
Introduction:
Active Ingredient Sameness in ANDA is demonstrated by proving that the generic drug’s API is identical to the Reference Listed Drug (RLD) in structure, properties, and impurity profile using advanced analytical techniques.
In Abbreviated New Drug Application (ANDA) submissions, one of the most critical scientific and regulatory requirements is establishing that the active pharmaceutical ingredient (API) in the generic product is the same as that of the RLD. This is not a simple checkbox—it requires a comprehensive, data-driven analytical approach.
For example, detailed case studies such as
Liraglutide sameness study and
Semaglutide sameness study
highlight how advanced analytical techniques are applied in real-world ANDA scenarios.
Regulatory bodies expect clear, reproducible, and scientifically justified evidence to ensure therapeutic equivalence, safety, and efficacy. This blog outlines how to effectively demonstrate Active Ingredient Sameness in ANDA submissions using modern analytical strategies.
Summary:
- Active Ingredient Sameness in ANDA ensures the generic drug matches the Reference Listed Drug (RLD) in identity, strength, quality, and purity.
- Regulatory agencies require robust analytical evidence, not just theoretical similarity.
- Techniques like LC-MS, HRMS, NMR, and impurity profiling are critical for proving sameness.
- Challenges include impurity differences, polymorphism, and complex APIs such as peptides.
- A structured analytical strategy significantly improves ANDA approval success rates.
1: What Does Active Ingredient Sameness in ANDA Mean?
Active Ingredient Sameness in ANDA means the API must be demonstrably identical to the Reference Listed Drug (RLD) in structure, properties, and impurity profile based on comprehensive analytical evidence.
In regulatory terms, this requirement ensures that the generic drug delivers the same therapeutic effect and safety profile as the innovator product—without the need for repeating full clinical trials.
Key Elements of Sameness
To establish Active Ingredient Sameness in ANDA, the following aspects must align with the RLD:
- Molecular Structure & Stereochemistry
The API must have the exact same chemical structure, including stereochemical configuration (e.g., chirality, geometric isomers). - Salt Form / Polymorphic Form (if applicable)
The same salt (e.g., hydrochloride, sodium) and solid-state form must be used, as differences can impact solubility and bioavailability. - Impurity Profile
The type and level of impurities must be comparable to the RLD, with no new or significantly higher impurities introduced. - Physicochemical Properties
Critical properties such as solubility, pKa, particle size, and crystalline form should be equivalent.
Why Even Minor Differences Matter
Even small variations can trigger regulatory concerns because they may affect:
- Drug absorption and bioavailability
- Stability and shelf-life
- Safety and toxicity profiles
For example:
- Isomeric differences can alter pharmacological activity
- Trace impurities may introduce unexpected toxicity
- Polymorphic changes can impact dissolution behavior
Bottom Line
Demonstrating Active Ingredient Sameness in ANDA is not just about matching a chemical formula—it requires a deep, multi-dimensional analytical comparison to ensure the generic product is truly equivalent to the RLD in every critical aspect.
For a deeper regulatory and scientific overview, refer to
Sameness evaluation in ANDA
2: Why is Demonstrating Sameness Critical for ANDA Approval?
Demonstrating Active Ingredient Sameness in ANDA is critical because it proves that the generic drug will perform identically to the Reference Listed Drug (RLD) in terms of safety, quality, and efficacy—without requiring full clinical trials.
In an ANDA pathway, regulators rely heavily on analytical and comparative data rather than clinical studies. Therefore, establishing sameness is the foundation of approval.
Key Reasons Why Sameness is Essential
- Regulatory Compliance
Regulatory agencies such as the U.S. FDA mandate clear evidence that the API in the generic product is the same as the RLD. Without this, the application cannot proceed. - Patient Safety
Ensures that no new impurities, degradation products, or structural variations introduce unexpected toxicity risks. - Therapeutic Equivalence
Confirms that the generic drug will deliver the same clinical effect, dosage performance, and bioavailability as the innovator product. - Reduced Need for Clinical Trials
A strong demonstration of sameness allows manufacturers to avoid costly and time-consuming clinical studies. - Faster Regulatory Approval
Well-documented sameness data minimizes regulatory questions, reduces review cycles, and accelerates time-to-market.
Regulatory Risks of Inadequate Sameness Data
Failure to properly demonstrate sameness can result in:
- Application rejection in severe cases
- Complete Response Letters (CRLs) requesting additional data
- Major review delays due to deficiencies or inconsistencies
For peptide-based drugs, regulatory expectations are even more stringent. Learn more here:
FDA peptide sameness study requirements
3: Key Analytical Techniques for Demonstrating Sameness
A combination of orthogonal analytical techniques is required to conclusively demonstrate Active Ingredient Sameness in ANDA by confirming identity, structure, and impurity equivalence with the Reference Listed Drug (RLD).
1. LC-MS and HRMS (High-Resolution Mass Spectrometry)
- Accurate molecular weight confirmation
- Detection of trace-level impurities
- Structural elucidation of unknown compounds
- Comparative profiling with RLD
2. Nuclear Magnetic Resonance (NMR) Spectroscopy
- Structural confirmation using 1D and 2D NMR
- Stereochemistry and isomer identification
- Functional group verification
- Detection of subtle structural differences
3. Chromatographic Techniques (HPLC/UPLC/GC)
- Impurity profiling and comparison
- Quantitative analysis of API and related substances
- Stability-indicating methods
- Batch-to-batch consistency evaluation
4. Spectroscopic Methods (FTIR & UV-Vis)
- Functional group identification (FTIR)
- Absorption characteristics (UV-Vis)
- Fingerprint comparison with RLD
- Complementary structural confirmation
For peptide-focused analytical workflows, explore:
Peptide sameness study for ANDA
4: Analytical Strategy for Active Ingredient Sameness in ANDA
A structured analytical strategy for Active Ingredient Sameness in ANDA integrates multiple complementary techniques to generate robust, reproducible, and regulatory-acceptable evidence of equivalence with the Reference Listed Drug (RLD).
Real-world implementation examples include:
Liraglutide sameness study for ANDA submission
Octreotide sameness study for ANDA submission
Step-by-Step Approach
| Step | Objective | Techniques Used |
|---|---|---|
| API Characterization | Confirm identity and chemical structure | LC-MS, HRMS, NMR |
| Impurity Profiling | Compare impurity types and levels | HPLC, UPLC |
| Forced Degradation | Understand degradation pathways | Stability studies |
| Comparative Analysis | Match API with RLD | Overlay chromatograms, spectral comparison |
| Method Validation | Ensure accuracy and reproducibility | ICH guidelines |
Key Considerations
- Use Orthogonal Methods
Apply multiple independent techniques to confirm results and eliminate ambiguity. - Ensure High Sensitivity
Analytical methods must detect trace-level impurities and minor structural differences. - Maintain Reproducibility and Accuracy
Methods should consistently generate reliable data across batches and laboratories.
5: Challenges in Demonstrating Active Ingredient Sameness
Active Ingredient Sameness in ANDA can be challenging due to structural complexity, impurity variations, and limited access to Reference Listed Drug (RLD) samples, requiring highly advanced analytical approaches.
Common Challenges
- Trace-Level Impurity Detection
Identifying impurities at very low concentrations is difficult but critical for regulatory acceptance. - Isomer Differentiation
Distinguishing between stereoisomers or structural isomers can be complex, especially for chiral APIs. - Polymorphic Variations
Different solid-state forms can affect solubility, stability, and bioavailability. - Complex APIs
Peptides, large synthetic molecules, and structurally intricate compounds require advanced characterization techniques. - Limited RLD Availability
Restricted access to sufficient RLD samples can limit comprehensive comparative analysis.
Mitigation Strategies
- Use High-Resolution Analytical Tools
Techniques such as HRMS and 2D NMR help detect subtle structural and impurity differences. - Develop Sensitive and Selective Methods
Optimize analytical methods to accurately quantify trace impurities and minor variations. - Perform Forced Degradation Studies
Understand degradation pathways and compare them with the RLD. - Apply Advanced Data Interpretation Techniques
Use sophisticated software and expert analysis to interpret complex datasets.
Common industry pitfalls are discussed here:
Peptide sameness study deficiencies
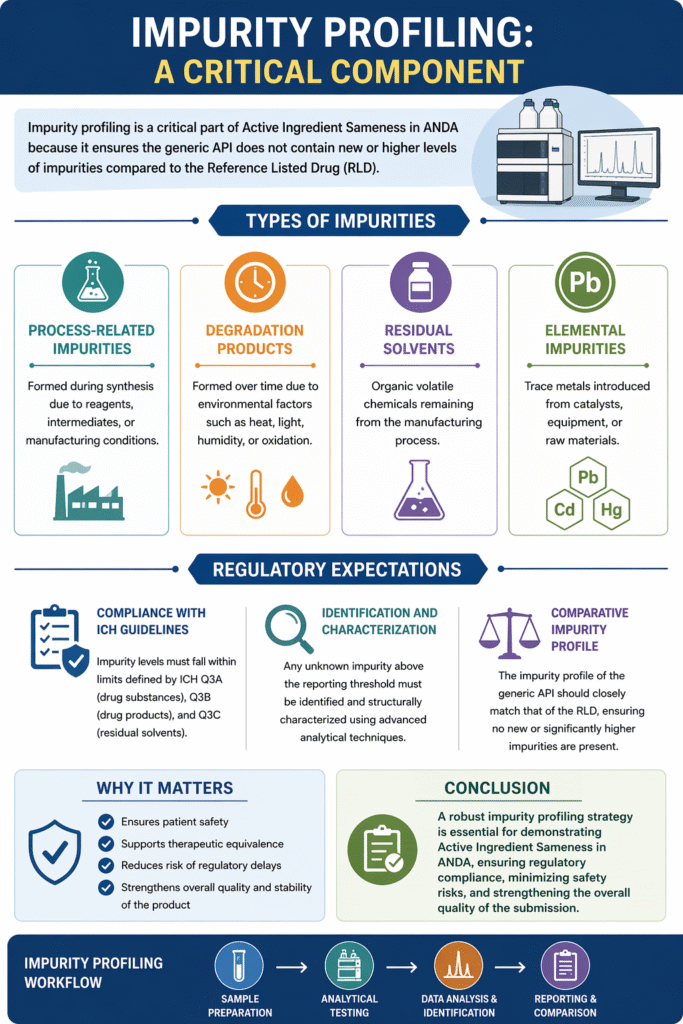
6: Impurity Profiling: A Critical Component
Impurity profiling is a critical part of Active Ingredient Sameness in ANDA because it ensures the generic API does not contain new or higher levels of impurities compared to the Reference Listed Drug (RLD).
Regulatory agencies place strong emphasis on impurity control, as even trace impurities can impact drug safety, efficacy, and stability.
Types of Impurities
- Process-Related Impurities
Formed during synthesis due to reagents, intermediates, or manufacturing conditions. - Degradation Products
Formed over time due to environmental factors such as heat, light, humidity, or oxidation. - Residual Solvents
Organic volatile chemicals remaining from the manufacturing process. - Elemental Impurities
Trace metals introduced from catalysts, equipment, or raw materials.
Regulatory Expectations
- Compliance with ICH Guidelines
Impurity levels must fall within limits defined by ICH Q3A, Q3B, and Q3C guidelines. - Identification and Characterization
Unknown impurities above threshold levels must be identified and structurally characterized. - Comparative Impurity Profile
The impurity profile of the generic API should closely match that of the RLD, with no new or significantly higher impurities.
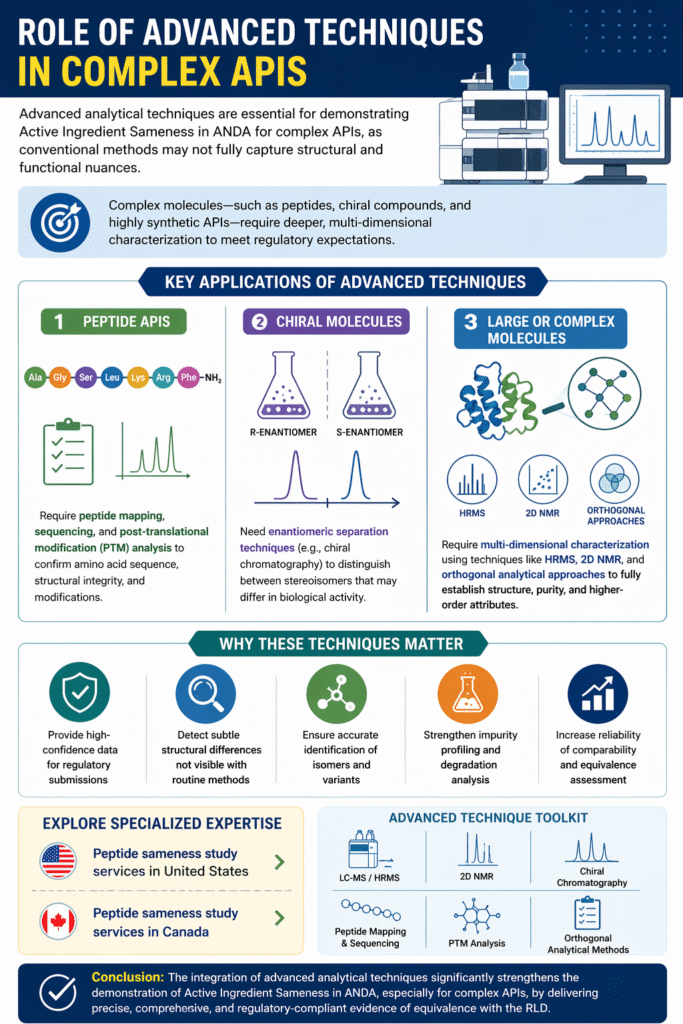
7: Role of Advanced Techniques in Complex APIs
Advanced analytical techniques are essential for demonstrating Active Ingredient Sameness in ANDA for complex APIs, as conventional methods may not fully capture structural and functional nuances.
Complex molecules—such as peptides, chiral compounds, and highly synthetic APIs—require deeper, multi-dimensional characterization to meet regulatory expectations.
Key Applications of Advanced Techniques
- Peptide APIs
Require peptide mapping, sequencing, and post-translational modification (PTM) analysis to confirm amino acid sequence, structural integrity, and modifications. - Chiral Molecules
Need enantiomeric separation techniques (e.g., chiral chromatography) to distinguish between stereoisomers that may differ in biological activity. - Large or Complex Molecules
Require multi-dimensional characterization using techniques like HRMS, 2D NMR, and orthogonal analytical approaches to fully establish structure and purity.
Why These Techniques Matter
- Provide high-confidence data for regulatory submissions
- Detect subtle structural differences not visible with routine methods
- Ensure accurate identification of isomers and variants
- Strengthen impurity and degradation analysis
For specialized services and expertise, explore:
Peptide sameness study services in United States
Peptide sameness study services in Canada
8: Regulatory Expectations and Compliance
Regulatory authorities require comprehensive, well-documented analytical evidence—supported by validated methods—to demonstrate Active Ingredient Sameness in ANDA submissions.
This ensures that the generic product meets the same quality, safety, and efficacy standards as the Reference Listed Drug (RLD).
Key Guidelines
- ICH Q6A (Specifications)
Defines acceptance criteria and test procedures for drug substances and products. - ICH Q3A/B (Impurities)
Provides guidance on identification, qualification, and control of impurities in drug substances and products. - FDA Guidance for ANDA Submissions
Outlines regulatory expectations for demonstrating sameness, including analytical and comparative requirements.
Documentation Requirements
- Detailed Analytical Reports
Comprehensive data covering API characterization, impurity profiling, and comparative analysis. - Method Validation Data
Evidence that analytical methods are accurate, precise, specific, and reproducible as per ICH guidelines. - Comparative Study Results
Side-by-side comparison of the generic API with the RLD using validated analytical techniques. - Justification of Differences (if any)
Scientific rationale explaining any observed differences, supported by data demonstrating no impact on safety or efficacy.
For peptide-specific regulatory expectations, refer again to:
FDA peptide sameness study requirements
9: Best Practices for Successful ANDA Submission
Following best practices ensures a smoother regulatory review and faster approval.
Recommended Practices
- Develop a robust analytical strategy early
- Use orthogonal techniques for confirmation
- Ensure high sensitivity for impurity detection
- Maintain complete documentation
- Perform thorough comparative studies
10: How ResolveMass Laboratories Supports Sameness Studies
ResolveMass Laboratories provides advanced analytical solutions to demonstrate Active Ingredient Sameness in ANDA with high precision and regulatory compliance.
Core Capabilities
- High-resolution LC-MS and HRMS analysis
- 1D and 2D NMR characterization
- Comprehensive impurity profiling
- Peptide mapping and sequencing
- Method development and validation
With deep expertise in complex API characterization, ResolveMass ensures scientifically robust and regulatory-ready data packages.
From small molecules to complex peptides like
Liraglutide sameness study and
Semaglutide sameness study,
the team delivers high-quality, regulatory-ready data.
Conclusion:
Active Ingredient Sameness in ANDA is demonstrated through a comprehensive analytical approach that confirms identity, structure, and impurity equivalence with the RLD.
A successful ANDA submission depends on:
- Strong analytical strategy
- Advanced instrumentation
- Accurate impurity profiling
- Regulatory-compliant documentation
By leveraging cutting-edge techniques and scientific expertise, pharmaceutical companies can significantly improve approval success rates and reduce regulatory risks.
Frequently Asked Questions:
Active Ingredient Sameness in ANDA refers to proving that the generic drug’s API is identical to the Reference Listed Drug (RLD) in structure, physicochemical properties, and impurity profile. This is demonstrated using advanced analytical techniques such as LC-MS, NMR, and chromatography. It ensures that the generic product performs the same as the innovator drug. Regulatory agencies rely on this evidence instead of clinical trials. A strong demonstration of sameness is essential for ANDA approval.
Demonstrating API sameness is critical because it ensures therapeutic equivalence, safety, and quality of the generic drug. Regulatory bodies like the FDA require this evidence to confirm that the generic will behave identically to the RLD. It reduces the need for extensive clinical trials, saving time and cost. Inadequate demonstration can lead to delays or rejection. Therefore, it is a foundational requirement in ANDA submissions.
Regulatory agencies evaluate sameness based on comprehensive analytical data and validated methods. They review structural characterization, impurity profiles, and comparative studies with the RLD. Compliance with ICH guidelines and FDA expectations is essential. Any observed differences must be scientifically justified. A well-documented submission increases the likelihood of approval.
Advanced techniques are essential for analyzing complex APIs such as peptides and chiral molecules. Methods like HRMS, 2D NMR, and peptide mapping provide detailed structural insights. These tools help detect subtle differences that conventional methods may miss. They also support impurity and degradation analysis. Their use strengthens the overall evidence for API sameness.
If API sameness is not adequately demonstrated, the ANDA may face regulatory delays or rejection. Agencies may issue a Complete Response Letter (CRL) requesting additional data. This increases time-to-market and development costs. In some cases, the application may need significant rework. Proper analytical strategy helps avoid these risks.
Companies can improve approval chances by adopting a robust analytical strategy early in development. Using orthogonal techniques ensures comprehensive characterization. High sensitivity methods help detect trace impurities accurately. Maintaining complete and compliant documentation is essential. Collaborating with experienced analytical partners can also enhance success rates.
Yes, peptide drugs are more complex due to their structure, sequence variability, and potential modifications. They require advanced techniques like peptide mapping, sequencing, and PTM analysis. Small changes can significantly impact their biological activity. Regulatory expectations for peptides are also more stringent. Therefore, demonstrating sameness for peptides requires a more detailed and sophisticated approach.
Reference
- Raw AS, Furness MS, Gill DS, Adams RC, Holcombe Jr FO, Yu LX. Regulatory considerations of pharmaceutical solid polymorphism in Abbreviated New Drug Applications (ANDAs). Advanced drug delivery reviews. 2004 Feb 23;56(3):397-414.https://www.sciencedirect.com/science/article/pii/S0169409X03002242
- Wittayanukorn S, Rosenberg M, Schick A, Hu M, Wang Z, Babiskin A, Lionberger R, Zhao L. Factors that have an impact on abbreviated new drug application (ANDA) submissions. Therapeutic Innovation & Regulatory Science. 2020 Nov;54(6):1372-81.https://link.springer.com/article/10.1007/s43441-020-00163-x
- Pawar J, Hegde N, Sharma S. Focusing on First Cycle Approval in ANDA Submission: Understanding Common Deficiencies & Case Study Insights. Therapeutic Innovation & Regulatory Science. 2025 May;59(3):426-37.https://link.springer.com/article/10.1007/s43441-025-00755-5
- Cohen EM, Lin LY. Active Pharmaceutical Ingredients. InGeneric Drug Product Development 2014 Oct 31 (pp. 39-52). CRC Press.https://api.taylorfrancis.com/content/chapters/edit/download?identifierName=doi&identifierValue=10.1201/9781420030419-5&type=chapterpdf


