
INTRODUCTION
Extractables and Leachables (E&L) Requirements for U.S. Market Authorization are central to ensuring that materials contacting drug products do not release harmful chemicals. These E&L requirements are now among the most scrutinized sections of regulatory submissions, and sponsors must understand them clearly to avoid FDA delays.
This article provides a comprehensive, expert-level guide on how sponsors can meet Extractables and Leachables (E&L) Requirements for U.S. Market Authorization, integrating the latest scientific expectations and risk-based strategies.
SUMMARY
- U.S. FDA requires a rigorous, risk-based framework for Extractables and Leachables (E&L) studies to ensure patient safety.
- Sponsors must demonstrate that container-closure systems, medical devices, packaging materials, and manufacturing components do not release harmful chemical migrants.
- Early planning of E&L requirements for U.S. market authorization reduces development delays and deficiency letters.
- The ICH Q3E guideline strengthens expectations for extractables study design, leachables thresholds, toxicological evaluation, and risk-based approaches.
- A compliant E&L program includes material characterization, exhaustive extraction, simulated use studies, toxicology assessment, and clear justification of analytical sufficiency.
- FDA expects scientifically defensible, well-documented E&L data supported by validated methodologies.
Get in Touch with Us
What Are Extractables and Leachables (E&L) Requirements for U.S. Market Authorization?
Extractables and Leachables (E&L) Requirements for U.S. Market Authorization ensure that any material used in packaging, delivery, or manufacturing does not compromise patient safety through chemical migration. These requirements apply to all contact materials involved in storing, transporting, or administering the drug.
Key components of U.S. E&L expectations
- Characterization of all contact materials
- Determination of potential chemical migrants (extractables)
- Identification and quantification of actual migrants (leachables)
- Justification of toxicological safety
- Demonstration of compatibility between drug product and materials
1. Why E&L Requirements Matter for FDA Approval
Extractables and Leachables (E&L) Requirements for U.S. Market Authorization matter because inadequate E&L data is one of the top causes of FDA deficiency letters. FDA reviewers rely on these studies to confirm long-term product safety.
FDA’s main concerns
- Impurities that exceed permitted exposure limits
- Unknown compounds in E&L reports
- Incomplete toxicological risk assessment
- Poorly designed studies with limited analytical coverage
- Lack of justification for extraction conditions
2. Scope of Extractables and Leachables (E&L) Requirements for U.S. Market Authorization
The scope of these requirements covers all components that directly, indirectly, or intermittently contact the drug product.
Link for Extractables and Leachables (E&L) testing : https://resolvemass.ca/el-testing-usa-vs-europe/
Applies to:
- Primary and secondary packaging
- Container-closure systems
- Drug-device combination products
- Manufacturing single-use systems
- Tubings, filters, seals, stoppers, syringes
- Inhalation, ophthalmic, parenteral, and transdermal systems
FDA focuses heavily on:
- Elastomers
- Polymers
- Adhesives
- Coatings
- Metals
3. Key Elements of a U.S.-Compliant E&L Study
A compliant program must address all scientific and regulatory expectations embedded in Extractables and Leachables (E&L) Requirements for U.S. Market Authorization.
Upfront Requirements
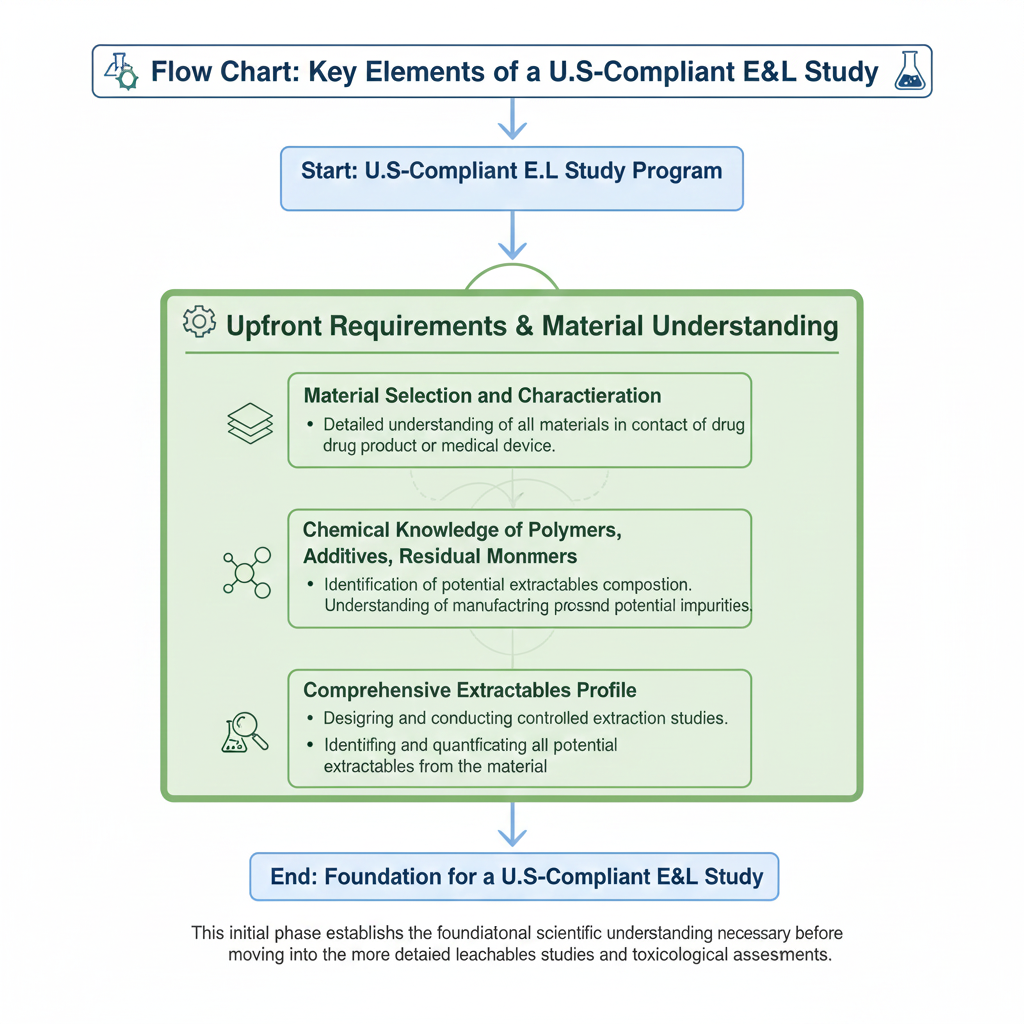
Extractables Study Requirements
Extractables studies must simulate the worst-case scenario to identify all potential chemical migrants.
Typical extraction conditions
| Extraction Type | Purpose | Common Solvents |
|---|---|---|
| Exhaustive Extraction | Maximum migration potential | Water, IPA, hexane |
| Simulated Use | Drug-specific environments | pH buffers, formulation simulants |
| Elevated Temperature/Time | Stress testing | Accelerated conditions |
Leachables Study Requirements
Leachables studies are required to show what actually migrates into the drug product during real-time storage.
FDA expectations for leachables
- Leachables monitored through entire shelf life
- Identification of unknowns above thresholds
- Quantitation using validated methods
- Control strategy for all confirmed toxicological risks
4. Toxicological Risk Assessment Requirements
Toxicology evaluation is essential to meeting Extractables and Leachables (E&L) Requirements for U.S. Market Authorization.
Primary toxicology expectations
- Identify each compound above the analytical evaluation threshold
- Compare exposure against permitted daily exposure (PDE)
- Assess mutagenicity, carcinogenicity, and systemic toxicity
- Provide NOAEL-based calculations
- Justify safety margins for vulnerable populations
5. How ICH Q3E Influences U.S. E&L Requirements
ICH Q3E directly informs the Extractables and Leachables (E&L) Requirements for U.S. Market Authorization by standardizing approach, thresholds, and risk-based decision-making.
Learn more about ICH Q3 for Extractables and Leachables (E&L):
Q3E enhances:

6. Analytical Methods Accepted by the FDA
FDA expects orthogonal, sensitive, validated methods to meet Extractables and Leachables (E&L) Requirements for U.S. Market Authorization.
Typical Analytical Toolkit
- GC-MS for volatile/semi-volatile extractables
- LC-MS for non-volatile organics
- ICP-MS for elemental impurities
- FTIR & NMR for structural confirmation
- HRMS for unknown identification
7. Common Pitfalls That Lead to FDA Delays
Many FDA delays occur because sponsors underestimate the complexity of Extractables and Leachables (E&L) Requirements for U.S. Market Authorization.
Most frequent issues
- Conducting extractables studies without knowing formulation
- Insufficient leachables time-points
- Limited mass spectrometry sensitivity
- Failing to justify worst-case conditions
- Missing toxicological justifications
8. Best Practices for Meeting E&L Requirements
To successfully meet Extractables and Leachables (E&L) Requirements for U.S. Market Authorization, sponsors should apply proven best practices.
Best Practice Checklist
- Start E&L planning early (pre-clinical stage)
- Characterize every component thoroughly
- Use extraction conditions that mimic actual use
- Apply orthogonal analytical platforms
- Document complete identification workflows
- Align toxicology justification with FDA expectations
- Maintain high data integrity and traceability
Conclusion
Meeting Extractables and Leachables (E&L) Requirements for U.S. Market Authorization is no longer optional—it is central to FDA’s safety evaluation framework. Sponsors who design strong extractables profiles, accurate leachables monitoring, and detailed toxicology justifications will accelerate approval timelines and reduce regulatory risk. The organizations that adopt high-integrity, science-driven E&L programs are the most likely to meet Extractables and Leachables (E&L) Requirements for U.S. Market Authorization successfully.
Get in Touch with Us
FAQs on Extractables and Leachables (E&L) Requirements for U.S. Market Authorization
U.S. E&L requirements stand out because the FDA expects a significantly higher level of toxicological justification, analytical rigor, and data transparency compared to many other markets. While Europe relies heavily on MDR principles and device-focused risk evaluation, the U.S. regulatory framework emphasizes the following:
Key U.S.-Specific Features
-Full identification of every unknown compound above AET
FDA rarely accepts “unknowns” unless justified with extensive analytical attempts using GC-MS, LC-MS, HRMS, NMR, and FTIR.
-Toxicology-first evaluation
The U.S. system heavily integrates toxicological thresholds such as PDE, TTC, SCT, and MOE—often requiring a board-certified toxicologist’s justification.
-Study design justification
~FDA reviewers routinely request documentation explaining:
~Why certain solvents were used
~Why extraction conditions represent a worst-case scenario
~How analytical methods were selected
~How mass spectrometry sensitivity aligns with the drug’s risk
-Alignment with ICH Q3E
ICH Q3E is now harmonizing expectations globally, but the FDA tends to interpret it in the strictest, most comprehensive manner.
Because of these factors, U.S. E&L submissions are generally more detailed, more exhaustive, and more thoroughly scrutinized than many global counterparts.
Sponsors should initiate E&L planning during the formulation development phase, not after packaging components have been chosen. Early E&L strategy prevents complications, redesigns, and regulatory delays.
Why Early Planning Is Critical
-Material choice influences E&L risk
Polymers, elastomers, adhesives, and coatings all vary drastically in extractables potential.
-Late-stage E&L issues can delay approval by 6–12 months
FDA deficiency letters frequently result from inadequate E&L packages.
-Biologics and gene therapies require even earlier planning
Single-use systems (bags, tubing, filters) contribute major extractables in biologics manufacturing, and this must be characterized early.
-Analytical method development takes time
Establishing GC-MS, LC-MS, ICP-MS workflows and validating sensitivity requires weeks or months.
In short: E&L planning must not wait until packaging selection. A proactive approach saves time, reduces cost, and ensures regulator-ready data.
Any product where materials contact the drug product requires E&L evaluation, but certain product categories fall under high-risk routes where FDA expects near-zero tolerance for unidentified leachables.
Products Under High Scrutiny
-Parenteral medications (IV, IM, SC injections)
Direct systemic exposure means even low-level leachables can cause toxicity.
-Ophthalmic products (eye drops, ointments, surgical lubricants)
The eye’s sensitivity requires strict E&L control.
-Inhalation products (MDIs, DPIs, nebulizers)
Inhalation bypasses natural metabolic barriers.
-Transdermal systems (patches, microneedles)
Adhesives, membranes, and backing layers can release extractables.
-Combination products
Syringes, auto-injectors, pumps, cartridges, and tubing expose the drug to multiple materials.
Also required for
-Prefilled syringes
-Infusion sets
-PVC and TPE-based tubings
-Manufacturing single-use systems (bags, filters, connectors)
If the material touches the drug—even indirectly—FDA requires E&L justification.
Materials with complex chemical compositions, high additive content, or reactive components tend to generate more extractables.
Highest-Risk Material Groups
-Elastomers
Stoppers, plungers, seals; contain curing agents, antioxidants, accelerators, nitrosamines.
-Polymers
PP, PE, PVC, PETG, TPEs—variable formulations and additives lead to diverse extractables.
-Adhesives
Acrylic, silicone, and rubber-based adhesives used in labels, patches, and components.
-Coatings & lubricants
Fluoropolymer coatings, silicone oils, molybdenum disulfide.
-Tubing systems
PVC (plasticizers), silicone (oligomers), Tygon® blends.
-Multilayer packaging films
EVOH, PVDC, aluminum laminates, polyamides.
Each material contains dozens of potential extractables—plasticizers, antioxidants, pigments, oligomers, slip agents, and monomers.
FDA expects leachables studies to span the full real-time shelf life of the drug product. Accelerated studies can supplement, but cannot replace real-time data.
FDA Expectations
-Full duration
If shelf life is 24 months, leachables must be monitored up to 24 months.
-Multiple time points
Early, mid, and end-of-life points are required for trend analysis.
-Simulated-use conditions
Accepted but must not substitute real-time data.
-Worst-case batches
Highest contact-surface-area packaging units must be evaluated.
For high-risk routes (parenteral, ophthalmic, inhalation), FDA is especially strict about real-time data completeness.
The #1 reason is inadequate toxicological justification. FDA prioritizes patient safety over analytical sophistication.
Top Rejection Causes
-Insufficient toxicology analysis
Missing PDE justification, unsupported TTC application, or lack of genotoxicity evaluation.
-Unidentified compounds above AET
Unknowns create unresolved safety risks.
-Inadequate leachables timepoints
FDA does not accept incomplete shelf-life data.
-Poor analytical sensitivity
Inability to detect key classes of compounds (semi-volatiles, metals, oligomers).
-Unjustified extraction conditions
FDA expects scientifically rigorous worst-case design—not arbitrary parameters.
Sponsors must submit a scientifically defensible, complete data package to avoid delays in market authorization.
Get in Touch with Us
Reference
- United States Pharmacopeial Convention. (n.d.). Extractables and leachables. Retrieved October 10, 2025, from https://www.usp.org/impurities/extractables-and-leachables
- Balfour, H. (2022, April 29). Advancing extractables and leachables testing. European Pharmaceutical Review. https://www.europeanpharmaceuticalreview.com/article/170814/advancing-extractables-and-leachables-testing/
- Rozio, M. G. (2025). Correcting detection and quantitation bias in extractables and leachables testing. Journal of Pharmaceutical Analysis, 15(2), 123–134. https://doi.org/10.1016/j.jpha.2025.04.004
- Balfour, H. (2022, April 29). Advancing extractables and leachables testing. European Pharmaceutical Review. https://www.europeanpharmaceuticalreview.com/article/170814/advancing-extractables-and-leachables-testing/

