species attack the lone electron pair present on the secondary amine. For many aliphatic secondary amines, the reaction rate reaches its maximum within a pH range of approximately 3 to 4 because nitrous acid (HNO₂) exists in a sufficiently unionized and reactive form while the amine remains partially unprotonated and chemically available for reaction.
Key Variables That Accelerate Nitrosamine Formation in API Synthesis
| Variable | Risk-Amplifying Condition | Mechanistic Reason |
|---|---|---|
| pH | 3.0–4.5 | Optimal coexistence of unionized HNO₂ and reactive free amine |
| Temperature | Elevated temperatures (>50°C) | Faster reaction kinetics and accelerated degradation of amide solvents |
| Nitrite concentration | Even ppm-level residual nitrite | Trace nitrite concentrations remain sufficiently reactive with susceptible amines |
| Amine structure | Cyclic secondary amines such as morpholine or piperazine | Greater nucleophilicity and reduced steric hindrance increase reactivity |
| Reaction time | Extended process hold times | Equilibrium progressively favors nitrosamine accumulation |
| Ionic strength | High salt concentration environments | Transition-state stabilization may enhance nitrosation efficiency |
Developing new testing protocols? Learn about GC-MS Method Development for Nitrosamine Testing
Solvent-Derived Nitrosamine Precursors: The Hidden Risk Associated with DMF, DMA, and NMP
Amide solvents, particularly N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMA), and N-methylpyrrolidone (NMP), represent significant secondary amine precursor sources when exposed to hydrolytic conditions. Their use in the presence of nitrosating agents creates a substantial and frequently underestimated nitrosamine risk during API synthesis.
This pathway remains one of the most overlooked nitrosamine formation mechanisms in pharmaceutical manufacturing. These solvents are widely used because of their outstanding solvating capability and compatibility with diverse reaction systems. However, under acidic aqueous conditions or elevated temperatures, they undergo hydrolysis reactions that release reactive amine species:
- DMF → Dimethylamine (DMA) + Formic acid
- DMA (solvent) → Dimethylamine + Acetic acid
- NMP → 4-(methylamino)butanoic acid → ultimately methylamine-containing species
Dimethylamine, which is a secondary amine, readily reacts with nitrosating agents present within the process stream to generate NDMA (N-nitrosodimethylamine), the nitrosamine impurity central to the well-known valsartan contamination incident.
Situations Where DMF Hydrolysis Becomes a Significant Risk
- Aqueous workup operations following DMF-based reactions, particularly when acidic conditions are used
- Spray-drying or distillation operations where DMF removal occurs at elevated temperatures under vacuum conditions, accelerating hydrolysis
- Extended reaction hold periods above 60°C in the presence of even trace quantities of water
- Processes using DMF as a co-solvent together with acid chlorides or acid anhydrides, both of which promote amide hydrolysis
The critical point is that the API structure itself may not contain a secondary amine. Instead, the nitrosamine precursor may originate entirely from the process solvent. Without targeted analytical screening, the release of dimethylamine can remain undetected throughout routine manufacturing operations.
Mitigate risk in your solvent systems: Nitrosamine Solvent and Catalyst Mitigation Strategies
Nitrosating Agent Sources Within API Synthesis: Beyond Sodium Nitrite
Nitrosating agents encountered during API synthesis are not limited to intentionally added sodium nitrite used in diazotization chemistry. They may also arise from degradation products of nitrogen-containing reagents, atmospheric nitrogen oxide exposure, or trace impurities present in commonly used raw materials.
This distinction is extremely important because risk assessments focused exclusively on explicit sodium nitrite usage frequently fail to identify many real-world nitrosamine formation pathways encountered in commercial pharmaceutical manufacturing.
Nitrosating Agent Sources That Require Investigation
1. Diazotization and Sandmeyer Chemistry
The deliberate use of sodium nitrite for diazonium salt generation is the most widely recognized nitrosamine risk step. However, the associated risk extends beyond the diazotization reaction itself. Residual nitrite carried into downstream processing stages can react with secondary amines introduced later in the synthesis route. Therefore, quenching efficiency for excess nitrite must be experimentally validated rather than assumed.
2. Nitric Acid Residuals and Nitrogen Oxide Species
Processes involving nitric acid during nitration reactions generate nitrogen oxide byproducts including NO, NO₂, and N₂O₃. If residual secondary amines remain present downstream, these species can function as highly effective nitrosating agents. Consequently, closed-system processing and efficient NOx scrubbing are not merely operational preferences but mechanistically necessary risk mitigation measures.
3. Nitrogen Gas Contamination
Industrial nitrogen gas used for reactor blanketing may contain trace levels of NOx contaminants, particularly when generated through pressure swing adsorption (PSA) systems lacking sufficient deoxygenation capability. In the presence of moisture, trace NO₂ converts into HNO₂, which acts as a potent nitrosating agent.
Ensuring compliance for complex submissions? Nitrosamine Risk Assessment for ANDA Submission
4. Sodium Azide in API Synthesis
Azide chemistry, commonly utilized in the preparation of biopharmaceutical intermediates, introduces highly nitrogen-rich species into the synthetic environment. Under acidic conditions, hydrazoic acid (HN₃) may decompose through pathways capable of generating reactive nitrogen-containing intermediates. Although this source is less frequently highlighted in standard assessments, it deserves careful evaluation in any synthetic route involving azide chemistry.
5. Nitrite Impurities in Common Reagents
Several commonly used process materials may introduce trace nitrite contamination:
- Sodium nitrate (NaNO₃), frequently employed as an oxidizing agent, may contain trace sodium nitrite impurities
- Sodium chloride used during aqueous workups may contain nitrite contamination depending on supplier and manufacturing origin
- Recycled process water and recovered solvent systems are documented but frequently overlooked nitrite sources
Tertiary Amine Reagents and N-Dealkylation: TEA, DIPEA, and Pyridine as Precursor Sources
Although tertiary amines cannot directly form nitrosamines, they should never be considered chemically inert with respect to nitrosamine risk. Under conditions involving strong acid, elevated temperature, or highly reactive electrophiles, tertiary amines may undergo N-dealkylation reactions that produce secondary amines capable of following conventional nitrosation pathways.
Triethylamine (TEA) and diisopropylethylamine (DIPEA) are among the most frequently used bases in pharmaceutical synthesis, and their degradation behavior under process conditions has been well documented.
- TEA exposed to acidic conditions at elevated temperature may slowly undergo N-deethylation to generate diethylamine, which is a secondary amine
- DIPEA subjected to strongly acidic aqueous workup conditions may generate diisopropylamine, especially during prolonged extraction procedures
- Pyridine, often used as both a solvent and a base, does not generate a classical secondary amine; however, N-nitrosopyridinium species may form under certain reaction conditions
This risk should not be underestimated. Commercial-scale manufacturing commonly involves kilogram-scale quantities of these reagents and solvents. Even a conversion rate as low as 0.1% of TEA to diethylamine can generate sufficient precursor quantities to produce measurable levels of N-nitrosodiethylamine (NDEA) in the presence of nitrite species.
Facing challenges with ultra-trace levels? Achieving Ultra-Low Limit of Quantitation (LOQ) in Nitrosamine Testing
Process Decisions That Increase This Risk
- Acidic aqueous workups performed after reactions using TEA or DIPEA as base
- Distillation of TEA at elevated process temperatures
- Long-term storage of TEA-containing solutions under acidic conditions
- Repeated recycling or reuse of amine-containing mother liquors
Cross-Campaign Contamination and Shared Equipment as a Nitrosamine Formation Pathway
Nitrosamine risk associated with shared manufacturing equipment should not be viewed solely as a contamination issue. In many situations, it represents an active nitrosamine formation pathway. If a reactor previously used for nitrite-containing synthesis is subsequently utilized for an API process involving secondary amines, residual nitrosating species on equipment surfaces may react directly with incoming amine-containing materials, generating nitrosamines in situ.
Mechanistically, this differs from simple carryover contamination because the nitrosamine impurity is newly formed on or near the equipment surface before dissolving into the active batch.
Critical Equipment-Related Risk Scenarios
- Dedicated versus multi-product facilities: Multi-product manufacturing facilities require careful evaluation of campaign sequencing involving nitrite-containing and amine-containing processes. Cleaning validation procedures and swab testing should specifically assess residual nitrite rather than relying solely on conventional cleaning criteria.
- Filter dryers and centrifuges: Porous equipment surfaces, seals, and internal crevices can retain nitrite-containing filtrates from previous production campaigns. Visual inspection alone is insufficient for evaluating cleanliness in these systems.
- Heat exchangers: Nitrite-containing solutions may accumulate in dead legs and internal tubing areas within heat exchangers, creating a frequently overlooked contamination and reaction source.
- Solvent recovery systems: Shared solvent recovery systems may accumulate trace nitrite contamination from earlier batches, leading to contamination of recovered solvents reused in later manufacturing campaigns.
Managing complex high-risk drug classes? Nitrosamine Testing for High-Risk Drug Classes
pH, Temperature, and Reaction Sequence: Process Parameters That Control Nitrosamine Risk
Process parameters are not independent variables in nitrosamine chemistry. They directly govern nitrosamine formation potential. The identical reaction performed at pH 7 versus pH 3.5 may exhibit nitrosamine formation rates differing by two to three orders of magnitude.
pH Control as a Nitrosamine Risk Mitigation Strategy
Maintaining process conditions above pH 6 significantly reduces nitrosation rates because HNO₂ exists primarily as ionized NO₂⁻ under alkaline conditions and therefore becomes substantially less reactive. Whenever process chemistry allows, alkaline environments are strongly preferred in systems where secondary amines and potential nitrosating species may coexist.
Temperature Staging
High-temperature operations involving amide solvents should ideally be completed before introducing reagents capable of generating nitrosating species. Removing the solvent before downstream nitrosating conditions are encountered is often an effective strategy for minimizing risk. In this context, reaction sequence design becomes just as important as the chemistry of the individual reaction steps themselves.
Reaction Sequence Decisions That Create or Reduce Risk
| Risk-Creating Sequence | Risk-Reducing Alternative |
|---|---|
| Diazotization followed by amine coupling in a single pot | Two-pot processing with nitrite quench before amine introduction |
| DMF-based reaction followed by acidic aqueous workup | Replacement with DMSO or THF combined with basic aqueous workup |
| TEA base followed by downstream acidification and nitrite quench | Use of solid-supported base and nitrite neutralization before acidification |
| Reuse of recovered solvent without nitrite analysis | Validation and testing of recovered solvent streams before reuse |
| High-temperature distillation of amine in amide solvent | Lower-temperature operation with molecular sieves to reduce water activity |
Evaluating multiple impurities? View Acceptable Intake for Multiple Nitrosamines
Nitrosamine Formation Pathways in API Synthesis: Analytical Confirmation Strategies
Determining the specific nitrosamine formation pathway active within a process requires pathway-focused analytical investigation rather than generic nitrosamine screening alone. Broad-spectrum screening identifies which nitrosamine impurity is present, whereas mechanistic investigation determines how the impurity was generated.
At ResolveMass Laboratories Inc., nitrosamine root-cause investigations are approached using structure-driven analytical strategy. The identified nitrosamine structure is correlated with plausible amine precursors present within the synthetic route, followed by targeted experimental stress studies that systematically vary pH, temperature, nitrite concentration, solvent composition, and process conditions. This approach provides not only identification of the root cause but also a scientifically defensible process model capable of supporting CAPA implementation and regulatory submissions.
Analytical Techniques Used for Pathway Confirmation
- LC-MS/MS using positive ion electrospray ionization (ESI) for trace-level nitrosamine quantitation in process intermediates and in-process streams
- Headspace GC-MS and GC-TEA for analysis of volatile nitrosamines such as NDMA and NDEA
- Stable isotope dilution (SID) methodologies for accurate sub-ppm quantitation with reduced matrix interference
- Nitrite-specific ion chromatography for mapping nitrite distribution throughout the synthetic process
- Forced degradation studies to confirm solvent hydrolysis pathways as precursor sources for nitrosamine formation
Optimize your testing methodology: Direct Injection vs. Headspace Techniques for Nitrosamines
Conclusion: Chemistry Decisions Are Ultimately Risk Decisions
Every solvent selection, base choice, quench reagent, and process parameter introduced during API synthesis carries an associated nitrosamine risk profile. Nitrosamine formation pathways in API synthesis are mechanistically understandable and therefore preventable when chemistry decisions are evaluated through a nitrosamine-focused risk framework from the earliest stages of process and route development.
Regulatory expectations have evolved far beyond simple batch testing strategies. Process chemists, analytical scientists, manufacturing specialists, and regulatory affairs teams must now work collaboratively to build nitrosamine risk assessments that are predictive, scientifically defensible, and firmly grounded in real process chemistry rather than generic checklist approaches.
Partner with a specialist lab: Check our Guide on Nitrosamine Testing CRO Selection
ResolveMass Laboratories Inc. provides deep expertise in analytical characterization, nitrosamine root-cause investigation, and control strategy development. Whether the challenge involves an unexpected nitrosamine detection, a regulatory inquiry, or a proactive process risk evaluation, the team delivers scientifically rigorous support focused at the chemistry level.
📩 Ready to evaluate nitrosamine risk directly at its source?
Contact ResolveMass Laboratories Inc..
Frequently Asked Questions (FAQs)
The highest nitrosamine risk is typically associated with diazotization reactions involving sodium nitrite, acidic aqueous workups, and process steps where secondary amines are exposed to nitrite-containing environments. Reactions performed in solvents such as DMF or DMA under acidic and high-temperature conditions can further increase susceptibility. Since every synthetic route behaves differently, each manufacturing step should be assessed individually through a detailed process risk evaluation.
DMF can degrade under acidic or high-temperature conditions and release dimethylamine, which is a reactive secondary amine. Once formed, dimethylamine can interact with nitrosating agents present in the process stream to generate NDMA. In such cases, the nitrosamine precursor originates from the solvent itself rather than from the API structure or intermediate.
pH plays a major role in controlling the rate of nitrosamine generation. Nitrosation reactions are most favorable within acidic conditions, particularly around pH 3 to 4, where nitrous acid remains highly reactive. At higher or alkaline pH levels, nitrous acid converts predominantly into nitrite ions, which are significantly less reactive toward secondary amines, thereby reducing nitrosamine formation potential.
Tertiary amines do not directly form nitrosamines under normal reaction conditions, but they should not be considered entirely risk-free. Under acidic environments or elevated temperatures, reagents such as triethylamine (TEA) and DIPEA may undergo partial N-dealkylation, producing secondary amines capable of nitrosamine formation. The extent of this risk depends heavily on process conditions and overall reaction design.
Risk assessment for shared manufacturing equipment should include analytical testing specifically designed to detect residual nitrite contamination. Techniques such as ion chromatography performed on swab samples or rinse solutions are commonly used for this purpose. Particular attention should also be given to hard-to-clean areas including filter dryers, dead legs, heat exchangers, and solvent recovery systems where trace residues may accumulate.
The order in which reactions and processing steps occur determines which reactive species are exposed to one another and under what environmental conditions. Separating nitrosating steps from secondary amine-containing operations can significantly reduce the possibility of nitrosamine generation. Strategies such as intermediate quenching, vessel segregation, and controlled neutralization are often highly effective risk mitigation approaches.
Confirming a nitrosamine formation pathway requires more than standard impurity screening. Laboratories typically perform targeted stress studies in which synthesis conditions such as pH, temperature, solvent composition, and reagent concentration are systematically varied. Advanced analytical techniques including LC-MS/MS, GC-TEA, and ion chromatography are then used to identify nitrosamines and trace the source of nitrite within the process.
Although most reported nitrosamine incidents have involved small molecule pharmaceuticals, biologic manufacturing processes are not completely exempt from risk. Certain cell culture media, excipients, or downstream processing aids may introduce nitrite or amine-containing materials into the manufacturing environment. However, current regulatory focus and the majority of high-risk scenarios remain concentrated within small molecule API synthesis processes.
Reference:
- López Rodríguez, R., McManus, J. A., Murphy, N. S., Ott, M. A., & Burns, M. J. (2020). Pathways for N-nitroso compound formation: Secondary amines and beyond. Organic Process Research & Development, 24(9), 1558–1585. https://doi.org/10.1021/acs.oprd.0c00323
- U.S. Food and Drug Administration. (2024). Control of nitrosamine impurities in human drugs: Guidance for industry (Revision 2). U.S. Department of Health and Human Services. https://www.fda.gov/media/141720/download
- Cioc, R. C., Joyce, C., Mayr, M., & Bream, R. N. (2023). Formation of N-nitrosamine drug substance related impurities in medicines: A regulatory perspective on risk factors and mitigation strategies. Organic Process Research & Development, 27(10), 1736–1750. https://doi.org/10.1021/acs.oprd.3c00153



