
Introduction:
Active Ingredient Characterization for ANDA submission is a fundamental requirement to establish that the generic product matches the reference listed drug (RLD) in identity, strength, quality, and purity. This process is closely aligned with sameness evaluation in ANDA and active ingredient sameness in ANDA, which are critical for regulatory success.
In finished dosage forms, this task becomes significantly more complex due to excipients, degradation pathways, and formulation-specific interactions. For pharmaceutical companies pursuing ANDA approval, precise characterization—supported by analytical strategies for sameness study—is not just a regulatory checkbox but a scientific necessity.
Summary:
- Active Ingredient Characterization for ANDA submission is critical to demonstrate sameness, quality, and regulatory compliance.
- Requires orthogonal analytical techniques for ANDA sameness evaluation such as HRMS, LC-MS, and NMR.
- Must confirm identity, purity, potency, and structural integrity within the finished dosage form.
- Regulatory bodies expect robust, reproducible, and validated analytical data.
- Supports peptide sameness study for ANDA and complex molecule evaluation.
- Challenges include matrix interference, degradation, and excipient interactions.
- Strategic analytical design improves ANDA approval success rates and reduces review cycles.
1: What is Active Ingredient Characterization in Finished Dosage Forms?
Active ingredient characterization in finished dosage forms refers to the comprehensive analytical evaluation of the API within its final formulation to confirm identity, purity, potency, and structural integrity.
In simple terms, it ensures that the active drug inside a tablet, capsule, or injection is exactly what it should be, in the right amount, and free from harmful impurities, even in the presence of complex formulation components.
This process is especially critical in peptide sameness study for ANDA and sameness evaluation of synthetic peptides for ANDA where molecular complexity is high.
Unlike raw API testing, finished dosage form analysis must deal with formulation complexity, including excipients, coatings, preservatives, and potential degradation products that can interfere with accurate measurement and identification.
Why is it More Complex Than API Testing Alone?
In finished products, the API is no longer isolated—it is embedded within a matrix of excipients that can:
- Interfere with analytical signals
- Mask impurities or degradation products
- Affect extraction and detection efficiency
- Alter stability and chemical behavior
This makes characterization more challenging and more critical for regulatory submissions like ANDA.
Key Objectives:
1. Confirm API Identity
Ensure the correct active ingredient is present in the formulation, even in the presence of excipients and additives.
2. Quantify Assay (Potency) Accurately
Measure the exact amount of API to confirm it matches the labeled claim and regulatory specifications.
3. Detect and Identify Impurities
Identify process-related impurities, degradation products, and unknown compounds that may impact safety and efficacy.
4. Establish Structural Integrity
Verify that the API maintains its correct molecular structure, stereochemistry, and conformation within the formulation.
5. Demonstrate Equivalence to RLD
Prove that the generic product is pharmaceutically equivalent to the Reference Listed Drug (RLD), a key requirement for ANDA approval.
2: Why is Active Ingredient Characterization Critical for ANDA Submission?
Active Ingredient Characterization for ANDA submission is critical because regulatory agencies require definitive scientific proof that a generic drug is pharmaceutically equivalent to the innovator (Reference Listed Drug, RLD).
In other words, it is the foundation for demonstrating that your product delivers the same safety, quality, and performance as the original drug.
This is closely aligned with FDA peptide sameness study requirements, which emphasize deep structural and impurity-level understanding.
Failure to meet these expectations often leads to issues highlighted in peptide sameness study deficiencies.
Key Regulatory Expectations
- Demonstration of Sameness
The API must match the RLD in identity, strength, and quality attributes. - Use of Validated Analytical Methods
Methods must comply with ICH guidelines and demonstrate accuracy, precision, specificity, and robustness. - Detailed Impurity Profiling
Identification and quantification of process-related impurities and degradation products at trace levels. - Stability-Indicating Methods
Analytical methods must detect changes in API quality over time under various conditions.
Why It Matters for ANDA Approval
Without robust characterization, regulators cannot confirm equivalence—making approval unlikely.
Direct Impact on Submission Success
| Aspect | Impact of Proper Characterization |
|---|---|
| Regulatory Review | Faster and smoother evaluation |
| Data Acceptance | Higher credibility and compliance |
| Risk Reduction | Fewer deficiencies or queries |
| Time-to-Market | Accelerated approval timelines |
Consequences of Inadequate Characterization
Failure to properly characterize the API in finished dosage forms can lead to serious regulatory setbacks:
- Complete Response Letters (CRLs)
Indicating deficiencies that must be addressed before approval - Delayed Approvals
Additional review cycles due to insufficient data - Additional Costly Studies
Rework in analytical development, validation, or impurity identification
3: What Does Active Ingredient Characterization Involve?
Active Ingredient Characterization for ANDA submission involves confirming the identity, strength, purity, and structural integrity of the API within the final drug product.
In practice, it is a multi-dimensional analytical process that ensures the API performs consistently and safely within the finished dosage form, despite the presence of excipients and formulation complexities.
This involves confirming identity, strength, purity, and structural integrity of the API.
For complex molecules like peptides, case-specific studies such as:
- liraglutide sameness study
- semaglutide sameness study
- tirzepatide sameness study
- retatrutide sameness study
- exenatide sameness study
demonstrate how characterization strategies are applied in real-world ANDA scenarios.
Core Elements of Active Ingredient Characterization
1. Identity Confirmation
Ensures that the correct API is present in the formulation.
This step verifies the chemical identity of the active ingredient using techniques such as spectroscopy and chromatography.
- Confirms molecular identity
- Differentiates API from excipients
- Matches API with Reference Listed Drug (RLD)
2. Assay (Potency)
Measures the exact amount of API in the dosage form.
- Confirms label claim compliance (e.g., 100 mg/tablet)
- Ensures dose accuracy and uniformity
- Critical for efficacy and safety
3. Impurity Profiling
Detects and quantifies impurities and degradation products.
- Identifies process-related impurities
- Detects degradation under stress conditions
- Ensures impurities are within ICH limits
- Supports toxicological safety assessment
4. Structural Characterization
Verifies the molecular structure and integrity of the API.
- Confirms chemical structure and stereochemistry
- Detects structural modifications or transformations
- Ensures no unintended changes during formulation
5. Stability Assessment
Evaluates how the API degrades over time under various conditions.
- Monitors shelf-life stability
- Identifies degradation pathways
- Ensures product remains within specifications throughout its lifecycle
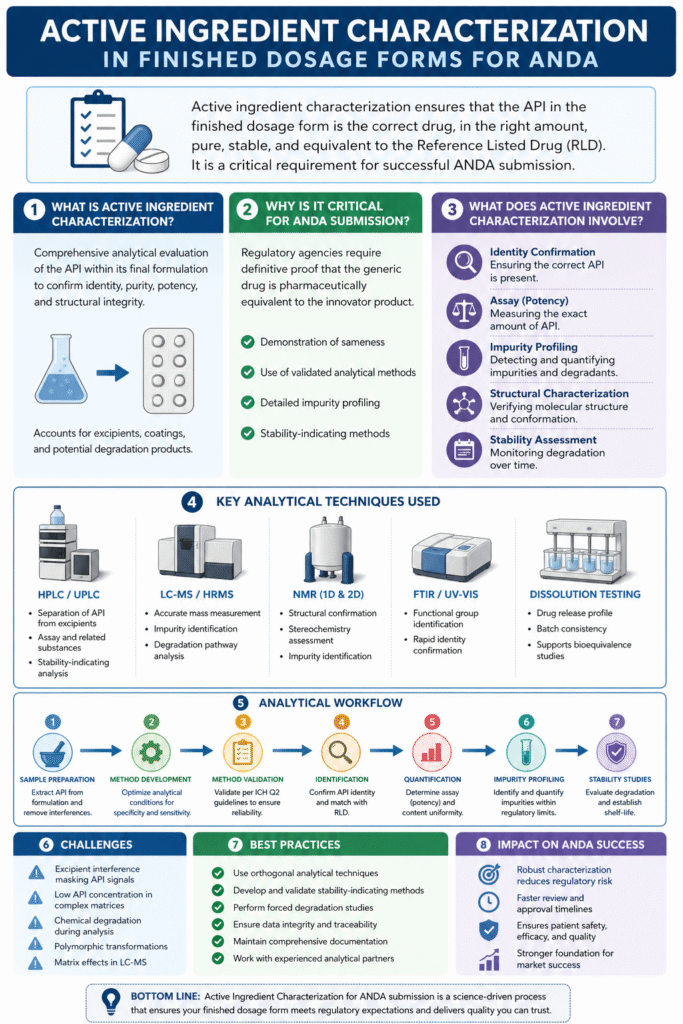
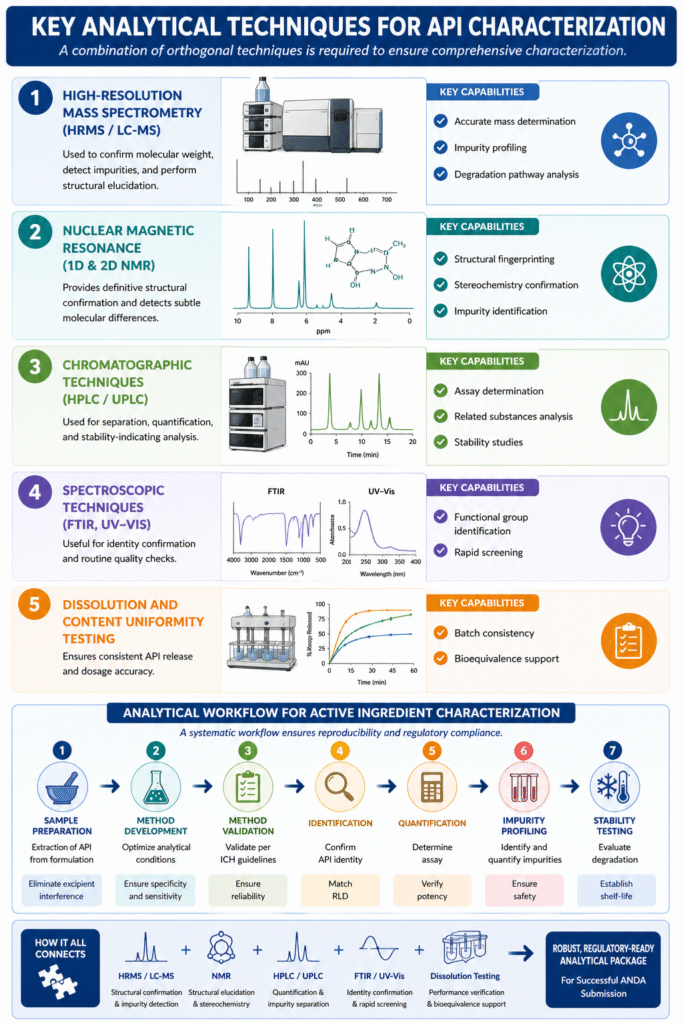
4: Key Analytical Techniques for API Characterization
Active Ingredient Characterization for ANDA submission relies on a combination of orthogonal analytical techniques to ensure accurate, reliable, and comprehensive evaluation of the API within finished dosage forms.
No single technique is sufficient—regulatory acceptance depends on complementary data from multiple methods that collectively confirm identity, purity, potency, and stability.
A combination of orthogonal techniques is required to ensure comprehensive characterization.
These approaches are central to orthogonal analytical techniques for ANDA sameness evaluation.
For peptide-based APIs, advanced studies like:
highlight the importance of combining HRMS, NMR, and chromatography.
1. High-Resolution Mass Spectrometry (HRMS / LC-MS)
Used to confirm molecular weight, detect impurities, and perform structural elucidation.
Key Capabilities
- Accurate mass determination for API confirmation
- Impurity profiling at trace levels
- Degradation pathway analysis under stress conditions
Why it matters:
HRMS provides high sensitivity and specificity, making it essential for identifying unknown impurities and confirming molecular composition.
2. Nuclear Magnetic Resonance (1D & 2D NMR)
Provides definitive structural confirmation and detects subtle molecular differences.
Key Capabilities
- Structural fingerprinting of the API
- Stereochemistry confirmation
- Impurity identification and structural elucidation
Why it matters:
NMR is considered a gold standard for structural verification, especially when demonstrating equivalence to the Reference Listed Drug (RLD).
3. Chromatographic Techniques (HPLC / UPLC)
Used for separation, quantification, and stability-indicating analysis.
Key Capabilities
- Assay determination (API content)
- Related substances analysis
- Stability studies with degradation monitoring
Why it matters:
Chromatography is the workhorse technique for routine quality control and regulatory submissions.
4. Spectroscopic Techniques (FTIR, UV-Vis)
Useful for identity confirmation and routine quality checks.
Key Capabilities
- Functional group identification
- Rapid identity confirmation
- Non-destructive analysis
Why it matters:
These methods provide quick and cost-effective verification, supporting primary analytical data.
5. Dissolution and Content Uniformity Testing
Ensures consistent API release and dosage accuracy.
Key Capabilities
- Batch consistency evaluation
- Drug release profiling
- Bioequivalence support
Why it matters:
These tests link analytical characterization to in vivo performance, which is critical for ANDA approval.
Analytical Workflow for Active Ingredient Characterization
A systematic workflow ensures reproducibility, regulatory compliance, and data integrity throughout the characterization process.
Step-by-Step Approach
| Step | Activity | Purpose |
|---|---|---|
| Sample Preparation | Extraction of API from formulation | Eliminate excipient interference |
| Method Development | Optimize analytical conditions | Ensure specificity and sensitivity |
| Method Validation | Validate as per ICH guidelines | Ensure accuracy and reliability |
| Identification | Confirm API identity | Match Reference Listed Drug (RLD) |
| Quantification | Determine assay (potency) | Verify label claim |
| Impurity Profiling | Identify and quantify impurities | Ensure safety and compliance |
| Stability Testing | Evaluate degradation over time | Establish shelf-life |
5: Challenges in Characterizing APIs in Finished Dosage Forms
The primary challenge in Active Ingredient Characterization for ANDA submission is isolating and accurately analyzing the API within a complex formulation matrix.
In finished dosage forms, the API coexists with excipients, coatings, and other components that can interfere with analytical measurements—making precise characterization significantly more difficult than testing the API alone.
Challenges such as excipient interference and matrix effects are commonly encountered, particularly in peptide formulations.
Organizations often rely on specialized providers offering:
to overcome these analytical complexities.
Common Challenges
1. Excipient Interference
Excipients can mask or overlap API signals, leading to inaccurate identification or quantification.
- Co-elution in chromatography
- Signal suppression in LC-MS
- Overlapping spectral peaks
2. Low API Concentration
In some formulations, the API is present in very low amounts relative to excipients, making detection challenging.
- Reduced sensitivity
- Difficulty in impurity detection
- Increased analytical variability
3. Degradation During Sample Preparation
APIs may degrade during extraction or handling, leading to false impurity profiles.
- Exposure to heat, light, or pH changes
- Formation of artificial degradants
- Misleading stability conclusions
4. Polymorphic Transformations
The API may exist in different crystalline forms (polymorphs) that can alter performance.
- Changes in solubility and dissolution
- Impact on bioavailability
- Regulatory concerns if not controlled
5. Matrix Effects in LC-MS Analysis
Complex formulation components can interfere with ionization in LC-MS.
- Signal suppression or enhancement
- Reduced accuracy in quantification
- Difficulty in trace impurity detection
Mitigation Strategies
Overcoming these challenges requires a strategic and scientifically robust analytical approach.
1. Selective Extraction Techniques
- Isolate API efficiently from excipients
- Use solvent optimization and sample cleanup methods
- Minimize interference from formulation components
2. Stability-Indicating Methods
- Develop methods that can separate API from its degradants
- Perform forced degradation studies
- Ensure accurate stability assessment
3. Orthogonal Analytical Approaches
- Combine LC-MS, NMR, HPLC, and spectroscopy
- Cross-verify results across techniques
- Improve confidence in data
4. Robust Method Validation
- Validate methods as per ICH guidelines
- Ensure accuracy, precision, specificity, and robustness
- Maintain reproducibility across batches
6: Regulatory Considerations for ANDA Submission
Active Ingredient Characterization for ANDA submission must meet strict regulatory expectations, requiring scientifically sound, reproducible, and well-documented analytical data.
Regulatory agencies such as the FDA evaluate whether your data package clearly demonstrates identity, purity, potency, and equivalence to the Reference Listed Drug (RLD).
Regulatory compliance aligns closely with expectations outlined in:
- ICH guidelines
- FDA ANDA requirements
- Advanced frameworks like analytical strategies for sameness study
A strong regulatory package must clearly demonstrate active ingredient sameness using validated and reproducible methods.
Key Regulatory Guidelines
1. ICH Q2 (R1): Method Validation
Defines the requirements for validating analytical methods used in characterization.
- Accuracy and precision
- Specificity and selectivity
- Linearity and range
- Robustness and reproducibility
Why it matters:
Ensures that all analytical methods generate reliable and defensible data.
2. ICH Q3A / Q3B: Impurity Guidelines
Provides limits and reporting requirements for impurities in drug substances and products.
- Identification thresholds
- Qualification thresholds
- Reporting limits
Why it matters:
Ensures patient safety by controlling and qualifying impurities.
3. FDA Guidance for ANDA Submissions
Outlines expectations for demonstrating pharmaceutical equivalence and quality.
- Comparative analysis with RLD
- Comprehensive analytical characterization
- Stability data requirements
- Batch consistency evidence
Why it matters:
Aligns your submission with regulatory review expectations, reducing approval risk.
Documentation Requirements
Regulators require complete, transparent, and well-structured documentation to support your analytical findings.
Essential Documents
- Analytical Method Validation Reports
Demonstrating compliance with ICH Q2 requirements - Chromatograms and Spectral Data
Raw and processed data from HPLC, LC-MS, NMR, etc. - Impurity Qualification Data
Identification, quantification, and toxicological assessment - Comparative Data with RLD
Side-by-side evidence showing equivalence
7: Best Practices for Successful ANDA Characterization
Active Ingredient Characterization for ANDA submission succeeds when a scientifically rigorous, reproducible, and well-documented approach is followed from method development to final reporting.
Adopting best practices not only ensures data integrity and regulatory acceptance, but also reduces the risk of delays, deficiencies, and rework during review.
Best practices include:
- Use of orthogonal analytical techniques
- Strong alignment with analytical strategies for sameness study
- Deep understanding of active ingredient sameness in ANDA
These practices significantly improve submission quality and reduce regulatory risk.
Recommended Approach
1. Use Multiple Orthogonal Techniques
Rely on complementary analytical methods to build strong, defensible data.
- Combine LC-MS, HRMS, NMR, and HPLC/UPLC
- Cross-verify identity, impurities, and structure
- Increase confidence in results and regulatory acceptance
Why it matters:
No single technique is sufficient—regulators expect converging evidence from multiple methods.
2. Ensure Method Robustness and Reproducibility
Develop methods that consistently perform under varied conditions.
- Validate per ICH Q2 guidelines
- Test robustness (pH, temperature, flow rate variations)
- Ensure reproducibility across analysts and instruments
Why it matters:
Reliable methods reduce variability and ensure consistent batch release and regulatory confidence.
3. Perform Forced Degradation Studies
Evaluate how the API behaves under stress conditions.
- Stress under acid/base, oxidation, heat, and light
- Identify degradation pathways
- Develop stability-indicating methods
Why it matters:
Helps demonstrate product stability and impurity control, a key regulatory requirement.
4. Maintain Comprehensive Documentation
Ensure complete and transparent recording of all analytical activities.
- Method development and validation reports
- Raw data (chromatograms, spectra)
- Impurity identification and qualification data
- Stability study reports
Why it matters:
Well-organized documentation supports audit readiness and smooth regulatory review.
5. Partner with Experienced Analytical Laboratories
Leverage specialized expertise for complex characterization challenges.
- Access to advanced instrumentation (HRMS, 2D NMR)
- Expertise in complex formulations and impurities
- Regulatory-compliant workflows
Why it matters:
Experienced partners help avoid common pitfalls and accelerate ANDA approval timelines.
8: How ResolveMass Laboratories Supports ANDA Characterization
ResolveMass Laboratories Inc. provides advanced analytical solutions tailored for complex pharmaceutical formulations.
Core Expertise
- HRMS-based impurity profiling
- Peptide and complex molecule characterization
- 1D/2D NMR structural elucidation
- Customized analytical method development
Why It Matters
- Reduces regulatory risk
- Accelerates ANDA approval timelines
- Ensures data reliability and compliance
Conclusion:
Active Ingredient Characterization for ANDA submission is a critical scientific and regulatory process that ensures generic drug equivalence, safety, and quality.
By leveraging advanced analytical techniques, implementing robust workflows, and addressing formulation-specific challenges, pharmaceutical companies can significantly improve their chances of ANDA approval.
A strategic, data-driven approach—supported by experienced analytical partners—transforms characterization from a regulatory hurdle into a competitive advantage.
Frequently Asked Questions:
API characterization is critical because regulators require proof of pharmaceutical equivalence. It ensures the generic drug matches the innovator in quality, safety, and efficacy. Without proper characterization, approval is unlikely. It also reduces regulatory queries and deficiencies. Strong data improves review confidence. Ultimately, it accelerates time-to-market.
Key parameters include identity, assay (potency), impurity profile, and structural integrity. Stability is also evaluated to understand degradation behavior. These parameters ensure the API meets quality standards. They confirm equivalence with the RLD. Each parameter is assessed using validated methods. Together, they form a complete analytical profile.
Finished dosage forms present challenges due to excipient interference and complex matrices. Low API concentration can make detection difficult. Degradation during sample preparation may affect results. Polymorphic changes can alter drug performance. Matrix effects in LC-MS can impact accuracy. These issues require advanced analytical strategies to overcome.
Regulatory agencies assess whether the data demonstrates identity, purity, and potency. They require validated methods as per ICH guidelines. Complete documentation including chromatograms and spectra is essential. Comparative data with the RLD is also reviewed. Consistency and reproducibility are key factors. Any gaps may lead to deficiencies or delays.
Inadequate characterization can lead to regulatory setbacks. Companies may receive Complete Response Letters (CRLs). Approval timelines can be significantly delayed. Additional studies may be required, increasing costs. It can also impact product launch plans. Proper characterization helps avoid these risks.
Reference
- Cohen EM, Lin LY. Active Pharmaceutical Ingredients. InGeneric Drug Product Development 2014 Oct 31 (pp. 39-52). CRC Press.https://api.taylorfrancis.com/content/chapters/edit/download?identifierName=doi&identifierValue=10.1201/9781420030419-5&type=chapterpdf
- Allen L, Ansel HC. Ansel’s pharmaceutical dosage forms and drug delivery systems. Lippincott Williams & Wilkins; 2013 Dec 23.https://books.google.com/books?hl=en&lr=&id=JteCAwAAQBAJ&oi=fnd&pg=PP1&dq=How+to+Characterize+Active+Ingredients+in+Finished+Dosage+Forms+for+ANDA&ots=j18m6xRB33&sig=NzWkrbWQsts-Q67KIy9UxIa8G6k
- Raw AS, Furness MS, Gill DS, Adams RC, Holcombe Jr FO, Yu LX. Regulatory considerations of pharmaceutical solid polymorphism in Abbreviated New Drug Applications (ANDAs). Advanced drug delivery reviews. 2004 Feb 23;56(3):397-414.https://www.sciencedirect.com/science/article/pii/S0169409X03002242
- Bhattycharyya L, Dabbah R, Hauck W, Sheinin E, Yeoman L, Williams R. Equivalence studies for complex active ingredients and dosage forms. The AAPS journal. 2005 Dec;7(4):77.https://link.springer.com/article/10.1208/aapsj070477
- Ahmed SS, Shailesh T, Hemanth KS, Gangadharappa HV, Gowrav MP. An overview on post approval changes to an approved ANDA in US-FDA. Research Journal of Pharmacy and Technology. 2021;14(1):506-12.https://search.proquest.com/openview/00b2fc82d5e3a108e803912132bff3e7/1?pq-origsite=gscholar&cbl=1096441


