Introduction
Understanding What Are Nitrosamines is essential when evaluating the chemical safety, purity, and structural stability of raw materials, water systems, pharmaceutical products, and consumer goods. Since mid-2018, these compounds have become a major global concern after trace-level contamination was unexpectedly identified in several widely prescribed blood pressure medications commonly referred to as “sartans.” This discovery initiated extensive international investigations, revealing that nitrosamine contamination was not an isolated manufacturing issue but part of a broader systemic challenge affecting multiple sectors within the chemical and pharmaceutical supply chain.
Subsequent investigations demonstrated that nitrosamines can develop through numerous subtle and often difficult-to-detect pathways during routine manufacturing operations, environmental exposure, prolonged storage conditions, and even product packaging interactions. These findings significantly reshaped global regulatory expectations and quality assurance practices.
To minimize contamination risks and avoid expensive recalls, import restrictions, or supply interruptions, pharmaceutical manufacturers and analytical laboratories now rely heavily on advanced predictive risk modeling, robust process controls, and highly sensitive mass spectrometric testing methodologies capable of detecting impurities at extremely low concentrations.
Explore our specialized services: Comprehensive Nitrosamine Analysis and Testing
Share via:
Summary of Nitrosamine Dynamics
- Nitrosamines are chemical compounds containing an N-nitroso group and are considered highly toxic because many are mutagenic and carcinogenic. Global concern increased after contamination was discovered in blood pressure medicines (“sartans”) in 2018.
- These compounds can form through multiple pathways during pharmaceutical manufacturing, water treatment, storage, packaging interactions, and environmental exposure, making contamination difficult to detect and control.
- Nitrosamines are produced when amines react with nitrosating agents such as nitrites under acidic, moist, or high-temperature conditions. Even small changes in pH, humidity, or contamination can accelerate their formation.
- In water systems, chloramination processes can create nitrosamines like NDMA from organic nitrogen compounds. In pharmaceuticals, excipients such as starch or cellulose may contain trace nitrites that react with drug ingredients during long-term storage.
- Nitrosamines are broadly classified into:
- Small-molecule nitrosamines (e.g., NDMA, NDEA), which often arise from solvents, raw materials, or cross-contamination.
- NDSRIs (Nitrosamine Drug Substance-Related Impurities), which are structurally linked to the active drug itself and are more complex to analyze.
- These compounds become dangerous after metabolic activation in the body. Liver enzymes convert them into reactive intermediates that can damage DNA, trigger mutations, and potentially initiate cancer development.
- Regulatory agencies such as the FDA and EMA enforce extremely low acceptable intake limits for nitrosamines, often measured in nanograms per day, and use frameworks like CPCA to estimate cancer risk for newer compounds lacking full toxicology data.
- Detecting nitrosamines requires advanced analytical methods such as LC-MS/MS and GC-MS/MS capable of identifying trace-level impurities. Modern mitigation strategies include cleaner manufacturing processes, low-nitrite excipients, antioxidants, active packaging systems, and continuous risk assessment throughout a product’s lifecycle.
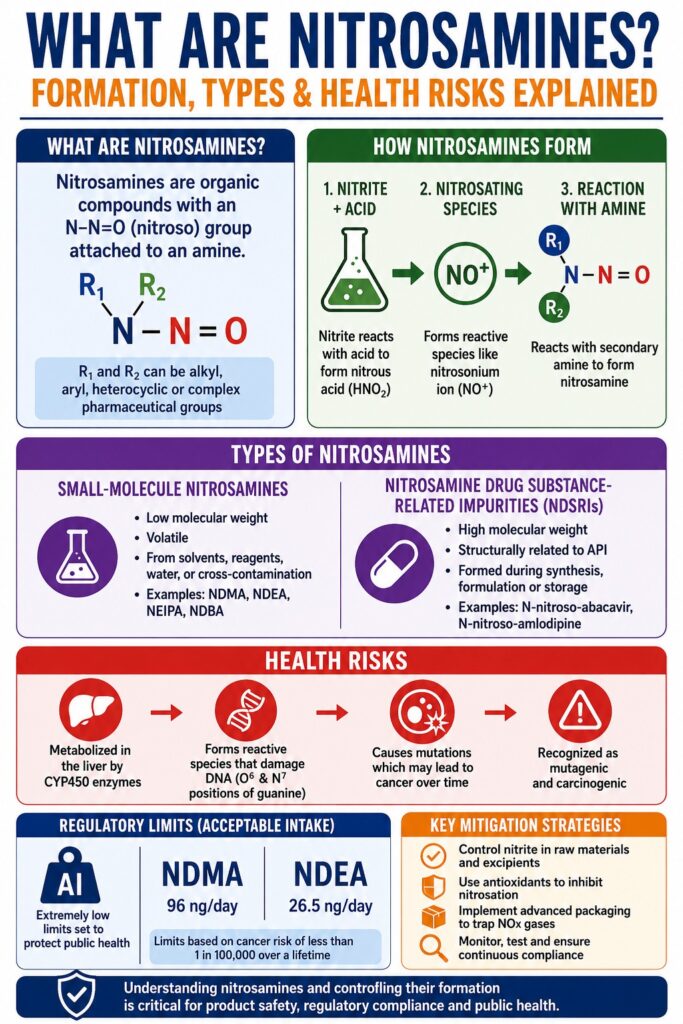
Understanding the Chemical Identity of Nitrosamines
Nitrosamines are a group of organic chemical compounds characterized by the presence of an NNN-nitroso functional group (−N−N=O)(-\text{N}-\text{N}=\text{O})(−N−N=O) directly attached to an organic amine nitrogen atom. Due to their recognized mutagenic and carcinogenic potential, they are classified as high-priority impurities of toxicological concern. Their general molecular structure follows the configuration:R1R2N−N=O\text{R}_1\text{R}_2\text{N}-\text{N}=\text{O}R1R2N−N=O
In this structure, the R1\text{R}_1R1 and R2\text{R}_2R2 substituents may consist of alkyl groups, aryl groups, heterocyclic systems, or even large pharmaceutical molecular fragments.
At the molecular level, the lone electron pair located on the amino nitrogen atom becomes partially delocalized toward the strongly electronegative oxygen atom within the nitroso group. This electron delocalization creates multiple resonance stabilization contributors, ultimately making the oxygen atom the most basic region of the molecule.
For commonly encountered dialkylnitrosamines such as NNN-nitrosodimethylamine (NDMA), the pKa\text{p}K_apKa value associated with the corresponding OOO-protonated form remains below 1. As a result, this protonated structure becomes the dominant species detected through nuclear magnetic resonance (NMR) spectroscopy under acidic conditions.
Under strongly acidic environments, typically within a pH\text{pH}pH range of 1–2, a short-lived and generally unobserved NNN-protonated intermediate may also form in kinetically meaningful amounts. This transient species can initiate protolytic denitrosation, a decomposition pathway in which the nitrosamine reverts back to its parent secondary amine through nucleophilic attack mechanisms.
Certain nucleophilic substances, including bromide ions, thiocyanate, and thiourea, can accelerate this acid-mediated decomposition process. These molecular behaviors demonstrate that although nitrosamines may remain relatively stable under neutral storage conditions, they become highly sensitive to localized changes in pH\text{pH}pH, trace catalytic contaminants, moisture accumulation, and microenvironmental chemical variations.
Learn more about high-risk products: Nitrosamine Testing for High-Risk Drug Classes
Core Chemical Formation Mechanisms and Precursor Kinetics
Nitrosamine formation generally occurs through a nucleophilic substitution reaction involving an amine precursor and a reactive nitrosating species. The amine precursor may be secondary, tertiary, or quaternary in nature, while the nitrosating agent is typically generated under acidic conditions, elevated temperatures, or fluctuating moisture environments.
The fundamental mechanism requires the conversion of relatively stable nitrogen-containing compounds into highly electrophilic nitrosating intermediates.
Nitrous acid (HNO2)(\text{HNO}_2)(HNO2), although chemically unstable in isolation, forms readily in situ when inorganic nitrite salts (NO2−)(\text{NO}_2^-)(NO2−) encounter mildly acidic conditions. Once protonated, nitrous acid undergoes dehydration to produce the highly reactive nitrosonium ion (NO+)(\text{NO}^+)(NO+) or decomposes into dinitrogen trioxide (N2O3)(\text{N}_2\text{O}_3)(N2O3), both of which function as major nitrosating species.
When a secondary amine interacts with these electrophilic nitrogen oxides, the lone electron pair on the amine nitrogen attacks the nitroso center. Rapid deprotonation subsequently occurs, producing a stable dialkylnitrosamine.
Tertiary and quaternary amines may also participate in nitrosation reactions through more complicated oxidative cleavage or dealkylation pathways, particularly when exposed to elevated processing temperatures or localized pH\text{pH}pH fluctuations.
Beyond classical laboratory reactions, several distinct nitrosamine formation pathways exist across environmental systems and pharmaceutical manufacturing environments.
Technical insight: Direct Injection vs. Headspace Techniques for Nitrosamines
Aqueous Disinfection Mechanisms
Within municipal water treatment facilities and public water distribution systems, nitrosamines may form as nitrogenous disinfection byproducts (N-DBPs) during chloramination processes.
When monochloramine (NH2Cl)(\text{NH}_2\text{Cl})(NH2Cl) and dichloramine (NHCl2)(\text{NHCl}_2)(NHCl2) coexist under standard treatment conditions, they react with organic nitrogen-containing precursors originating from agricultural runoff, industrial waste streams, or naturally occurring biological material.
Secondary amines, including dimethylamine, undergo nucleophilic substitution reactions with dichloramine to generate chlorinated unsymmetrical dimethylhydrazine (Cl-UDMH) intermediates. Dissolved oxygen then gradually oxidizes these intermediates over several days, ultimately yielding NDMA.
Because this oxidative conversion occurs slowly, nitrosamines may continue forming even after treated water exits the treatment facility and travels through municipal pipeline networks. This delayed formation mechanism explains why nitrosamine concentrations can increase over time during water distribution.
Solid-State Excipient Microenvironments
Nitrosation reactions may also occur within solid pharmaceutical dosage forms such as tablets and capsules during long-term storage.
Even when active pharmaceutical ingredients (APIs) possess extremely high purity, commonly used excipients including microcrystalline cellulose (MCC), starches, and magnesium stearate frequently contain residual nitrite contamination. These trace nitrite concentrations generally range from 200020002000 to 3000 ppb3000\text{ ppb}3000 ppb (2 to 3 ppm)(2\text{ to }3\text{ ppm})(2 to 3 ppm), originating from raw material synthesis, processing water contamination, or microbial metabolic activity.
When these excipients are combined with APIs containing amine functionalities, ambient moisture can facilitate localized dissolution within the tablet matrix. As storage temperatures exceed 40∘C40^\circ\text{C}40∘C and humidity levels fluctuate, reactive species become increasingly mobile.
Furthermore, binder systems such as povidone (PVP) and hydroxypropyl methylcellulose (HPMC) may create localized microenvironmental pH\text{pH}pH shifts that accelerate solid-state nitrosation pathways. Over extended storage periods, these reactions can gradually increase nitrosamine concentrations throughout the product shelf life.
Mitigation strategy: Using Secondary Amine Scavengers to Prevent Nitrosamines
Advanced Structural Classification: Deciphering What Are Nitrosamines
Nitrosamine impurities are generally categorized into two major structural groups based on molecular weight, chemical complexity, and formation origin:
- Small-molecule nitrosamines
- Nitrosamine Drug Substance-Related Impurities (NDSRIs)
Understanding these classifications is essential for identifying contamination sources, designing analytical methods, and assessing toxicological risk.
Small-Molecule Nitrosamines
Small-molecule nitrosamines are relatively low-molecular-weight volatile compounds that do not structurally resemble the active pharmaceutical ingredient itself.
These impurities commonly enter pharmaceutical products through environmental contamination, solvent degradation, manufacturing side reactions, or equipment cross-contamination. For instance, dimethylformamide (DMF), a frequently used processing solvent, may degrade into dimethylamine. This amine can subsequently react with trace nitrites to form NDMA.
Other commonly monitored small-molecule nitrosamines include:
- NNN-nitrosodiethylamine (NDEA)
- NNN-nitrosodiisopropylamine (NDIPA)
- NNN-nitrosoethylisopropylamine (NEIPA)
- NNN-nitrosodibutylamine (NDBA)
Because these compounds possess relatively simple molecular architectures, they dissolve readily in polar solvents, exhibit substantial volatility, and can easily migrate between manufacturing systems if cleaning validation procedures are insufficient.
Nitrosamine Drug Substance-Related Impurities (NDSRIs)
Nitrosamine Drug Substance-Related Impurities (NDSRIs) are structurally complex, higher-molecular-weight compounds directly related to the active pharmaceutical ingredient.
NDSRIs form when the API itself contains vulnerable secondary or tertiary amine sites capable of undergoing nitrosation during synthesis, formulation processing, or extended storage.
Examples include:
- NNN-nitroso-abacavir
- NNN-nitroso-amlodipine
- NNN-nitroso-fluoxetine
Unlike small-molecule nitrosamines, NDSRIs are generally non-volatile and exhibit physicochemical behaviors closely resembling the parent drug molecule. Their detection and toxicological assessment require highly customized analytical strategies because compound-specific long-term safety data are often unavailable.
| Nitrosamine Generic Name | Structural Subcategory | Primary Source or Mechanism | Documented Drug Association Examples |
| N-nitrosmdimethylamine (NDMA) | Small-Molecule | Reused solvents, chloraminated water, DMF degradation | Valsartan, Metformin, Ranitidine |
| N-nitrosdiethylamine (NDEA) | Small-Molecule | Contaminated raw reagents, cross-contamination | Losartan, Irbesartan, Nizatidine |
| N-nitrosoethylisopropylamine (NEIPA) | Small-Molecule | Quaternary amine reactions, raw materials | Early sartan-class blockages |
| N-nitrosodibutylamine (NDBA) | Small-Molecule | Packaging leaching, industrial rubber items | Container-closure extractables |
| N-nitroso-abacavir | NDSRI | API secondary amine site solid-state nitrosation | Abacavir antiretroviral treatments |
| N-nitroso-fluoxetine | NDSRI | Interaction with residual excipient nitrites | Fluoxetine antidepressant formulations |
| N-nitroso-amlodipine | NDSRI | Amine function alteration under thermal stress | Amlodipine cardiovascular options |
Regulatory update: Impact of ICH M7(R2) Updates on Nitrosamine Risk Assessment
Molecular Characteristics and Evolving Understandings of What Are Nitrosamines
The structural differences between small-molecule nitrosamines and NDSRIs significantly influence their chemical behavior, toxicological profile, and regulatory classification.
Small-molecule nitrosamines typically display relatively predictable metabolic activation pathways and toxicological characteristics across different matrices. In contrast, NDSRIs possess structurally diverse frameworks that mirror the complexity of their corresponding APIs. These variations affect their solubility, stability, tissue distribution, and metabolic transformation pathways within the human body.
As a result, predictive computational modeling and structure-based carcinogenicity assessment tools have become increasingly important for evaluating NDSRI-related risk.
Recent investigations have also expanded contamination concerns beyond APIs and excipients to include packaging-related leachables. Vulcanization accelerators used in rubber manufacturing and plasticizers incorporated into liquid containers may release reactive nitrites or small nitrosamine compounds into pharmaceutical products such as eye drops and oral liquid formulations.
Consequently, modern nitrosamine risk assessments must evaluate the complete product lifecycle, including:
- Raw material sourcing
- API synthesis
- Formulation design
- Packaging compatibility
- Long-term storage stability
Toxicological Mechanisms and Metabolic Carcinogenicity Pathways
Nitrosamines present substantial health concerns because many function as highly potent mutagenic carcinogens capable of chemically altering DNA.
Importantly, nitrosamines are not inherently reactive in their native state. Instead, they require metabolic activation within the body before exerting genotoxic effects.
After ingestion, nitrosamines undergo hepatic metabolism primarily through Cytochrome P450 enzymes, especially the CYP2E1 isoform. These enzymes catalyze α\alphaα-hydroxylation reactions, introducing a hydroxyl group onto the carbon atom adjacent to the NNN-nitroso moiety.
The resulting α\alphaα-hydroxylated intermediate is highly unstable and rapidly undergoes spontaneous dealkylation, producing aldehydes and transient primary nitrosamine species. These unstable compounds subsequently decompose to generate highly electrophilic diazonium ions.
The diazonium ion attacks nucleophilic sites on DNA bases, particularly the O6O^6O6 and N7N^7N7 positions of guanine. This alkylation disrupts normal DNA geometry and causes base-pairing errors such as G-A\text{G-A}G-A transition mutations during replication.
If these mutations evade cellular DNA repair systems, they may deactivate tumor suppressor genes or activate oncogenes, thereby initiating carcinogenesis.
In addition to direct DNA damage, nitrosamine metabolism also generates reactive oxygen species (ROS), contributing to oxidative stress and widespread cellular injury involving lipids, proteins, and nucleic acids.
Emerging mammalian studies further indicate that gut microbiota may metabolize certain nitrosamines into secondary genotoxic compounds. For example, butyl-hydroxybutylnitrosamine (BBN) may convert into butyl-carboxypropylnitrosamine (BCPN), which can accumulate within distal tissues such as the bladder wall and induce localized tissue damage.
Regulatory Compliance Thresholds and the CPCA Framework
Due to the severe genotoxic risks associated with nitrosamines, global health authorities including the U.S. Food and Drug Administration and the European Medicines Agency enforce extremely strict Acceptable Intake (AI) limits.
These thresholds are established using compound-specific carcinogenicity data or predictive structural assessment tools such as the Carcinogenic Potency Categorization Approach (CPCA).
The AI limit represents the maximum daily exposure expected to produce less than a 1 in 100,000 increase in lifetime cancer risk when consumed daily over 70 years.
For well-characterized small-molecule nitrosamines, regulators have established highly specific intake thresholds. Examples include:
- NDMA: 96 ng/day96\text{ ng/day}96 ng/day
- NDEA: 26.5 ng/day26.5\text{ ng/day}26.5 ng/day
NDEA receives a significantly lower threshold because of its greater carcinogenic potency.
To address NDSRIs lacking direct toxicological datasets, regulators developed the CPCA framework. This predictive system evaluates molecular structure, electronic configuration, and activation potential to classify compounds into five carcinogenic potency categories.
CPCA Potency Categories
| Potency Category | Acceptable Intake Limit | Key Structural Characteristics |
|---|---|---|
| Category 1 | 26.5 ng/day26.5\text{ ng/day}26.5 ng/day | Strong activating features and highly reactive α\alphaα-hydrogen configurations |
| Category 2 | 100 ng/day100\text{ ng/day}100 ng/day | Moderate activation characteristics with limited shielding |
| Category 3 | 400 ng/day400\text{ ng/day}400 ng/day | Restricted activation potential or cyclic stabilization |
| Category 4 | 1500 ng/day1500\text{ ng/day}1500 ng/day | Bulky substituents reducing metabolic activation |
| Category 5 | 1500 ng/day1500\text{ ng/day}1500 ng/day | Strong deactivating features such as tertiary carbons or carboxylic acids |
Regulatory agencies may also issue temporary interim AI limits during manufacturing transition periods. Examples include:
- 3000 ng/day3000\text{ ng/day}3000 ng/day for 1-methyl-4-nitrosopiperazine (MNP) in rifampin
- 12,000 ng/day12,000\text{ ng/day}12,000 ng/day for NNN-nitroso-ciprofloxacin
However, manufacturers remain obligated to complete comprehensive risk assessments and implement permanent corrective actions within established regulatory timelines.
Deep dive into limits: Nitrosamine AI Limits and the CPCA Approach
State-of-the-Art Analytical Strategies for Trace Quantification
Detecting nitrosamines at parts-per-billion (ppb) and parts-per-trillion (ppt) concentrations requires highly advanced chromatographic and mass spectrometric methodologies.
Because these impurities exist at extremely low concentrations within chemically complex matrices, analytical laboratories must utilize specialized extraction procedures, matrix cleanup techniques, and selective ion-monitoring workflows.
Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS)
LC-MS/MS remains the gold-standard analytical platform for nitrosamine detection, particularly when analyzing polar, non-volatile, or thermally unstable compounds such as NDSRIs.
Modern workflows frequently combine:
- Solid-Phase Extraction (SPE)
- Hydrophilic Interaction Chromatography (HILIC)
These cleanup methods enrich target analytes while minimizing interference from complex pharmaceutical matrices.
Mass spectrometers typically operate in Multiple Reaction Monitoring (MRM) mode using positive Atmospheric Pressure Chemical Ionization (APCI) or Electrospray Ionization (ESI) sources.
This approach enables highly selective monitoring of characteristic fragmentation transitions, including loss of the nitroso functional group, allowing precise quantification at ppt-level sensitivity.
Gas Chromatography-Mass Spectrometry (GC-MS/MS)
For highly volatile nitrosamines such as NDMA, NDEA, and NDIPA, GC-MS/MS provides exceptional analytical reliability.
Laboratories commonly utilize Electron Ionization (EI) sources operating at 70 eV70\text{ eV}70 eV together with ultra-inert capillary columns such as the Agilent DB-WAX UI configuration:30 m×0.25 mm,0.25\mum or 0.5\mum30\text{ m} \times 0.25\text{ mm}, 0.25\text{\mu m} \text{ or } 0.5\text{\mu m}30 m×0.25 mm,0.25\mum or 0.5\mum
Both headspace injection and direct liquid injection workflows enable simultaneous multi-analyte screening within rapid analytical runtimes of approximately 16 minutes.
Ultra-inert flow paths minimize thermal degradation within the injector system, ensuring reproducible peak symmetry and reliable trace-level quantification.
To achieve regulatory acceptance, these analytical methods must undergo rigorous validation according to USP and ICH Q2(R2) requirements.
Typical Validation Requirements
| Validation Parameter | Acceptance Criteria |
|---|---|
| Linear Range | 50%50\%50% to 150%150\%150% of target AI concentration |
| Correlation Coefficient | r2≥0.99r^2 \ge 0.99r2≥0.99 |
| Accuracy | 70%70\%70% to 130%130\%130% recovery |
| System Repeatability | %RSD<25%\%\text{RSD} < 25\%%RSD<25% |
| Intermediate Precision | %RSD<30%\%\text{RSD} < 30\%%RSD<30% |
| Limit of Quantitation | ≤30%\le 30\%≤30% of AI limit |
| Signal-to-Noise Ratio | S/N≥10S/N \ge 10S/N≥10 |
Methodology expertise: GC-MS Method Development for Nitrosamine Testing
Comprehensive Risk Mitigation and Formulation Strategies
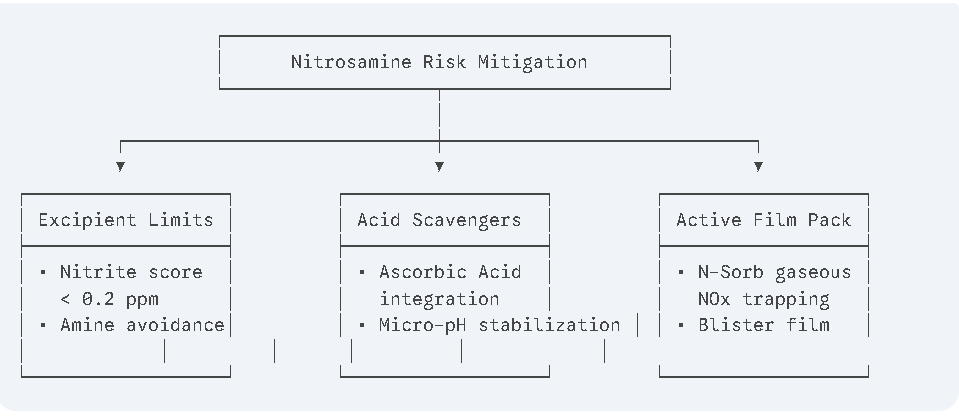
Effective nitrosamine mitigation requires proactive process design rather than relying exclusively on final-product testing.
Modern mitigation strategies involve:
- Synthetic route optimization
- Excipient control
- Antioxidant scavenging
- Packaging engineering
- Environmental monitoring
Excipient Control
Manufacturers increasingly source excipients with certified nitrite levels below 0.2 ppm0.2\text{ ppm}0.2 ppm. Avoiding unnecessary amine-containing stabilizers and maintaining controlled microenvironmental pH\text{pH}pH conditions further reduces nitrosation potential.
Antioxidant Integration
Incorporating antioxidants such as:
- Ascorbic acid (Vitamin C)
- α\alphaα-tocopherol (Vitamin E)
can suppress nitrosation by rapidly reacting with nitrosating species before they interact with susceptible amine groups.
These antioxidants convert reactive nitrous acid into relatively unreactive nitric oxide, thereby interrupting nitrosamine formation pathways.
Active Packaging Technologies
Advanced packaging systems now incorporate scavenging materials such as N-Sorb Activ-Polymer films directly into blister packs.
These active materials trap volatile nitrogen oxide gases released from excipients or packaging components during storage. By removing gaseous nitrosating species from the sealed environment, manufacturers can significantly reduce long-term nitrosamine formation throughout product shelf life.
Analytical precision: Ultra-Low Limit of Quantitation (LOQ) in Nitrosamine Testing
Conclusion: The Future Landscape of Impurity Control
Achieving sustainable compliance regarding What Are Nitrosamines requires a continuous, science-driven strategy that combines advanced analytical technologies with evolving regulatory standards.
As understanding of solid-state chemistry, packaging interactions, and excipient microenvironments continues to improve, pharmaceutical manufacturers are increasingly capable of identifying contamination risks during early-stage product development rather than after market release.
At the same time, regulatory authorities continue refining acceptable exposure limits using predictive structural modeling and expanded carcinogenicity frameworks. Consequently, highly sensitive trace-level analytical testing remains essential for ensuring pharmaceutical quality, maintaining regulatory compliance, and protecting public health.
Assess your risk: Calculate Less-Than-Lifetime (LTL) Exposure for Nitrosamines
Organizations seeking technical guidance for nitrosamine risk assessment, validated analytical workflows, or regulatory compliance strategies can access specialized support through ResolveMass Laboratories.
Frequently Asked Questions
Nitrosamine impurities are unwanted chemical compounds that may appear in medications at extremely low concentrations. These substances contain an N-nitroso functional group and are categorized as probable human carcinogens based on toxicological and structural evidence. They can develop unintentionally during drug manufacturing, packaging, or long-term storage, which is why regulatory agencies closely monitor their levels in pharmaceutical products.
Nitrosamines typically form when amine-containing compounds react with nitrosating agents such as nitrous acid or nitrogen oxides. These reactions are more likely to occur under acidic conditions, elevated temperatures, or high-moisture environments during manufacturing or storage. Residual nitrites in excipients, contaminated solvents, or degraded raw materials can also contribute to nitrosamine generation.
Small-molecule nitrosamines are relatively simple, low-molecular-weight compounds such as NDMA and NDEA that generally arise from solvents, environmental contamination, or process-related reactions. In contrast, Nitrosamine Drug Substance-Related Impurities (NDSRIs) are structurally linked to the active pharmaceutical ingredient itself. NDSRIs form when the drug molecule undergoes nitrosation, making them more chemically complex and difficult to evaluate.
Excipients may contribute to nitrosamine formation because some contain trace amounts of residual nitrites introduced during raw material production or environmental exposure. Under humid or high-temperature storage conditions, these nitrites can dissolve and react with amine-containing APIs inside the dosage form. This interaction may gradually promote solid-state nitrosation throughout the product’s shelf life.
The Carcinogenic Potency Categorization Approach, commonly called CPCA, is a predictive scientific framework used to estimate the cancer-causing potential of nitrosamines that lack direct toxicological data. It analyzes structural characteristics such as α\alphaα-hydrogen arrangements and electron-withdrawing groups to classify compounds into different potency categories. These categories help regulators establish appropriate acceptable intake limits for pharmaceutical safety evaluations.
The rise in nitrosamine-related recalls is largely linked to improvements in analytical testing technologies and stricter global regulatory oversight. Modern mass spectrometry instruments can now identify impurities at significantly lower concentrations than older detection systems. As testing sensitivity has advanced, manufacturers and health authorities have been able to uncover previously undetectable contamination across pharmaceutical supply chains.
Patients should never discontinue prescribed medication without first consulting a qualified healthcare professional. In many situations, the immediate risks associated with untreated medical conditions are greater than the long-term risks posed by trace nitrosamine exposure. Regulatory exposure limits are based on continuous daily intake over several decades, meaning short-term exposure generally does not represent an immediate health emergency.
Reference:
- U.S. Food and Drug Administration (FDA). (2023). Recommended acceptable intake limits for nitrosamine drug substance-related impurities (NDSRIs).https://www.fda.gov/regulatory-information/search-fda-guidance-documents/recommended-acceptable-intake-limits-nitrosamine-drug-substance-related-impurities
- U.S. Food and Drug Administration (FDA). (2024). Control of nitrosamine impurities in human drugs: Guidance for industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/control-nitrosamine-impurities-human-drugs
- U.S. Food and Drug Administration (FDA). (2024). Determining recommended acceptable intake limits for N-nitrosamine impurities in pharmaceuticals: Development and application of the carcinogenic potency categorization approach. https://www.fda.gov/drugs/spotlight-cder-science/determining-recommended-acceptable-intake-limits-n-nitrosamine-impurities-pharmaceuticals
- European Medicines Agency (EMA). (n.d.). Nitrosamine impurities. European Medicines Agency. https://www.ema.europa.eu/en/human-regulatory-overview/post-authorisation/referral-procedures-human-medicines/nitrosamine-impurities
- U.S. Food and Drug Administration (FDA). (2023). Recommended acceptable intake limits for nitrosamine drug substance-related impurities (NDSRIs). U.S. Department of Health and Human Services. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/recommended-acceptable-intake-limits-nitrosamine-drug-substance-related-impurities



