Introduction:
Bioanalytical Method Development: A Comprehensive Guide highlights the critical role analytical techniques play in modern pharmaceutical research and life-science innovation. As drug molecules become more complex and biologic therapies continue to expand, accurately measuring compounds within biological matrices has become increasingly essential. Scientists depend on precise bioanalytical methods to understand how drugs behave in the body, assess therapeutic performance, and ensure safety throughout the drug development process.
From early drug discovery to large-scale clinical trials, bioanalytical techniques provide reliable data on drug concentrations, metabolism, and biomarker activity. These insights support regulatory submissions, therapeutic monitoring, and informed scientific decisions. By establishing robust and validated analytical methods, laboratories can ensure that experimental results remain accurate, reproducible, and scientifically dependable.
Share via:
Article Summary
- Bioanalytical method development enables accurate measurement of drugs, metabolites, and biomarkers in biological matrices such as plasma, urine, and tissues.
- These analytical methods are critical for drug discovery, pharmacokinetic studies, clinical research, and regulatory approval processes.
- Key components include selection of appropriate analytical techniques (LC-MS/MS, HPLC, ELISA), biological matrix selection, and rigorous method validation.
- The development process typically follows structured stages including method design, optimization, validation, and application to biological sample analysis.
- Proper sample preparation, calibration standards, and quality control samples ensure reliable and reproducible analytical results.
- With advances such as automation, artificial intelligence, and high-resolution mass spectrometry, bioanalytical methods continue to evolve to support modern pharmaceutical and biopharmaceutical research.
Why Is Bioanalytical Method Development Important?
Bioanalytical methods are integral to ensuring the safety, efficacy, and quality of pharmaceutical and biotechnological products. They provide the foundation for reliable data in drug development and clinical research. Key reasons include:
- Drug Development Support: Accurate measurement of drugs and metabolites ensures proper pharmacokinetic (PK) and pharmacodynamic (PD) profiling.
- Regulatory Compliance: Regulatory authorities like the FDA and EMA require validated methods to confirm data reliability and reproducibility.
- Clinical Research Enhancement: Biomarker quantification enables disease monitoring, diagnosis, and treatment efficacy assessment.
- Quality Assurance: Ensures consistent results in pharmaceutical and clinical applications.
Our bioanalytical services provide the high-quality data necessary to support every phase of your clinical and preclinical research.
Role of Bioanalytical Methods in Modern Drug Discovery
Bioanalytical methods are fundamental tools that support decision-making during early drug discovery. Scientists use these techniques to determine how candidate molecules behave in biological systems before they move into clinical development. Accurate analytical data allows researchers to evaluate parameters such as bioavailability, metabolic stability, and toxicity risks. Without reliable measurement techniques, it would be difficult to identify promising drug candidates or eliminate compounds that may fail later in development.
In addition to measuring drug concentrations, bioanalytical testing helps researchers understand complex biological interactions. For example, researchers can monitor how drug molecules interact with enzymes, receptors, or transport proteins in the body. These insights help scientists optimize molecular structures and improve drug performance. As a result, bioanalytical method development contributes directly to more efficient research pipelines and reduces the risk of costly failures during advanced development stages.
Key Components of Bioanalytical Method Development
Several critical elements form the backbone of bioanalytical method development, ensuring robust and reliable outcomes.
1. Selection of Analytical Techniques
The choice of analytical technique depends on the nature of the analyte and the study objectives. Common methods include:
- Liquid Chromatography-Mass Spectrometry (LC-MS/MS): Highly sensitive and specific for drug quantification.
- High-Performance Liquid Chromatography (HPLC): Widely used for separation and analysis of compounds.
- Enzyme-Linked Immunosorbent Assay (ELISA): Commonly employed for biomarker detection in immunoassays.
Achieve superior precision in your separation science with our professional HPLC analysis services.
2. Biological Matrices
Bioanalytical methods often involve complex biological samples such as:
- Plasma: Ideal for measuring drug concentrations.
- Urine: Useful for metabolite excretion studies.
- Tissue Samples: Suitable for localized drug distribution analysis.
3. Method Validation
Method validation is crucial to ensure accuracy, precision, reproducibility, and specificity. It forms the basis for regulatory acceptance and reliable data generation.
Ensure your protocols meet strict regulatory standards with our analytical method development and validation service.
Importance of Sample Preparation Techniques
Sample preparation is one of the most critical steps in bioanalytical workflows because biological samples contain numerous endogenous compounds that may interfere with analysis. Proteins, lipids, salts, and other biomolecules present in biological matrices can affect detection accuracy if not properly removed. Effective sample preparation techniques help isolate the analyte of interest while minimizing potential interference from these matrix components.
Several strategies are commonly used to prepare biological samples for analysis. Protein precipitation is a quick method used to remove proteins from plasma or serum samples, while liquid-liquid extraction separates analytes based on solubility differences. Solid-phase extraction provides higher purification efficiency and is often used for sensitive analytical assays. By carefully selecting and optimizing these preparation methods, researchers can improve analytical accuracy, sensitivity, and reproducibility.
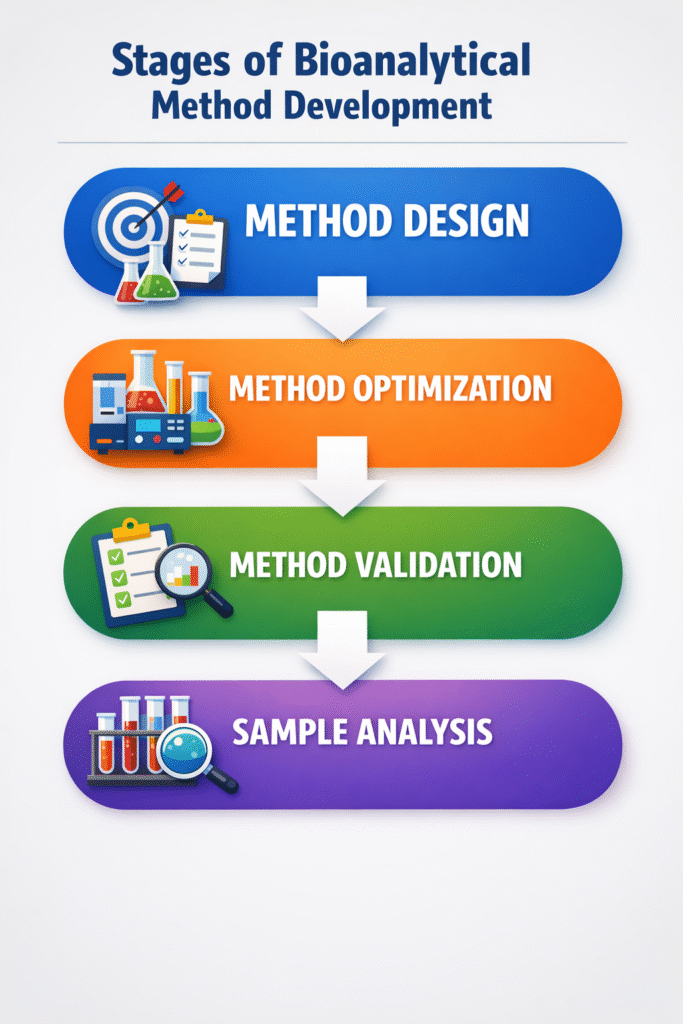
Stages of Bioanalytical Method Development
A structured approach ensures that the developed bioanalytical method is suitable for its intended application.
1. Method Design
- Defining Objectives: Establish the target analyte, matrix, and desired detection limits.
- Choosing Techniques: Select between LC-MS/MS, HPLC, or ELISA based on analyte properties.
2. Method Optimization
- Sample Preparation: Optimize extraction protocols such as protein precipitation, liquid-liquid extraction, or solid-phase extraction.
- Chromatographic Conditions: Fine-tune mobile phase composition, flow rates, and column selection.
- Detection Parameters: Adjust sensitivity and resolution settings for accurate measurements.
3. Method Validation
Validation ensures the method meets predefined criteria for:
- Accuracy and Precision: Reliable and reproducible results.
- Specificity: Minimal interference from biological matrix components.
- Linearity: Establishing a valid calibration curve over the required concentration range.
- Stability: Ensuring the analyte remains stable throughout sample collection, storage, and analysis.
4. Application to Sample Analysis
Once validated, the method is applied to analyze biological samples in preclinical or clinical studies.
Importance of Calibration and Quality Control Samples
Calibration standards and quality control samples are essential for ensuring the reliability of bioanalytical measurements. Calibration curves establish the relationship between analyte concentration and instrument response, allowing researchers to accurately quantify unknown samples. Without properly prepared calibration standards, analytical data may become unreliable or inconsistent.
Quality control samples are used throughout analytical runs to verify that the method continues to perform as expected. These samples are prepared at different concentration levels to monitor accuracy, precision, and instrument performance. By evaluating these controls during analysis, scientists can quickly identify potential errors, instrument drift, or sample processing issues. This systematic monitoring helps maintain confidence in analytical results and supports regulatory compliance.
Common Challenges in Bioanalytical Method Development
- Matrix Interference: Complex biological matrices can introduce variability.
- Detection Sensitivity: Achieving low detection limits for trace analytes.
- Analyte Stability: Degradation during collection or processing.
Addressing Matrix Effects in Bioanalytical Analysis
Matrix effects occur when components present in biological samples influence the analytical signal of the target analyte. These effects are particularly common in techniques such as LC-MS/MS, where endogenous substances can suppress or enhance ionization during mass spectrometric detection. Such interference may lead to inaccurate quantification if not properly controlled during method development.
To minimize matrix effects, researchers often employ strategies such as matrix-matched calibration curves, improved chromatographic separation, or the use of stable isotope-labeled internal standards. Careful evaluation of matrix interference during validation studies helps ensure that analytical methods remain robust across different biological samples. Addressing matrix effects early in development significantly improves method reliability and data accuracy.
Identify and quantify potential contaminants with high sensitivity using our impurity profiling using LCMS.
Overcoming Challenges
- Implement robust sample preparation techniques.
- Use advanced instruments like LC-MS/MS for improved sensitivity.
- Conduct thorough stability studies to optimize handling and storage.
Protect patient safety with our specialized nitrosamine analysis to detect trace levels of genotoxic impurities.
Applications of Bioanalytical Method Development
Bioanalytical methods play a pivotal role in various scientific and industrial applications:
- Pharmacokinetics and Pharmacodynamics: Quantifying drug absorption, distribution, metabolism, and excretion.
- Therapeutic Drug Monitoring (TDM): Ensuring drug levels stay within therapeutic windows.
- Biomarker Discovery: Facilitating disease diagnosis and treatment monitoring.
- Bioequivalence Studies: Comparing generic and branded drug formulations.
Role in Biologics and Biopharmaceutical Research
As biologic therapies such as monoclonal antibodies, peptides, and nucleic acid-based drugs gain popularity, bioanalytical methods have adapted to measure these complex molecules. Unlike small-molecule drugs, biologics often require specialized analytical approaches due to their larger size, structural complexity, and potential immunogenicity. Techniques such as ligand-binding assays and hybrid LC-MS methods are commonly used to analyze these therapeutic agents.
Bioanalytical testing also plays a key role in evaluating the pharmacokinetics and immunogenic responses of biologic therapies. For instance, assays may be designed to detect anti-drug antibodies that could impact treatment effectiveness. Accurate measurement of these parameters helps researchers better understand therapeutic behavior and optimize dosing strategies. As biologics continue to expand within the pharmaceutical industry, bioanalytical method development remains essential for supporting their safe and effective use.
Our gel permeation chromatography (GPC) analysis provides essential data for characterizing the molecular weight and distribution of complex polymers.
Workflow for Bioanalytical Method Development
Below is a simplified visual representation of the development process:
Define Objectives
↓
Select Analytical Technique
↓
Optimize Parameters
↓
Validate Method
↓
Apply to Sample Analysis
Data Integrity and Documentation in Bioanalytical Studies
Maintaining data integrity is a critical requirement in bioanalytical research, particularly when analytical results are used for regulatory submissions. Every stage of the analytical process—from sample collection to data reporting—must follow well-documented procedures to ensure transparency and reproducibility. Proper documentation helps verify that experiments were conducted according to validated protocols and that analytical results are trustworthy.
Regulatory agencies emphasize strict compliance with Good Laboratory Practice (GLP) and data integrity guidelines. Laboratories must maintain detailed records of instrument settings, sample preparation steps, calibration data, and validation results. Digital data management systems are increasingly used to store and track analytical data securely. By implementing strong documentation practices, organizations can maintain regulatory compliance and support the credibility of their scientific findings.
Future Trends in Bioanalytical Method Development
The field is rapidly evolving with advancements in technology and methodology. Key trends include:
- Automation: High-throughput systems to improve efficiency and reduce human error.
- AI Integration: Optimizing method development and enhancing data interpretation.
- Miniaturization: Microfluidic devices for low-volume sample analysis.
- Advanced Techniques: High-resolution mass spectrometry for multiplex analysis.
Conclusion
Bioanalytical method development remains a cornerstone of pharmaceutical research, biotechnology innovation, and clinical diagnostics. Through carefully designed analytical strategies, scientists can accurately measure drugs, metabolites, and biomarkers within complex biological matrices. These measurements provide valuable insights into drug behavior, therapeutic effectiveness, and patient safety throughout the development process.
As the pharmaceutical industry continues to evolve with new therapeutic modalities and advanced analytical technologies, the importance of robust bioanalytical methods will only increase. By combining precise instrumentation, optimized workflows, and rigorous validation practices, researchers can generate reliable data that supports regulatory approvals and scientific progress. Ultimately, effective bioanalytical method development contributes to the successful discovery, development, and monitoring of life-saving therapies.
Contact Us for Expert Solutions
For further information or to discuss your specific bioanalytical requirements, ResolveMass Laboratories Inc. is here to help. We offer state-of-the-art solutions tailored to meet your research and regulatory needs.
Frequently Asked Questions
Bioanalytical method development is essential because it helps researchers measure drug concentrations and understand how medicines behave inside the body. Accurate analytical data supports pharmacokinetic and pharmacodynamic studies, which guide dosing strategies and safety evaluations. Reliable methods also help generate data required for regulatory approval.
The development process generally includes method design, optimization, validation, and sample analysis. Researchers first define the analytical objectives and select appropriate techniques. The method is then optimized for accuracy and sensitivity before being validated to ensure it produces reliable and reproducible results.
Researchers often encounter challenges such as matrix interference, low analyte concentration, and analyte instability during analysis. Biological samples contain many endogenous compounds that may affect detection accuracy. Proper sample preparation techniques and optimized analytical conditions help overcome these difficulties.
Bioanalytical methods play a crucial role in clinical studies by measuring drug levels and monitoring biomarker responses in patient samples. These measurements help determine how effectively a drug works and whether it is safe for patients. The data generated also assists researchers in refining dosing regimens and evaluating treatment outcomes.
Reference:
- Bhadra, S., Mukherjee, B., & Bhattacharya, S. (2024). Bioanalytical method development, in-vivo pharmacokinetic evaluation, and ex-vivo platelet aggregation inhibition activity of a novel solid dispersion formulation of ticagrelor. Journal of Pharmaceutical Analysis. https://pmc.ncbi.nlm.nih.gov/articles/PMC11839408/
- Nagavi, J. B., Ghosh, S., & Anantharaju, P. G. (2024). RP-HPLC bioanalytical method development and validation for the simultaneous estimation of metformin and levothyroxine in human plasma: Drug interaction studies. International Journal of Pharmaceutical Sciences and Research, 15(4), 1190–1197. https://doi.org/10.13040/IJPSR.0975-8232.15(4).1190-97
- Jagtap, J., Borhade, A., Mhetar, M., & Chavan, R. (2026). A mini review on bioanalytical method development and validation. International Journal of Pharmaceutical Sciences and Research, 17(2), 437–446. https://doi.org/10.13040/IJPSR.0975-8232.17(2).437-46



