
Introduction:
A well-planned Bioanalytical Strategy Drug Development framework is critical for the success of early drug development programs. Without reliable bioanalytical data, pharmaceutical and biotech companies cannot accurately measure drug concentration, evaluate pharmacokinetics (PK), or understand pharmacodynamic (PD) responses using advanced bioanalytical services.
In many cases, promising drug candidates fail not because the molecule lacks therapeutic potential, but because bioanalytical strategies were poorly designed or implemented too late in development. This leads to unreliable data, regulatory setbacks, and costly delays. Understanding why bioanalysis is important in drug development can help companies avoid these risks.
Early integration of a strong Bioanalytical Strategy Drug Development approach ensures that drug developers obtain precise, reproducible, and regulatory-compliant analytical data throughout the development lifecycle through bioanalytical services in drug development.
Share via:
Summary:
- A strong Bioanalytical Strategy Drug Development plan is essential for accurate PK/PD data generation in early drug programs.
- Many early drug development failures occur due to inadequate bioanalytical planning, poor assay sensitivity, or unreliable data.
- Without robust analytical workflows such as LC-MS/MS bioanalytical services and bioanalytical quantification, developers cannot correctly evaluate drug exposure, efficacy, safety, and biomarker response.
- Early integration of Bioanalytical Strategy Drug Development reduces regulatory risks, accelerates decision-making, and improves success rates.
- Experienced bioanalytical CROs provide validated assays, biomarker analysis, and regulatory-compliant data required for IND-enabling bioanalytical studies.
- Strategic bioanalysis helps companies avoid costly delays and ensures confident progression from preclinical to clinical stages.
1: Why Bioanalytical Strategy Drug Development Is Critical in Early Drug Programs
A strong Bioanalytical Strategy Drug Development ensures accurate measurement of drug concentrations and biological responses during early drug research using specialized bioanalytical laboratory services.
Drug development depends heavily on bioanalytical data to answer critical questions such as:
- How much drug is present in biological samples?
- How long does the drug remain in the body?
- Does the drug interact with its target?
- What exposure level produces therapeutic effect?
Reliable answers often require validated PK/PD bioanalysis and clinical bioanalytical services.
Without reliable answers to these questions, developers cannot confidently advance candidates to clinical trials.
Core Elements of a Bioanalytical Strategy Drug Development Plan
| Component | Purpose |
|---|---|
| Bioanalytical Method Development | Create sensitive analytical assays using bioanalytical method development |
| Method Validation | Ensure accuracy, precision, and reproducibility through bioanalytical method validation |
| PK/PD Analysis | Understand drug exposure and biological response using PK-TK bioanalysis |
| Biomarker Quantification | Evaluate therapeutic activity through biomarker bioanalytical services |
| Regulatory Compliance | Meet FDA/EMA guidelines through regulated bioanalytical services |
These components form the backbone of a successful Bioanalytical Strategy Drug Development framework.
2: Common Reasons Early Drug Development Programs Fail Without Bioanalytical Strategy
Early drug programs frequently fail when bioanalytical considerations are overlooked or delayed. Many of these failures stem from challenges such as bioanalytical matrix effects, poor assay design, or data integrity issues.
Below are some of the most common failure points.
1. Poorly Designed Bioanalytical Strategy Drug Development Plan
Many companies underestimate the complexity of bioanalysis and treat it as a late-stage activity rather than a core development component.
Without a well-defined Bioanalytical Strategy Drug Development, teams often face issues frequently discussed in common bioanalytical mistakes.
These include:
- Inadequate assay sensitivity
- Poor sample preparation methods
- Insufficient method validation
- Inconsistent analytical results
These challenges often arise during rapid bioanalytical method development or when facing challenges in bioanalytical method development.
This leads to unreliable PK data and difficulty interpreting study outcomes.
2. Inadequate Assay Sensitivity
Modern drug candidates often require ultra-sensitive analytical methods, especially for biologics, peptides, and oligonucleotides.
Sensitive workflows such as high-sensitivity bioanalysis and LC-MS bioanalysis for oligonucleotides are increasingly required.
If analytical methods cannot detect drug concentrations at very low levels, developers may encounter:
- False negative results
- Misinterpretation of drug exposure
- Incorrect dosing decisions
Technologies such as LC-MS/MS bioanalysis of xenobiotics help overcome these challenges.
A robust Bioanalytical Strategy Drug Development ensures the use of highly sensitive technologies such as LC-MS/MS or high-resolution mass spectrometry to achieve accurate quantification.
3. Lack of Reliable Pharmacokinetic Data
Pharmacokinetic analysis is essential for determining drug absorption, distribution, metabolism, and excretion.
Without strong Bioanalytical Strategy Drug Development, PK data may suffer from:
- High variability
- Poor assay reproducibility
- Limited sampling analysis
- Incorrect exposure estimation
These issues are often addressed through toxicokinetic bioanalysis and high-throughput bioanalysis.
Reliable PK data are essential for dose selection and regulatory submissions.
4. Poor Biomarker Measurement
Biomarkers provide evidence that a drug is interacting with its biological target.
Without validated biomarker workflows such as biomarker bioanalytical services or proteomics bioanalytical services, biomarker analysis may fail due to:
- Lack of validated assays
- High biological variability
- Sample instability
- Insufficient analytical sensitivity
These measurements are particularly important for complex therapies such as cell and gene therapy bioanalysis or biosimilar bioanalysis.
Accurate biomarker measurement is particularly important for precision medicine and targeted therapies.
5. Regulatory Compliance Challenges
Regulatory agencies expect bioanalytical methods to meet strict validation standards.
A weak Bioanalytical Strategy Drug Development can lead to:
- Incomplete validation documentation
- Non-compliance with regulatory guidelines
- Data integrity issues
- Delays in IND or clinical trial approvals
Maintaining data integrity in bioanalytical studies and strong bioanalytical stability testing is essential for regulatory success.
Working with experienced bioanalytical laboratories helps ensure compliance with global regulatory expectations.

3: How a Strong Bioanalytical Strategy Drug Development Improves Success Rates
Implementing a strong Bioanalytical Drug Development early in drug research dramatically increases development efficiency and success rates.
Organizations increasingly rely on advanced bioanalytical strategies for complex drug modalities and AI in bioanalysis to improve analytical performance.
Key advantages include:
1. Faster Decision Making
Reliable bioanalytical data allows researchers to quickly determine:
- Whether a drug candidate is viable
- Optimal dosing strategies
- Target engagement confirmation
2. Reduced Development Risk
A robust Bioanalytical Strategy Drug Development reduces the likelihood of unexpected analytical failures during critical development phases.
3. Improved Data Quality
High-quality analytical data improves:
- PK modeling
- Exposure-response relationships
- Clinical trial design
4. Better Regulatory Readiness
Regulatory agencies prioritize validated analytical methods and reliable datasets.
Strong bioanalysis ensures smoother regulatory interactions.
4: Key Technologies Supporting Bioanalytical Strategy Drug Development
Modern drug development relies on advanced analytical technologies.
These technologies are commonly used by bioanalytical CRO services for PK and TK and specialized integrated chemistry and bioanalytical CROs.
Below are commonly used platforms.
| Technology | Application |
|---|---|
| LC-MS/MS | Quantification of small molecules |
| High-Resolution Mass Spectrometry | Ultra-trace detection |
| Ligand Binding Assays | Biologics and antibodies |
| Hybrid LC-MS Methods | Peptides and oligonucleotides |
| Multiplex Biomarker Platforms | Target engagement and PD analysis |
These technologies enable accurate and reproducible results within a comprehensive Bioanalytical Drug Development framework.
5: Role of Experienced Bioanalytical CROs in Bioanalytical Strategy Drug Development
Partnering with a specialized bioanalytical CRO can significantly strengthen Bioanalytical Strategy Drug Development.
Sponsors often rely on outsourced bioanalysis for drug development or bioanalytical CRO partnerships to access expert scientists and advanced instrumentation.
For biotech startups, affordable bioanalytical services for biotech startups and cost-effective bioanalytical services help accelerate development while managing budgets.
Experienced CROs provide:
- Expert method development and validation
- Advanced analytical instrumentation
- Regulatory-compliant workflows
- Experienced analytical scientists
- End-to-end bioanalysis support
Companies like ResolveMass Laboratories Inc. support pharmaceutical and biotechnology organizations by delivering high-quality bioanalytical data across preclinical and clinical development stages.
This expertise allows sponsors to focus on advancing promising therapeutics while ensuring robust analytical support.
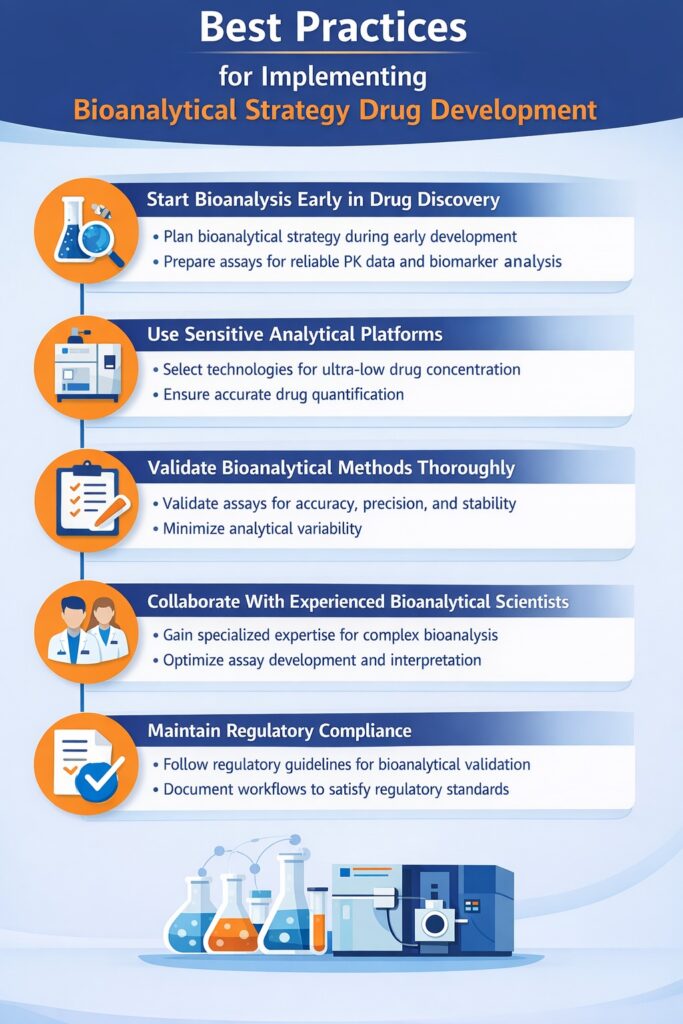
6: Best Practices for Implementing Bioanalytical Strategy Drug Development
A successful Bioanalytical Drug Development requires careful planning, advanced analytical technologies, and collaboration with experienced bioanalytical scientists.
Some companies also adopt virtual bioanalytical strategy or bioanalytical outsourcing models to optimize efficiency.
Biotech organizations increasingly outsource bioanalysis for biotech startups or rely on outsourced bioanalysis services to access specialized analytical expertise.
Below are key best practices that organizations should follow to strengthen their Bioanalytical Drug Development.
1. Start Bioanalysis Early in Drug Discovery
The most effective Bioanalytical Drug Development begins during the earliest stages of drug discovery. Integrating bioanalytical planning early allows development teams to design appropriate assays, establish analytical workflows, and ensure reliable data generation throughout the drug development lifecycle.
Early bioanalysis helps organizations:
- Generate reliable pharmacokinetic (PK) data
- Identify potential analytical challenges early
- Improve decision-making during candidate selection
- Reduce delays during preclinical and clinical studies
Starting bioanalysis early ensures that analytical methods are ready before critical development milestones.
2. Use Sensitive Analytical Platforms
Drug candidates often exist in extremely low concentrations in biological samples, particularly in early-stage studies. A strong Bioanalytical Drug Development requires analytical platforms capable of detecting and quantifying drugs with high sensitivity and accuracy.
Common technologies used in modern bioanalysis include:
| Technology | Application |
|---|---|
| LC-MS/MS | Quantification of small molecules |
| High-resolution mass spectrometry | Ultra-low level detection |
| Ligand binding assays | Biologics and monoclonal antibodies |
| Hybrid LC-MS methods | Peptides and oligonucleotides |
Selecting the right analytical platform ensures accurate drug measurement even at trace concentration levels.
3. Validate Bioanalytical Methods Thoroughly
Method validation is a critical component of Bioanalytical Drug Development. Validated assays ensure that analytical data are accurate, reproducible, and acceptable for regulatory submissions.
Bioanalytical methods must be validated for key parameters such as:
- Accuracy
- Precision
- Selectivity
- Sensitivity
- Stability
Thorough method validation minimizes analytical variability and ensures that data generated during preclinical and clinical studies are reliable.
4. Collaborate With Experienced Bioanalytical Scientists
Developing and implementing an effective Bioanalytical Strategy Drug Development requires specialized expertise. Experienced bioanalytical scientists understand complex biological matrices, analytical instrumentation, and regulatory expectations.
Working with skilled bioanalytical teams helps organizations:
- Optimize assay development
- Avoid analytical pitfalls
- Improve data interpretation
- Accelerate study timelines
Collaboration with experienced laboratories also ensures that analytical workflows remain efficient and scientifically robust.
5. Maintain Regulatory Compliance
Regulatory compliance is a critical aspect of Bioanalytical Drug Development. Global regulatory agencies expect bioanalytical methods to follow strict validation and documentation standards.
To maintain compliance, organizations should:
- Follow regulatory bioanalytical method validation guidelines
- Maintain detailed documentation and audit trails
- Implement strong data integrity practices
- Ensure reproducibility of analytical results
Adhering to regulatory expectations ensures that bioanalytical data can support IND submissions, clinical trials, and regulatory approvals.
Conclusion:
Early drug development programs frequently fail when bioanalytical strategy is weak, delayed, or poorly implemented. A strong Bioanalytical Strategy Drug Development framework ensures accurate pharmacokinetic data, reliable biomarker measurement, and regulatory-compliant analytical results supported by robust bioanalytical data and quality bioanalysis.
By integrating bioanalysis early and partnering with experienced laboratories or bioanalytical CROs for drug discovery, pharmaceutical and biotechnology companies can significantly reduce development risk and accelerate successful drug advancement.
Organizations like ResolveMass Laboratories Inc. provide the expertise, technology, and regulatory knowledge required to support robust Bioanalytical Drug Development across the entire drug development lifecycle through comprehensive ResolveMass bioanalytical services overview.
Frequently Asked Questions:
Bioanalytical Strategy Drug Development refers to the systematic planning and implementation of analytical methods used to measure drugs, metabolites, and biomarkers in biological samples during drug development. It ensures accurate pharmacokinetic (PK), pharmacodynamic (PD), and biomarker data required for regulatory submissions and clinical decision-making.
A strong Bioanalytical Strategy Drug Development is critical because it enables accurate measurement of drug exposure, safety, and biological response. Without reliable bioanalytical data, researchers cannot properly evaluate drug performance, which can lead to incorrect dosing decisions, regulatory delays, or program failure.
A well-designed Bioanalytical Strategy Drug Development provides validated analytical methods to measure drug concentrations in biological samples. This allows researchers to generate reliable pharmacokinetic data, including drug absorption, distribution, metabolism, and elimination, which are essential for dose optimization and safety assessment.
Regulatory agencies require bioanalytical methods to meet strict validation standards for accuracy, precision, selectivity, and reproducibility. A strong Bioanalytical Strategy Drug Development ensures that analytical data comply with regulatory guidelines, supporting successful IND submissions and clinical trial approvals.
Companies can strengthen their Bioanalytical Strategy Drug Development by:
-Integrating bioanalysis early in drug discovery
-Using highly sensitive analytical technologies
-Validating bioanalytical methods thoroughly
-Collaborating with experienced bioanalytical scientists
-Ensuring regulatory compliance throughout development
These best practices help reduce development risk and accelerate successful drug advancement.
Reference
- Aarzoo Thakur, Zhiyuan Tan, Tsubasa Kameyama, Eman El-Khateeb, Shakti Nagpal, Stephanie Malone. Bioanalytical strategies in drug discovery and development.https://www.tandfonline.com/doi/abs/10.1080/03602532.2021.1959606
- Saurabh Pandey , Preeti Pandey , Gaurav Tiwari , Ruchi Tiwari. Bioanalysis in drug discovery and development.https://www.sciencedirect.com/science/article/abs/pii/S2229470810110036
- Nuggehally R. Srinivas. Changing need for bioanalysis during drug development.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bmc.932
- Nuggehally R. Srinivas. Applicability of bioanalysis of multiple analytes in drug discovery and development: review of select case studies including assay development considerations.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bmc.594
- Brigitte Buscher, Sirpa Laakso, Hermann Mascher, Klaus Pusecker, Mira Doig, Lieve Dillen. Bioanalysis for Plasma Protein Binding Studies in Drug Discovery and Drug Development: Views and Recommendations of The European Bioanalysis Forum.https://www.tandfonline.com/doi/full/10.4155/bio.13.338
- Mohammad Mahdi Moein , Aziza El Beqqali , Mohamed Abdel-Rehim. Bioanalytical method development and validation: Critical concepts and strategies.https://www.sciencedirect.com/science/article/abs/pii/S1570023216308881


