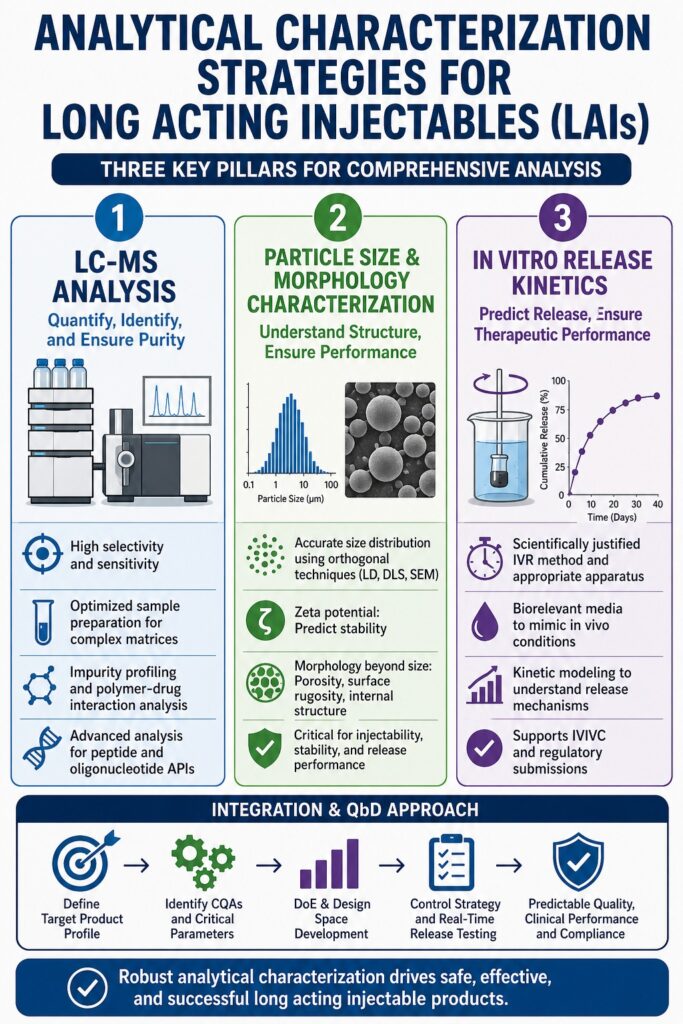
Introduction: Why Conventional Analytical Frameworks Fall Short for LAI Systems
The Analysis of Long Acting Injectable Formulations is one of the most technically demanding areas in pharmaceutical development. Long acting injectable (LAI) systems such as polymeric microparticles, lipid suspensions, in situ forming depots, and nanosuspensions are designed to release APIs over weeks or months. This extended release behavior introduces analytical challenges that cannot be addressed using conventional dissolution and assay methods. Because the formulation changes over time, analytical strategies must capture both early burst release and long-term diffusion-controlled drug release.
Depot formation, polymer degradation, and tissue interaction create evolving conditions throughout the dosing interval. These changes require analytical approaches that evaluate chemical stability, particle properties, and release kinetics together. Small variations in particle size, polymer composition, or drug distribution can significantly affect clinical performance. Therefore, the Analysis of Long Acting Injectable Formulations requires dedicated characterization strategies that combine chemical, physical, and biological understanding.
Explore our specialized services for complex drug delivery: CDMO for Long Acting Injectable Formulation Development
The following sections describe three major analytical pillars: LC-MS characterization, particle size and morphology analysis, and in vitro release kinetics testing. Together, these approaches provide a comprehensive understanding of depot performance and support regulatory submissions. A structured analytical workflow is essential for developing robust long acting injectable products.
Share via:
📋 Article Summary — Key Takeaways
- The Analysis of Long Acting Injectable Formulations requires multiple analytical techniques, as a single method cannot fully evaluate the structural and release complexity of LAI drug products. A combination of complementary tools is necessary to understand depot behavior and long-term performance.
- LC-MS (Liquid Chromatography–Mass Spectrometry) plays a central role in API quantification, impurity identification, and measurement of drug release during in vitro studies. Its high sensitivity and selectivity make it especially useful for complex matrices commonly seen in LAI formulations.
- Particle size and morphology function as critical quality attributes that influence injectability, depot stability, and release kinetics. Because different methods measure different particle characteristics, orthogonal sizing techniques such as laser diffraction, DLS, and microscopy are typically required.
- In vitro release testing for LAI systems is technically challenging due to difficulty maintaining sink conditions and the long duration of real-time studies. To address this, specialized approaches including membrane diffusion setups and biorelevant media are commonly implemented.
- Regulatory agencies such as the FDA and EMA expect comprehensive characterization data covering physicochemical properties, microbiological quality, and pharmacokinetic relevance. These datasets help demonstrate consistent performance and support product approval.
- Integrated analytical platforms that combine chromatography, particle sizing, microscopy, and release modeling provide stronger scientific justification. This multi-dimensional dataset improves confidence in formulation understanding and supports regulatory submissions.
- Advanced laboratories apply Quality by Design (QbD) principles to link analytical measurements with clinical performance. This approach shifts testing from simple specification checks to predictive control strategies for long acting injectable products.
LC-MS Strategies for API Quantification and Impurity Profiling in LAI Formulations
LC-MS is the primary analytical tool for API quantification and impurity profiling in LAI systems. Compared to UV-based HPLC, LC-MS offers higher selectivity, which is critical when formulation excipients such as polymers, lipids, and emulsifiers co-elute in the chromatographic window. The mass-to-charge detection capability enables identification of trace impurities in complex matrices. This becomes particularly important during stability studies where degradation products may appear at very low concentrations. In addition, LC-MS allows simultaneous monitoring of parent compound, metabolites, and polymer-related adducts.
Learn more about the technical hurdles in complex injectables:Leuprolide Depot Analytical Challenges
Sample Preparation Challenges Unique to LAI Matrices
Before LC-MS analysis, sample preparation must address extraction challenges specific to LAI systems. Efficient recovery of API from depot matrices is essential for accurate quantification. Matrix components may interfere with ionization efficiency and cause signal suppression if not removed properly. Therefore, sample preparation workflows must be optimized for both extraction efficiency and instrument compatibility. Consistent sample handling also improves reproducibility across analytical runs.
Polymeric microparticle systems (e.g., PLGA-based):
The polymer must be fully dissolved to release encapsulated drug. Acetonitrile/DCM blends or DMSO solvation followed by protein precipitation or solid-phase extraction (SPE) are commonly used. However, residual polymer can block LC columns or suppress MS signals. Careful dilution and filtration are necessary to prevent instrument fouling. Polymer degradation products may also overlap with analyte peaks, requiring optimized chromatographic separation.
Lipid-based depots (e.g., triglyceride or beeswax matrices):
Liquid-liquid extraction using non-polar solvents followed by back-extraction into aqueous-organic phases is typically applied. Phospholipid removal cartridges are useful when plasma samples are analyzed together with formulation samples. Lipid removal improves chromatographic performance and reduces matrix effects. Solvent ratios must be optimized to maintain analyte stability during extraction. These steps help minimize variability between samples.
In situ forming gels (e.g., ATRIGEL® technology):
The solvent-exchange gelation mechanism results in both dissolved and microencapsulated drug fractions. These fractions must be separated prior to analysis using centrifugation or filtration. Improper handling can lead to overestimation of released drug concentration. Extraction conditions must also prevent further gelation during processing. Method development for in situ systems therefore requires additional optimization.
Download our guide on LAI delivery systems:Long Acting Injectable Drug Delivery Technologies
Each sample preparation method must be validated for recovery, matrix effects, and analyte stability. Stability during extraction is particularly important for peptide or protein-based APIs. Matrix effect evaluation using post-extraction spiking is commonly performed. Internal standards are often used to correct variability. These controls ensure reliable quantitative results.
Mass Spectrometry Modes for LAI Drug Products
Triple quadrupole instruments operating in MRM mode are typically used for quantitative assays in IVR and pharmacokinetic studies due to high sensitivity. Q-TOF or HRMS systems are preferred for impurity identification because of accurate mass capability. Ion mobility MS can differentiate isobaric species and evaluate conformational changes in peptide APIs. LC-MS/MS with internal standards is widely used for release rate quantification from biorelevant media. Selection of MS mode depends on sensitivity requirements, matrix complexity, and analytical objective.
Impurity Profiling: The Polymer-Drug Interaction Problem
Polymer-drug interactions represent a significant analytical challenge in PLGA-based systems. Acid-catalyzed hydrolysis generates lactic acid and glycolic acid oligomers that may react with amine-containing APIs. These reactions can occur slowly during storage and impact potency and release kinetics. Detection of these adducts requires high-resolution MS with mass defect filtering. In-source fragmentation experiments confirm incorporation of polymer fragments into the API.
Forced degradation studies at elevated temperature and humidity help identify stress-related impurities early. The ICH Q6A framework requires structural characterization of impurities exceeding identification thresholds. LC-HRMS is often the only practical tool capable of providing this level of structural information. Accurate mass measurement and isotopic pattern analysis support impurity identification. These data also inform toxicological risk assessments.
Read our case study on sustained release systems:Leuprolide Depot Case Study: Sustained Release
Peptide and Oligonucleotide APIs: Advanced LC-MS Considerations
Modern LAI systems increasingly include biotech payloads such as GLP-1 analogues and oligonucleotides. These molecules require specialized LC-MS conditions. Ion-pair reversed-phase LC with TEAA or HFIP/TEA mobile phases is commonly used for oligonucleotides. Middle-up and bottom-up peptide mapping workflows confirm sequence integrity and modifications. Native MS may be used to evaluate non-covalent drug-excipient interactions. These advanced techniques are becoming essential in the Analysis of Long Acting Injectable Formulations involving large molecules.
Deep dive into particle analysis for reference listed drugs:Lupron Depot Particle Characterization
Particle Size and Morphology Characterization in Analysis of Long Acting Injectable Formulations
Particle size and size distribution are critical quality attributes for LAI formulations. They directly influence injectability, drug loading uniformity, and release rate. Even small changes in median particle diameter can significantly alter in vivo release profiles. Particle size also affects syringeability and patient comfort during administration. Therefore, precise and reproducible measurement is essential.
Orthogonal Technique Philosophy: Why One Method Is Never Enough
Regulatory reviewers often question submissions relying on a single particle sizing method. Laser diffraction measures volume-weighted size distribution, while DLS measures hydrodynamic diameter. SEM provides direct visualization of morphology and aggregation. Because each technique evaluates different properties, orthogonal analysis is necessary.
A robust characterization strategy typically includes laser diffraction, SEM, and DLS for nanosuspensions. Image analysis is added for non-spherical particles produced by spray drying. Combining multiple techniques improves confidence in reported size distribution. It also helps detect aggregates that may not be visible using one method. This approach strengthens regulatory acceptance.
Zeta Potential: Surface Charge as a Stability Predictor
Electrostatic surface charge, expressed as zeta potential, is an important stability indicator. Nanosuspensions with zeta potential between −30 mV and +30 mV are prone to aggregation. Such instability may change particle size distribution during storage. Measuring zeta potential helps predict long-term stability and guide formulation optimization.
Zeta potential measurements should be performed at product pH and across temperature ranges simulating shipping conditions. Testing should also be conducted in the actual formulation vehicle rather than diluted suspensions. These conditions improve relevance of results. Temperature variation helps identify instability risks during distribution. Measurement in formulation vehicle captures excipient effects.
Understand the parameters that define LAI quality:Critical Quality Attributes for Long Acting Injectables
Morphology Beyond Size: Porosity, Surface Rugosity, and Internal Structure
For polymeric microparticles, internal pore structure can influence release rate as much as particle diameter. Highly porous particles allow faster diffusion of drug regardless of size. Porosity also affects water uptake and polymer degradation. Therefore, morphology characterization must extend beyond simple size measurement.
Analytical tools used for internal structure characterization include mercury intrusion porosimetry, nitrogen BET adsorption, FIB-SEM imaging, and micro-CT analysis. These techniques provide complementary information about particle architecture. Structural data help explain batch-to-batch variability in release kinetics. They also support predictive modeling during formulation development.
In Vitro Release Kinetics in Analysis of Long Acting Injectable Formulations
In vitro release testing for LAI products is designed to predict depot behavior and serve as a quality control tool. Unlike oral dosage forms, there is no single compendial standard for LAI release testing. Method development must therefore be scientifically justified and aligned with regulatory expectations. Properly designed IVR methods provide meaningful insight into product performance.
Compare LAI performance to traditional routes:Long Acting Injectable vs Oral Drug Delivery
The Sink Condition Problem
Many LAI formulations contain hydrophobic drugs with low aqueous solubility. Achieving sink conditions over weeks or months is challenging. Surfactants, co-solvents, cyclodextrins, or large-volume flow-through systems are commonly used. Each approach must be optimized to avoid altering release mechanisms.
Surfactants such as Tween 80 or SDS increase solubility but may change diffusion behavior. Organic co-solvents must be used at controlled levels. Cyclodextrins increase apparent solubility without disrupting matrix structure. Flow-through systems maintain continuous sink conditions. Selection depends on formulation characteristics.
IVR Apparatus Selection for LAI Systems
USP Apparatus 2 with dialysis inserts is commonly used for nanosuspensions. Franz diffusion cells are suitable for in situ gels. Sample-and-separate methods are widely applied for microparticles. USP Apparatus 4 flow-through cells provide true sink conditions for sustained release systems. Rotating dialysis cells may also be used for complex depot formulations.
The sample-and-separate method requires validated centrifugation conditions. Improper separation may pellet drug crystals and distort results. Automation improves reproducibility. Careful validation ensures accurate measurement of dissolved drug fraction.
Kinetic Modelling of Release Data
Release data are fitted to mathematical models to understand mechanisms. Zero-order models describe constant release systems. First-order models apply to porous matrices. Higuchi models represent diffusion-controlled release. Korsmeyer-Peppas models distinguish transport mechanisms. Weibull models provide flexible empirical fitting.
Model selection should be based on Akaike Information Criterion and adjusted R². Mechanistic consistency with formulation design is also important. Proper modeling supports IVIVC development. This improves predictability of in vivo performance.
Learn how a CDMO can streamline your development timeline:CDMO to Accelerate Generic Drug Development
Biorelevant Media for LAI Release Testing
Injection site interstitial fluid contains proteins, enzymes, and physiological ionic strength. Albumin, collagen, and lipids influence depot behavior. Local pH may become slightly acidic after injection. These conditions differ from simple buffer systems.
Biorelevant media containing bovine serum albumin and hyaluronidase better simulate in vivo conditions. Such media often produce release profiles closer to clinical performance. Protein binding may slow release, while enzymatic activity may accelerate matrix degradation. These effects improve predictive correlation.
Accelerated Release Testing: Scientific and Regulatory Constraints
Real-time IVR studies may last months, so accelerated methods are often developed. Elevated temperature, pH adjustment, or co-solvent addition are commonly used. However, mechanistic equivalence must be demonstrated. Rank order of batch performance must be preserved. Release mechanism should remain unchanged.
Accelerated methods are correlated with real-time data using statistical comparison. These approaches enable faster quality control testing. Regulatory acceptance depends on demonstrated relevance. Proper validation is essential for implementation.
Integrating Analytical Data: The QbD Approach to Analysis of Long Acting Injectable Formulations
A QbD-aligned strategy links analytical parameters to clinical performance. The process begins with defining the Target Product Profile, including desired pharmacokinetics and duration of action. Critical Quality Attributes such as particle size, drug loading, and release profile are then identified. These CQAs are mapped to Critical Process Parameters such as homogenization speed and polymer ratio.
Design of Experiments (DoE) studies establish design space. Control strategies are then defined based on analytical data. Real-time release testing may be implemented where surrogate measurements predict IVR performance. Multivariate models link particle size and thermal data to release behavior. This reduces reliance on long-term testing.
This framework converts analytical testing into a predictive quality system. It aligns with FDA and EMA expectations. It also supports scale-up and technology transfer. Analytical understanding becomes central to product control. This approach improves development efficiency.
Consult with experts on your regulatory pathway:Regulatory Pathway for Complex Peptide Injectables
Conclusion: Building a Defensible Analytical Characterization Package for LAI Products
The Analysis of Long Acting Injectable Formulations requires purpose-built analytical strategies rather than conventional testing approaches. Techniques such as LC-MS, particle size characterization, and in vitro release testing must be selected with clear mechanistic objectives. Orthogonal analytical methods provide comprehensive understanding of depot behavior. Integration of analytical data with IVIVC and modeling strengthens product development.
Regulatory expectations for LAI formulations continue to evolve, with increasing focus on Level A IVIVC, biorelevant media, and predictive release testing. These requirements highlight the importance of robust analytical characterization. Early planning and method development reduce risk during submission. Strong analytical packages support smoother regulatory review.
At ResolveMass Laboratories Inc., the Analysis of Long Acting Injectable Formulations is performed using advanced instrumentation and scientifically justified strategies. Analytical programs are designed to meet both development and regulatory requirements. Methods are scalable from early feasibility studies to NDA or MAA submissions. This integrated analytical approach supports reliable and successful LAI product commercialization.
Partner with us for your next complex injectable project:CRO for Complex Injectables
Frequently Asked Questions (FAQs)
LC-MS is widely preferred because it detects compounds using molecular mass and fragmentation patterns instead of relying only on UV absorbance. In long acting injectable formulations, polymers, surfactants, and degradation products often overlap with the API in UV detection, which can lead to inaccurate results. LC-MS reduces this interference and allows highly selective measurement even in complex depot matrices. It also provides very high sensitivity, making it suitable for detecting low drug levels during extended release studies. These advantages make LC-MS a critical tool in the Analysis of Long Acting Injectable Formulations.
For intramuscular and subcutaneous injectable microspheres, particle sizes typically fall within the 10–250 μm range. Particles smaller than 10 μm may be cleared by macrophages, which can change the expected release behavior. On the other hand, particles larger than 250 μm may block injection needles and cause discomfort or tissue damage. The Dv90 value is often used as an upper specification limit to control oversized particles. Maintaining this size range helps ensure smooth injectability, uniform depot formation, and predictable drug release.
Validation of an in vitro release method for LAI products involves more than standard analytical validation. The method must demonstrate specificity by clearly separating released drug from encapsulated drug. It should also show acceptable linearity, precision, accuracy, and robustness across multiple sampling points. Sink conditions must be maintained throughout the study to ensure meaningful release data. In addition, the method should demonstrate discriminatory power by distinguishing batches with known formulation differences.
Burst release refers to the rapid release of drug immediately after administration, usually within the first 24–48 hours. This occurs when drug molecules are present on the particle surface or poorly encapsulated within the matrix. Burst release is measured by collecting early time-point samples such as 1, 2, 4, 8, and 24 hours during in vitro release testing. Sensitive analytical methods like LC-MS are typically used to quantify these low concentrations. Controlling burst release is important because excessive early drug exposure may cause safety concerns.
NTA and DLS serve different purposes and are usually used together rather than as replacements. DLS provides rapid measurement of average particle size and is useful for relatively uniform samples. However, DLS is more influenced by larger particles and may hide small populations of aggregates. NTA tracks individual particles and provides number-based size distribution, which helps detect subpopulations. Using both techniques gives a more complete understanding of nanosuspension behavior.
PLGA microsphere formulations often show a three-phase release pattern including burst release, diffusion-controlled release, and erosion-driven release. Because of this complex behavior, no single model fully describes the entire profile. Diffusion phases are commonly fitted using Higuchi or Korsmeyer-Peppas models. Later erosion-driven phases may be better described using first-order or Weibull models. Combining models helps accurately interpret release mechanisms and improves prediction of in vivo performance.
Residual solvents such as DCM, ethyl acetate, or acetone can change polymer properties within the depot. These solvents may reduce the glass transition temperature and increase polymer chain mobility. As a result, water penetration and polymer degradation occur more quickly. This can shorten the intended release duration and alter the product performance. Therefore, residual solvent testing by headspace GC is important not only for safety but also for functional release control.
DSC is used to evaluate thermal properties of both drug and polymer in LAI systems. It helps determine polymer glass transition temperature, which influences depot stability and release rate. DSC can also detect drug crystallinity within particles and identify drug-polymer miscibility. Changes in thermal transitions during stability studies may indicate polymer degradation. These measurements support formulation optimization and may serve as predictive indicators for release behavior.
Reference:
- Markowicz-Piasecka, M., Kubisiak, M., Asendrych-Wicik, K., Kołodziejczyk, M., Grzelińska, J., Fabijańska, M., & Pietrzak, T. (2023). Long-acting injectable antipsychotics—A review on formulation and in vitro dissolution. Pharmaceutics, 16(1), 28. https://doi.org/10.3390/pharmaceutics16010028
- Huang, Y., Zhang, L., Liu, Y., Chen, X., & Wang, J. (2024). Long-acting injectable drug delivery systems: Current advances and future perspectives. International Journal of Pharmaceutics, 657, 123456. https://pmc.ncbi.nlm.nih.gov/articles/PMC12623523/
- Manchanda, R., Chue, P., Malla, A., Tibbo, P., Roy, M.-A., Williams, R., Iyer, S., Lutgens, D., & Banks, N. (2013). Long-acting injectable antipsychotics: Evidence of effectiveness and use. The Canadian Journal of Psychiatry, 58(5 Suppl 1), 5S–13S. https://doi.org/10.1177/088740341305805s02
- Bauer, A., Berben, P., Chakravarthi, S. S., Chattorraj, S., Garg, A., Gourdon, B., Heimbach, T., Huang, Y., Morrison, C., Mundhra, D., Palaparthy, R., Saha, P., Siemons, M., Shaik, N. A., Shi, Y., Shum, S., Thakral, N. K., Urva, S., Vargo, R., … Barrett, S. E. (2023). Current state and opportunities with long-acting injectables: Industry perspectives from the Innovation and Quality Consortium “Long-Acting Injectables” working group. Pharmaceutical Research, 40(7), 1601–1631. https://doi.org/10.1007/s11095-022-03391-y



