
Introduction:
FDA Peptide Sameness Study Requirements define the scientific and regulatory framework needed to demonstrate that a generic synthetic peptide is structurally identical and therapeutically equivalent to its reference listed drug (RLD). For manufacturers pursuing peptide generics in the U.S. and Canada, understanding the regulatory expectations of the U.S. Food and Drug Administration and Health Canada is essential to achieving successful approval.
At ResolveMass Laboratories Inc., we specialize in high-resolution mass spectrometry–driven peptide characterization in drug development designed specifically to meet FDA Peptide Sameness Study Requirements and Health Canada comparability expectations. Our work aligns with regulatory expectations outlined in FDA requirements for peptide characterization and global ICH standards.
Summary:
- FDA Peptide Sameness Study Requirements focus on proving structural identity, impurity equivalence, and analytical comparability under a generic drug pathway (505(j) or 505(b)(2)).
- Synthetic peptides are regulated as drugs, not biologics, when chemically synthesized and well-characterized.
- Health Canada follows similar principles, requiring robust structural confirmation and impurity profiling for ANDS submissions.
- Analytical characterization must include LC-MS/MS, HRMS, peptide mapping, impurity profiling, and stability studies.
- Bioequivalence alone is not sufficient without comprehensive analytical sameness data.
- A scientifically defensible peptide sameness package reduces regulatory risk and accelerates approval timelines.
For sponsors filing in North America, our peptide sameness study services in United States and peptide sameness study services in Canada are structured to meet dual-jurisdiction requirements.
Share via:
Video Guide: How to Demonstrate Peptide Sameness for Regulatory Approval
1: What Are FDA Peptide Sameness Study Requirements?
FDA Peptide Sameness Study Requirements mandate comprehensive analytical evidence proving that a synthetic peptide generic is structurally identical to its Reference Listed Drug (RLD). This means confirming exact amino acid sequence, molecular weight, impurity profile, physicochemical characteristics, and stability equivalence before approval under an ANDA or 505(b)(2) pathway.
Under the regulatory oversight of the U.S. Food and Drug Administration, most chemically synthesized peptides are regulated as drugs under the Federal Food, Drug, and Cosmetic Act — not as biologics under the Public Health Service Act. Therefore, the expectation is molecular identity, not just high similarity.
These requirements are further detailed in our guide on characterization of peptides for FDA.
Core Components Required by FDA:
| Requirement Category | What FDA Expects | Analytical Techniques Commonly Used |
|---|---|---|
| Primary Structure Confirmation | Exact amino acid sequence match | LC-HRMS, MS/MS sequencing |
| Molecular Weight Verification | Accurate mass within tight tolerance | High-resolution mass spectrometry |
| Impurity Profiling | Comparable impurity profile | LC-MS, HPLC-UV |
| Counterion & Salt Form | Same salt and composition | Ion chromatography |
| Related Substances | Identification and quantification | LC-MS/MS |
| Stability Data | Comparable degradation pathways | Forced degradation studies |
Sequence confirmation often requires understanding peptide mapping vs peptide sequencing – key differences to select the correct analytical strategy.
FDA Peptide Sameness Study Requirements emphasize analytical rigor because even minor sequence deviations can impact safety or efficacy.
2: Why FDA Peptide Sameness Study Requirements Are Different from Biosimilar Comparability
Peptides are evaluated under generic drug frameworks, whereas biologics undergo biosimilar comparability exercises.
For a deeper comparison, see peptide sameness vs biosimilar comparability.
The FDA distinguishes synthetic peptides from large biologics based on:
- Molecular size
- Manufacturing process (chemical synthesis vs. recombinant DNA)
- Structural complexity
- Immunogenicity risk
For biosimilars, similarity — not identity — is demonstrated. In contrast, FDA Peptide Sameness Study Requirements demand demonstration of molecular identity, not just high similarity.
FDA Peptide Sameness Study Requirements emphasize analytical rigor because:
- Even a single amino acid substitution can alter biological activity.
- Trace impurities may impact safety or immunogenicity.
- Manufacturing route differences can introduce unique degradation products.
- Peptides may undergo oxidation, deamidation, or truncation.
For this reason, regulators expect orthogonal analytical confirmation rather than reliance on a single technique.
Even trace impurities can impact safety. This is why impurity profiling in peptides – why it matters in drug development is a central pillar of regulatory submissions.
This distinction significantly affects regulatory strategy and analytical depth.
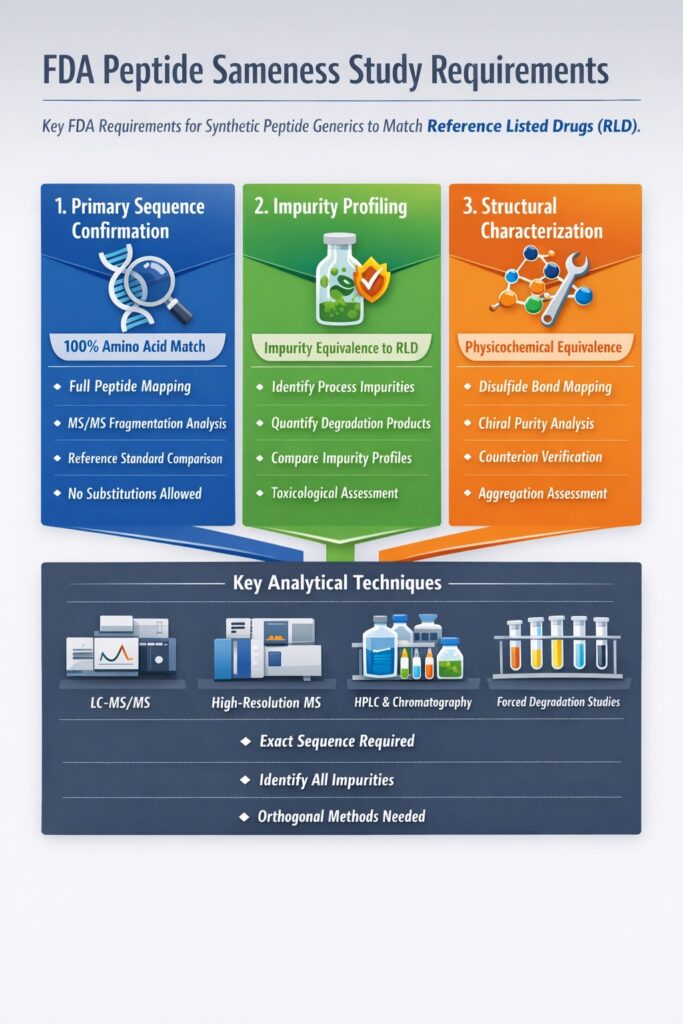
3: Detailed Breakdown of FDA Peptide Sameness Study Requirements
FDA Peptide Sameness Study Requirements demand complete structural, impurity, and physicochemical equivalence between a synthetic peptide generic and its Reference Listed Drug (RLD). Below is a regulator-focused breakdown of the three most critical analytical pillars required by the U.S. Food and Drug Administration.
1. Primary Sequence Confirmation Under FDA Peptide Sameness Study Requirements
The peptide’s amino acid sequence must be confirmed with 100% accuracy — no substitutions, deletions, or additions are permitted.
Advanced PTM analysis may require peptide mapping for PTM analysis and high-resolution LC-MS workflows.
When sequence identity is uncertain, how to identify unknown peptides by LCMS testing becomes essential.
FDA requires definitive structural proof that the generic peptide is molecularly identical to the RLD at the primary structure level.
Regulatory Expectations:
- Complete peptide mapping
- High-resolution MS/MS fragmentation analysis
- Coverage of N-terminal and C-terminal sequences
- Confirmation of post-synthetic modifications (if applicable)
- Direct comparison against a qualified reference standard
Analytical Techniques Commonly Used:
- LC-HRMS (High-Resolution Mass Spectrometry)
- Tandem mass spectrometry (MS/MS sequencing)
- Enzymatic digestion followed by fragment analysis
- Accurate mass determination within tight ppm tolerance
Why This Matters
Even a single amino acid substitution can:
- Alter receptor binding
- Change pharmacokinetics
- Increase immunogenicity risk
- Result in regulatory rejection
Under FDA Peptide Sameness Study Requirements, structural identity — not similarity — is mandatory.
2. Impurity Profiling in FDA Peptide Sameness Study Requirements
Impurity equivalence is a critical determinant of regulatory approval.
FDA expects the impurity profile of the generic peptide to be comparable to that of the RLD, both qualitatively and quantitatively.
Regulatory Expectations:
- Identification of process-related impurities
- Quantification of degradation products
- Comparative impurity fingerprinting vs. RLD
- Detection of low-level sequence variants
- Toxicological qualification of any novel impurity
Types of Impurities Commonly Evaluated:
- Truncated peptides
- Oxidized variants (e.g., methionine oxidation)
- Deamidated species
- Epimerized amino acids
- Residual reagents or synthesis by-products
Analytical Techniques Used:
- LC-MS and LC-MS/MS
- Stability-indicating HPLC-UV
- Forced degradation studies
- Accurate mass impurity identification
Advanced LC-MS platforms are essential to detect trace-level variants that may not be visible using conventional UV detection alone.
See our deep dive on peptide degradation product characterization and what is peptide purity by HPLC and why it matters.
For U.S.-specific testing standards, visit peptide purity testing in United States.
3. Structural Characterization Requirements
Structural confirmation extends beyond sequence and includes physicochemical equivalence.
FDA Peptide Sameness Study Requirements often demand orthogonal analytical approaches to ensure data robustness and integrity.
Structural Elements Evaluated:
- Disulfide bond mapping (if applicable)
- Chiral purity and stereochemical confirmation
- Counterion and salt form verification
- Aggregation assessment
- Conformational analysis (when relevant)
Why Orthogonal Methods Are Important
Regulators prefer multiple complementary analytical techniques to confirm results independently. For example:
| Structural Attribute | Primary Technique | Orthogonal Confirmation |
|---|---|---|
| Molecular Weight | HRMS | Amino acid analysis |
| Sequence Identity | MS/MS mapping | Enzymatic digestion profiling |
| Impurity Levels | LC-MS | HPLC-UV |
| Counterion | Ion chromatography | Elemental analysis |
Orthogonal confirmation strengthens regulatory confidence and minimizes deficiency risk.
Our overview of peptide characterization techniques and applications explains orthogonal strategies in detail.
Key Regulatory Insight
FDA Peptide Sameness Study Requirements are designed to eliminate structural uncertainty before bioequivalence studies are even considered. The regulatory philosophy is clear:
- Identity must be proven analytically.
- Impurity risk must be fully characterized.
- Structural equivalence must be defensible with high-resolution data.
Failure to meet these analytical expectations is one of the most common causes of regulatory delays in peptide ANDA submissions.
If needed, I can next expand this section to include real-world regulatory pitfalls and strategies to prevent Complete Response Letters (CRLs).
4: Health Canada Requirements for Peptide Sameness Studies
Health Canada requires comprehensive analytical evidence demonstrating identity, strength, purity, and quality for synthetic peptide generics filed through an Abbreviated New Drug Submission (ANDS). The scientific standard is rigorous and closely aligned with FDA Peptide Sameness Study Requirements.
Under the regulatory oversight of Health Canada, sponsors must provide robust structural and impurity characterization before market authorization is granted.
Our recent case example on Semaglutide Sameness Evaluation for Health Canada illustrates how peptide sameness packages are structured for Canadian regulators.
For Canadian ANDS filings, sponsors often leverage peptide sameness study services in Canada.
Core Analytical Requirements Under Health Canada
Manufacturers must demonstrate:
- Confirmed Amino Acid Sequence
Full primary structure verification using peptide mapping and MS/MS fragmentation analysis. - Verified Molecular Mass
Accurate molecular weight determination using high-resolution mass spectrometry within tight tolerances. - Comparable Impurity Profile
Identification and quantification of:- Process-related impurities
- Degradation products
- Sequence variants
- Oxidized or deamidated species
- Stability-Indicating Method Validation
Demonstration that analytical methods can reliably detect degradation under:- Thermal stress
- Oxidative conditions
- Hydrolytic environments
- Photolytic exposure
- Bioequivalence (When Applicable)
Required unless a biowaiver is scientifically justified.
Analytical Techniques Commonly Expected
| Requirement | Typical Methods Used |
|---|---|
| Sequence Confirmation | LC-HRMS, MS/MS mapping |
| Molecular Mass | Accurate mass HRMS |
| Impurity Profiling | LC-MS/MS, HPLC-UV |
| Counterion Verification | Ion chromatography |
| Stability Testing | Forced degradation + validated stability-indicating assays |
5: FDA Peptide Sameness Study Requirements vs Health Canada – Key Differences
FDA Peptide Sameness Study Requirements and Health Canada peptide sameness expectations are scientifically aligned, but the regulatory pathways and reference standards differ. While both agencies mandate full analytical sameness, submission structure and terminology vary.
For ANDA-focused filings, review peptide sameness study for ANDA.
Below is a clear regulatory comparison between the U.S. Food and Drug Administration and Health Canada.
Regulatory Comparison Table
| Aspect | FDA (U.S.) | Health Canada |
|---|---|---|
| Regulatory Pathway | 505(j) ANDA or 505(b)(2) | ANDS (Abbreviated New Drug Submission) |
| Analytical Sameness | Mandatory under FDA Peptide Sameness Study Requirements | Mandatory |
| Bioequivalence | Required unless scientifically waived | Required |
| Impurity Qualification | ICH Q3A/Q3B thresholds | ICH Q3A/Q3B thresholds |
| Reference Product | U.S. Reference Listed Drug (RLD) | Canadian Reference Product (CRP) |
Key Regulatory Differences Explained
1. Regulatory Submission Pathway
- In the U.S., synthetic peptide generics are typically filed under 505(j) ANDA or sometimes 505(b)(2) if additional data are required.
- In Canada, submissions are filed as an ANDS.
While naming differs, the scientific data package is largely comparable.
2. Reference Product Requirements
- FDA requires comparison against a U.S.-approved RLD.
- Health Canada requires comparison against a Canadian Reference Product (CRP).
This distinction is important when sourcing comparator material for analytical studies.
3. Bioequivalence Expectations
Both jurisdictions require bioequivalence unless:
- A biowaiver is justified.
- The peptide qualifies under specific exemption criteria.
However, analytical sameness must be established before bioequivalence is considered adequate.
4. Impurity Qualification Standards
Both agencies follow ICH guidance for impurity thresholds:
- Identification thresholds
- Qualification thresholds
- Reporting thresholds
Any novel impurity exceeding limits must be toxicologically justified.
Overall Regulatory Insight
Despite minor procedural differences, the analytical burden is comparable between FDA Peptide Sameness Study Requirements and Health Canada expectations. Both regulators require:
- Confirmed amino acid sequence
- Verified molecular mass
- Comparable impurity profile
- Stability-indicating methods
- Orthogonal analytical confirmation
A peptide sameness package designed to meet FDA Peptide Sameness Study Requirements will generally align well with Health Canada standards, provided jurisdiction-specific documentation and reference products are appropriately addressed.
If helpful, I can next generate a regulatory submission checklist for dual U.S.–Canada peptide filings.

6: Is Bioequivalence Alone Sufficient Under FDA Peptide Sameness Study Requirements?
No. Bioequivalence alone is not sufficient without analytical sameness data.
Bioequivalence cannot replace structural proof. Our work in peptide characterization for IND and NDA demonstrates how analytical depth evolves across regulatory stages.
FDA expects:
- Structural identity confirmation
- Analytical comparability package
- Stability equivalence
- Impurity risk assessment
Bioequivalence studies support therapeutic equivalence but cannot replace structural characterization.
7: Analytical Techniques Used to Meet FDA Peptide Sameness Study Requirements
ResolveMass Laboratories Inc. applies advanced analytical workflows tailored specifically for regulatory compliance.
Common Techniques Include:
- LC-HRMS (High-Resolution Mass Spectrometry)
- Triple Quadrupole LC-MS/MS
- Amino acid analysis
- Peptide mapping
- Chiral chromatography
- Ion chromatography for counterions
- Forced degradation studies
- Stability-indicating method validation
Our team of peptide mass spectrometry experts ensures regulatory-grade data quality.
Sponsors outsourcing analytical work should review peptide testing services for pharmaceutical R&D and top 5 things to look for in a peptide testing laboratory.
Each technique contributes a specific layer of evidence required to meet FDA Peptide Sameness Study Requirements.
8: Regulatory Risks of Inadequate Peptide Sameness Studies
Insufficient analytical characterization is one of the most common causes of regulatory deficiency letters.
Potential consequences include:
- Complete Response Letters (CRLs)
- Data request delays
- Additional toxicology requirements
- Repeat analytical studies
- Delayed market entry
Early analytical support in peptide synthesis – why it’s essential reduces these risks.
Manufacturing approach also matters — see solid vs liquid phase peptide synthesis – which method is better.
Investing in a robust, regulator-aligned peptide sameness study significantly reduces approval risk.
9: Best Practices for Meeting FDA Peptide Sameness Study Requirements
Meeting FDA Peptide Sameness Study Requirements requires proactive planning, rigorous analytical design, and regulator-aligned documentation from the earliest stages of development. The U.S. Food and Drug Administration expects clear scientific justification, reproducible data, and transparent reporting in every peptide generic submission.
Below are industry-proven best practices to strengthen your regulatory strategy and reduce deficiency risk.
1. Begin Analytical Planning Early in Development
Early analytical strategy prevents costly rework later.
- Define regulatory pathway (505(j) vs 505(b)(2)) at project initiation
- Identify required comparability attributes upfront
- Source qualified Reference Listed Drug (RLD) early
- Develop stability-indicating methods during formulation development
Late-stage analytical gaps are a common cause of review delays.
2. Use Orthogonal Analytical Methods
FDA Peptide Sameness Study Requirements favor confirmatory evidence from multiple independent techniques.
For example:
| Attribute | Primary Method | Orthogonal Confirmation |
|---|---|---|
| Sequence Identity | LC-MS/MS | Enzymatic peptide mapping |
| Molecular Weight | HRMS | Amino acid analysis |
| Impurity Levels | LC-MS | HPLC-UV |
| Counterion Form | Ion chromatography | Elemental analysis |
Orthogonal confirmation improves data credibility and regulatory confidence.
3. Compare Against a Certified Reference Standard
Direct side-by-side comparison with a qualified RLD is essential.
Best practices include:
- Same batch comparison where possible
- Parallel impurity profiling
- Overlay chromatograms
- Comparative degradation studies
Regulators expect objective comparability, not theoretical similarity.
4. Document Impurity Justification Thoroughly
Impurity risk assessment must be science-driven and defensible.
Include:
- Structural identification of each impurity
- Quantitative levels vs ICH thresholds
- Toxicological qualification if required
- Batch-to-batch consistency data
- Clear justification for absence/presence differences
Incomplete impurity documentation is one of the most common causes of deficiency letters.
5. Align with ICH Q3A/Q3B Impurity Guidelines
Both FDA and global regulators follow ICH guidance for:
- Reporting thresholds
- Identification thresholds
- Qualification thresholds
Ensure:
- Impurities above thresholds are identified
- Novel impurities are assessed toxicologically
- Specifications reflect validated analytical capability
Regulatory alignment avoids last-minute data requests.
6. Conduct Forced Degradation Studies
Forced degradation proves that your method is stability-indicating.
Stress conditions typically include:
- Heat
- Light
- Oxidation
- Acid/base hydrolysis
These studies demonstrate:
- Degradation pathways
- Detection capability
- Impurity resolution
- Analytical robustness
FDA Peptide Sameness Study Requirements expect stability data to support shelf-life justification.
7. Maintain Clear and Structured Regulatory Documentation
Regulators prioritize:
- Transparency
- Reproducibility
- Complete method validation data
- Logical scientific justification
Best practices:
- Provide clear study summaries
- Include raw data traceability
- Present side-by-side comparison tables
- Ensure consistency between analytical reports and submission modules
Well-organized documentation reduces review time and builds regulator confidence.
Regulatory Insight
FDA Peptide Sameness Study Requirements are not merely analytical checkboxes — they represent a structured scientific demonstration of molecular identity and safety assurance.
Sponsors who:
- Plan early
- Apply orthogonal methods
- Fully characterize impurities
- Align with ICH guidance
- Provide clear documentation
are significantly more likely to achieve smooth regulatory review and timely approval.
If helpful, I can next generate a regulatory risk mitigation checklist specifically designed to prevent Complete Response Letters (CRLs) in peptide ANDA submissions.
Choosing the right CRO partner is critical — read peptide synthesis service – how to choose the right CRO partner.

10: Why Choose ResolveMass for FDA Peptide Sameness Study Requirements?
ResolveMass Laboratories Inc. provides:
- Regulatory-focused peptide characterization
- Advanced mass spectrometry expertise
- ANDA and ANDS submission experience
- Impurity identification and qualification support
- High-resolution structural confirmation
- Customized study design aligned with FDA Peptide Sameness Study Requirements
ResolveMass has supported complex generic characterization projects such as:
- Peptide Characterization of Ganirelix – Generic Project
- Peptide Characterization of Lanreotide – Generic Project
Our regulatory-focused workflows are purpose-built to satisfy FDA Peptide Sameness Study Requirements and Health Canada expectations.
Our analytical scientists understand both the scientific complexity of peptides and the regulatory expectations of FDA and Health Canada.
Conclusion:
FDA Peptide Sameness Study Requirements demand rigorous analytical proof of structural identity, impurity equivalence, and stability comparability for synthetic peptide generics. Both the U.S. Food and Drug Administration and Health Canada require comprehensive mass spectrometry–based characterization before approval.
Manufacturers cannot rely on bioequivalence alone. A complete peptide sameness package must include:
- Sequence confirmation
- Impurity profiling
- Stability data
- Structural verification
- Regulatory documentation
Partnering with an experienced analytical laboratory significantly improves regulatory success and accelerates time to market.
Frequently Asked Questions:
FDA Peptide Sameness Study Requirements mandate comprehensive analytical proof that a synthetic peptide generic is structurally identical to the Reference Listed Drug (RLD).
This includes:
-Primary amino acid sequence confirmation
-Accurate molecular weight verification
-Impurity profiling and qualification
-Physicochemical characterization
-Stability-indicating method validation
These requirements ensure that even minor sequence or impurity differences do not compromise safety or efficacy.
No, most chemically synthesized peptides are regulated as small molecule drugs under the Federal Food, Drug, and Cosmetic Act—not as biosimilars under the Public Health Service Act.
Peptides typically follow the 505(j) ANDA or 505(b)(2) pathway with strict analytical sameness requirements instead of biosimilar comparability frameworks.
FDA expects orthogonal and high-resolution analytical methods to confirm structural identity and purity.
Common techniques include:
-LC-HRMS for accurate mass determination
-MS/MS peptide mapping
-LC-MS impurity profiling
-HPLC-UV for related substances
-Ion chromatography for counterion verification
-Forced degradation studies for stability assessment
Advanced mass spectrometry is critical for detecting low-level sequence variants and oxidation products.
Scientifically, FDA and Health Canada requirements are highly aligned, but regulatory pathways and reference products differ.
Key distinctions:
-FDA requires comparison to a U.S. Reference Listed Drug (RLD)
-Health Canada requires a Canadian Reference Product (CRP)
-Submissions are filed as ANDA/505(b)(2) in the U.S. and ANDS in Canada
Despite procedural differences, analytical rigor is comparable.
Yes, bioequivalence is generally required unless scientifically justified for waiver.
However:
-Analytical sameness must be established first
-A biowaiver may apply under specific regulatory guidance
-Justification must be clearly documented
Analytical data form the foundation before bioequivalence is considered sufficient.
Reference
- Chapter 1: Regulatory Considerations for Peptide Therapeutics.https://books.rsc.org/books/edited-volume/801/chapter/540098/Regulatory-Considerations-for-Peptide-Therapeutics
- Jessica A. Rogers-Crovak, Edward J. Delaney & David J. Detlefsen. Recommendation for Clarifying FDA Policy in Evaluating “Sameness” of Higher Order Structure for Generic Peptide Therapeutics.https://link.springer.com/article/10.1208/s12248-024-00994-8
- Doris Zane , Paul L. Feldman and Jessica Hawes. Development and Regulatory Challenges for Peptide Therapeutics.https://journals.sagepub.com/doi/full/10.1177/1091581820977846
- Larisa C. Wu , Fu Chen , Sau L. Lee , Andre Raw , Lawrence X. Yu. Building parity between brand and generic peptide products: Regulatory and scientific considerations for quality of synthetic peptides.https://www.sciencedirect.com/science/article/abs/pii/S0378517316311930
- Mayur S. Mitra , Steven DeMarco , Brad Holub , Lakshmanan Thiruneelakantapillai , Evan A. Thackaberry. Development of peptide therapeutics: A nonclinical safety assessment perspective.https://www.sciencedirect.com/science/article/abs/pii/S0273230020301926
- Carol F. Kirchhoff, Xiao-Zhuo Michelle Wang, Hugh D. Conlon, Scott Anderson, Anne M. Ryan, Arindam Bose. Biosimilars: Key regulatory considerations and similarity assessment tools.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/full/10.1002/bit.26438


