
Introduction to Bioanalytical Method Development
Bioanalytical Method Development is the systematic process of designing, optimizing, and preparing analytical methods to quantitatively measure drugs, metabolites, and biomarkers in biological matrices such as plasma, serum, urine, or tissue.
In regulated drug development programs, method development directly impacts data quality, regulatory acceptance, and decision-making throughout discovery, preclinical, and clinical phases.
At ResolveMass Laboratories Inc.,method development is driven by scientific expertise, regulatory knowledge, and real-world experience supporting global pharmaceutical and biotech clients.
Summary
- Bioanalytical Method Development ensures accurate, precise, and reproducible measurement of drugs, metabolites, and biomarkers.
- It is a foundational step for PK, TK, bioequivalence, and biomarker studies across preclinical and clinical phases.
- Robust method development reduces regulatory risk and supports FDA, EMA, and ICH submissions.
- Advanced platforms such as LC–MS/MS are central to modern bioanalytical method development.
- Partnering with an experienced CRO like ResolveMass Laboratories Inc. improves timelines, compliance, and data integrity.
1: What Is Bioanalytical Method Development?
Method Development is the scientific process of creating a sensitive, selective, and reproducible analytical method for measuring analytes in biological samples.
It typically involves:
- Selection of analytical technique
- Sample preparation optimization
- Chromatographic separation
- Detection and quantification strategy
- Method robustness evaluation
Why Bioanalytical Method Development Matters
Without a properly developed bioanalytical method:
- PK and TK data may be unreliable
- Dose-exposure relationships can be misinterpreted
- Regulatory submissions may be delayed or rejected
2: Why Bioanalytical Method Development Is Critical in Drug Development
Method Development is essential because it directly supports:
- Pharmacokinetic (PK) studies
- Toxicokinetic (TK) studies
- Bioavailability and bioequivalence studies
- Biomarker and ligand-binding assays
- Clinical trial sample analysis
Key Benefits
- Ensures accurate drug concentration measurement
- Supports regulatory compliance
- Reduces study repeat risks
- Enables confident go/no-go decisions
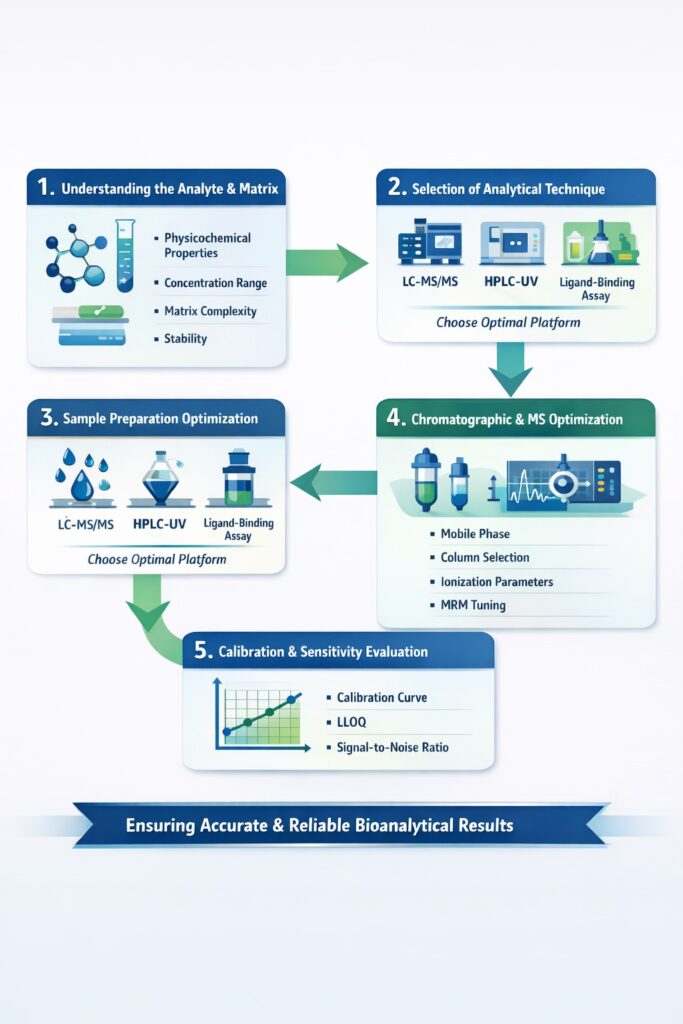
3: Key Stages of Bioanalytical Method Development
Each stage of Method Development must be approached systematically to ensure regulatory-ready performance.
1. Understanding the Analyte and Matrix
The first step in method development is understanding:
- Physicochemical properties of the analyte
- Expected concentration range
- Biological matrix complexity
- Stability considerations
This knowledge guides method selection and optimization.
2. Selection of Analytical Technique
The analytical platform defines sensitivity and specificity.
| Technique | Application |
|---|---|
| LC–MS/MS | Small molecules, high sensitivity |
| HPLC-UV | Higher concentration analytes |
| Ligand-Binding Assays | Large molecules, biologics |
Liquid Chromatography-Mass Spectrometry (LC-MS) remains the gold standard for small-molecule analysis.
3. Sample Preparation Optimization
Sample preparation is a critical determinant of method performance.
Common approaches include:
- Protein precipitation
- Liquid-liquid extraction
- Solid-phase extraction (SPE)
Well-optimized preparation improves:
- Recovery
- Matrix effect control
- Method reproducibility
4. Chromatographic and Mass Spectrometric Optimization
In method development, chromatographic separation ensures selectivity, while MS parameters ensure sensitivity.
Optimization includes:
- Mobile phase composition
- Column selection
- Ionization mode
- MRM transitions
5. Calibration Curve and Sensitivity Evaluation
A well-developed method must demonstrate:
- Linearity across concentration range
- Lower limit of quantification (LLOQ)
- Acceptable signal-to-noise ratio
This ensures the method is fit for PK and clinical sample analysis.
4: Regulatory Considerations in Bioanalytical Method Development
Method Development must align with global regulatory expectations.
Key Regulatory Guidelines
- FDA Bioanalytical Method Validation Guidance (2018)
- EMA Bioanalytical Method Validation Guideline
- ICH M10 Bioanalytical Method Validation
- 21 CFR Part 58 (GLP)
ResolveMass Laboratories Inc. integrates regulatory thinking at the method development stage, minimizing downstream compliance risks.
5: Bioanalytical Method Development vs Method Validation
Although closely related, they serve different purposes.
| Aspect | Method Development | Method Validation |
|---|---|---|
| Objective | Optimize method | Confirm performance |
| Stage | Early | Pre-study |
| Flexibility | High | Controlled |
| Regulatory Submission | No | Yes |
Strong method development simplifies validation and reduces rework.
6: Common Challenges in Bioanalytical Method Development
Even experienced teams face challenges during method development.
Typical Challenges
- Matrix interference
- Low analyte recovery
- Ion suppression/enhancement
- Analyte instability
- Cross-talk in MS detection
How ResolveMass Addresses Them
- Extensive feasibility studies
- Advanced sample cleanup strategies
- Experienced troubleshooting scientists
- State-of-the-art LC–MS/MS platforms
7: Bioanalytical Method Development for Complex Molecules
Modern drug pipelines increasingly include:
- Highly potent compounds
- Polar metabolites
- Combination therapies
Method Development must evolve to support these complexities with enhanced sensitivity and selectivity.
ResolveMass Laboratories Inc. brings proven experience handling complex bioanalytical challenges across therapeutic areas.
8: Why Outsource Bioanalytical Method Development to a Specialized CRO?
Outsourcing Method Development offers strategic advantages.
Key Advantages
- Access to specialized expertise
- Faster method readiness
- Regulatory-compliant documentation
- Cost-effective resource utilization
Why ResolveMass Laboratories Inc.
- Deep experience across PK, TK, and clinical bioanalysis
- GLP-compliant infrastructure
- Regulatory-inspection-ready processes
- Transparent communication and timelines
9: The Role of Experience and Scientific Expertise
True excellence in Method Development comes from hands-on experience.
At ResolveMass Laboratories Inc.:
- Methods are developed by seasoned scientists
- Regulatory expectations guide every decision
- Data integrity and reproducibility are non-negotiable
This practical expertise ensures confidence in every dataset delivered.
10: Future Trends in Bioanalytical Method Development
The future of Method Development is shaped by:
- Increased sensitivity requirements
- Automation and high-throughput workflows
- Integration with biomarker strategies
- Harmonized global regulatory standards
ResolveMass continues to invest in advanced technologies and scientific training to stay ahead of evolving bioanalytical demands.
Conclusion
Bioanalytical Method Development is not just a technical requirement—it is a strategic pillar of successful drug development.
When executed with scientific rigor, regulatory foresight, and real-world experience, it enables:
- Reliable PK and clinical data
- Faster development timelines
- Strong regulatory submissions
ResolveMass Laboratories Inc. delivers bioanalytical method development solutions built on trust, expertise, and proven performance.
Frequently Asked Questions:
Bioanalytical Method Development is the process of designing and optimizing analytical methods to accurately measure drugs, metabolites, or biomarkers in biological matrices such as plasma, serum, or urine for preclinical and clinical studies.
Bioanalytical method development is critical because it ensures accurate, reproducible, and reliable data for pharmacokinetic (PK), toxicokinetic (TK), and clinical studies, supporting regulatory submissions and informed decision-making.
The most commonly used techniques in bioanalytical method development include LC–MS/MS for small molecules, HPLC-UV for higher concentration analytes, and ligand-binding assays for large molecules and biologics.
Bioanalytical method development can be performed in various biological matrices, including plasma, serum, urine, tissue homogenates, cerebrospinal fluid (CSF), and whole blood, depending on study requirements.
Bioanalytical method development focuses on optimizing method conditions, while method validation formally confirms method performance according to regulatory guidelines such as FDA, EMA, and ICH M10.
Reference
- Mohammad Mahdi Moein , Aziza El Beqqali , Mohamed Abdel-Rehim.Bioanalytical method development and validation: Critical concepts and strategies.https://www.sciencedirect.com/science/article/abs/pii/S1570023216308881
- A REVIEW ON BIOANALYTICAL METHOD DEVELOPMENT AND VALIDATION.https://www.researchgate.net/profile/Lokesh-Tijare/publication/316004889_A_review_on_bioanalytical_method_development_and_validation/links/5d8c5276a6fdcc25549a54b5/A-review-on-bioanalytical-method-development-and-validation.pdf
- Bioanalytical Method Development and Validation by HPLC: A Review.https://www.researchgate.net/profile/Pushpa-Latha-18/publication/352834227_Bioanalytical_Method_Development_and_Validation_by_HPLC_A_Review/links/60dbd8d692851ca944974667/Bioanalytical-Method-Development-and-Validation-by-HPLC-A-Review.pdf
- Bioanalytical Method Development and Validation: from the USFDA 2001 to the USFDA 2018 Guidance for Industry.https://www.researchgate.net/publication/326495876_Bioanalytical_Method_Development_and_Validation_from_the_USFDA_2001_to_the_USFDA_2018_Guidance_for_Industry
- Matrix Effect Elimination During LC–MS/MS Bioanalytical Method Development.https://www.tandfonline.com/doi/abs/10.4155/bio.09.117
- Bioanalytical method validation: An updated review.https://www.sciencedirect.com/science/article/abs/pii/S2229470810110048


