The Strategic Imperative of Biosimilar Characterization Using Mass Spectrometry
Biosimilar Characterization Using Mass Spectrometry is a critical scientific approach used to confirm that a biosimilar closely matches its reference medicinal product (RMP). This method provides detailed molecular-level data and helps identify even very small differences in complex biologic drugs. Because biologics are naturally variable, this high-resolution technique is essential to ensure that any variation does not affect safety or effectiveness. As the global biopharmaceutical market continues to grow rapidly, mass spectrometry has become a key tool for generating a precise molecular “fingerprint” required by regulators.
Biologic drugs are large and complex molecules, often exceeding 150 kDa and containing thousands of atoms. Since biosimilars are produced in living cells, slight differences can occur due to post-translational modifications and protein folding. Biosimilar Characterization Using Mass Spectrometry helps overcome these challenges by offering high sensitivity and accuracy in detecting even trace-level differences. Compared to older techniques like circular dichroism (CD) or FT-IR spectroscopy, mass spectrometry provides much deeper insights, improving confidence in product comparability and strengthening quality control.
Learn how our high-resolution LC-MS/MS workflows provide the molecular “fingerprint” needed for your biosimilar. View Analytical Services for Generic Drug Development
| Analytical Characteristic | Small Molecule Generics | Biosimilars (Biologics) |
| Molecular Weight | Typically < 1,000\text{ Da} | Typically > 150,000\text{ Da} |
| Primary Method | HPLC-UV, NMR | High-Resolution LC-MS/MS |
| Structural Focus | Chemical identity | PTMs, HOS, Glycosylation |
| Heterogeneity | Minimal to none | High (Micro-heterogeneity) |
| Regulatory Threshold | Bioequivalence (PK) | Totality of Evidence (Structural + Functional) |
Share via:
Executive Summary
- Analytical Primacy and Regulatory Shifts: Modern biosimilar development relies on high-resolution analytical data, particularly mass spectrometry, which is now favored by the FDA over traditional clinical efficacy studies to establish biosimilarity.
- Primary Structure Verification: Exhaustive peptide mapping and intact mass analysis are essential to confirm $100\%$ amino acid sequence identity and detect low-abundance sequence variants.
- Advanced PTM Monitoring: The Multi-Attribute Method (MAM) enables the simultaneous quantification of post-translational modifications (PTMs) like deamidation, oxidation, and glycation at the site-specific level.
- Complex Glycan Profiling: Precise characterization of N- and O-linked glycans is required to ensure effector functions such as $ADCC$ and $CDC$ match the reference product.
- Higher-Order Structure (HOS) Comparability: Hydrogen/Deuterium Exchange Mass Spectrometry (HDX-MS) provides near-residue level resolution of protein folding and conformational dynamics.
- Statistical Rigor in Comparability: The implementation of frameworks like the Two One-Sided Test (TOST) and Maximum Deviation of True Equivalents (MDTE) provides the mathematical basis for demonstrating equivalence.
- Impurity and Residual Analysis: High-sensitivity proteomics workflows identify and quantify trace-level host cell proteins (HCPs) and process-related residuals.
- Instrumentation Strategy: Strategic selection between Orbitrap and Q-TOF mass analyzers is critical for balancing resolution, mass accuracy, and quantitative throughput.
Regulatory Landscape and the Analytical-First Paradigm
Regulatory frameworks for biosimilars have evolved toward an “analytical-first” paradigm, where comprehensive structural characterization can reduce or even eliminate the need for extensive clinical efficacy trials. Recent FDA draft guidances (2025 and 2026) emphasize that high-resolution analytical comparability, combined with human pharmacokinetic (PK) data, offers the most sensitive method for detecting product differences. This shift reflects the growing recognition that analytical tools, particularly mass spectrometry, can detect structural variations with greater precision than large-scale clinical studies. As a result, developers can rely more heavily on analytical data to support biosimilarity claims. This also allows regulatory bodies to streamline approval pathways without compromising safety.
The financial and operational advantages of this transition are significant. Comparative efficacy studies (CES) can account for up to 70% of total development costs, making them one of the most resource-intensive components of biosimilar development. By prioritizing risk-based analytical strategies, companies can shorten development timelines by one to three years while maintaining stringent safety and efficacy standards. ResolveMass Laboratories Inc. aligns with these expectations by delivering comprehensive, ICH Q6B-compliant characterization packages. These services help meet the “totality of evidence” requirements set by both the FDA and EMA. Additionally, this approach improves overall development efficiency and supports faster patient access to affordable biologics.
Ensure your submission meets the latest FDA and EMA analytical requirements with our specialized consulting. Access Regulatory Support for Generic Drug Development
The Totality of Evidence and the Structural Foundation
The “totality of evidence” refers to the complete dataset required to substantiate a biosimilarity claim, with analytical characterization forming the base of this framework. A robust analytical package demonstrates that the biosimilar closely matches the reference product in terms of quality attribute distribution. This minimizes uncertainty and provides a strong scientific basis for regulatory approval. Analytical data guide subsequent stages of development, including non-clinical and clinical studies, ensuring that each step addresses any remaining gaps in comparability. This structured approach enhances confidence in the final product. It also ensures that any residual risks are systematically evaluated and mitigated.
This stepwise strategy allows developers to build a comprehensive understanding of the biosimilar’s structural and functional properties. By starting with detailed molecular characterization, researchers can focus later studies on specific areas of concern rather than repeating broad evaluations. This targeted approach reduces redundancy and improves resource allocation. It also aligns with regulatory expectations for efficient and evidence-based development. Ultimately, it strengthens the scientific justification for biosimilarity.
Use high-resolution data to guide your formulation strategy and minimize structural variations early in the lifecycle. Learn about Formulation Development
Compliance with ICH Q6B and Q5E Guidelines
ICH Q6B establishes the standard framework for structural characterization of biotechnological products, requiring detailed analysis of amino acid sequences, terminal heterogeneity, and carbohydrate structures. ICH Q5E further provides guidance on comparability assessments when manufacturing changes occur, ensuring that the product remains highly similar throughout its lifecycle. Mass spectrometry is uniquely capable of addressing these requirements, as it can analyze multiple structural attributes within a single integrated workflow. This versatility makes it a cornerstone technology in biosimilar development. It also ensures consistency across different production batches.
By meeting these guidelines, developers can demonstrate regulatory compliance while maintaining product quality. Mass spectrometry enables simultaneous evaluation of primary structure, post-translational modifications, and impurity profiles. This integrated approach reduces the need for multiple independent assays. It also improves data consistency and reliability. As a result, regulatory submissions become more streamlined and robust.
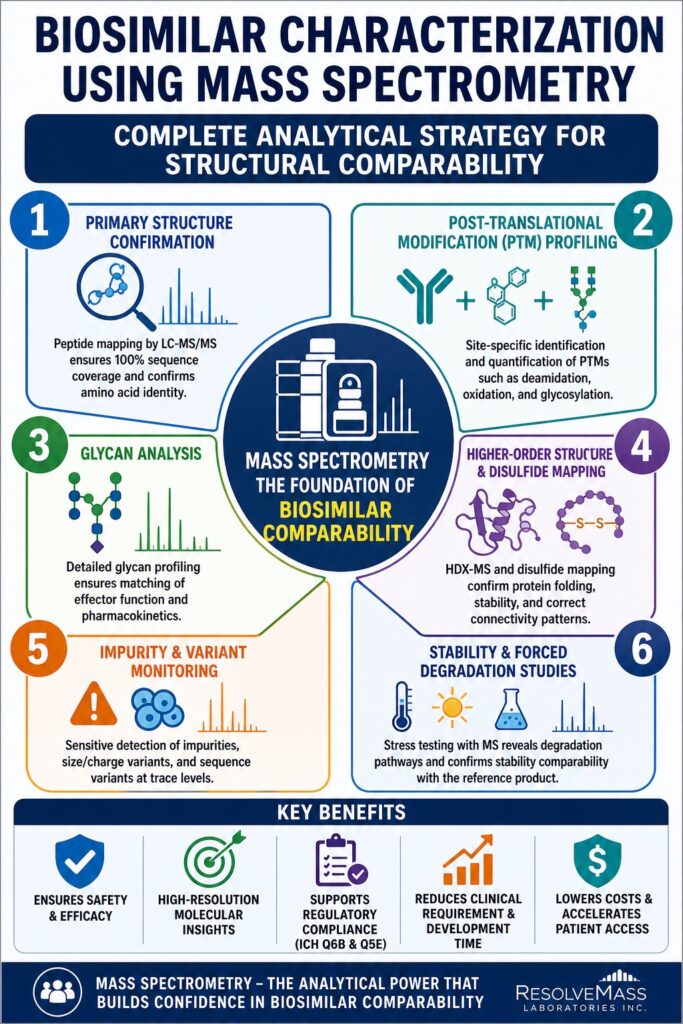
Primary Structure Confirmation: Exhaustive Peptide Mapping
Primary structure verification ensures that the amino acid sequence of the biosimilar matches that of the reference product, which is a fundamental requirement for approval. This is achieved through peptide mapping, where the protein is enzymatically digested into smaller fragments and analyzed using LC-MS/MS. ResolveMass aims for complete sequence coverage, identifying every amino acid in both heavy and light chains. This comprehensive mapping provides high confidence in sequence identity. It also helps detect any unexpected variations early in development.
The workflow involves protein denaturation, reduction of disulfide bonds using TCEP, and alkylation with iodoacetamide to prevent bond reformation. The protein is then digested using enzymes such as trypsin, followed by separation through reversed-phase UPLC and analysis using high-resolution MS/MS. This process ensures accurate identification of peptide fragments and confirms sequence integrity. Additional enzymatic approaches can be used to improve coverage and resolve challenging regions. This multi-enzyme strategy enhances analytical robustness and reliability.
| Enzyme | Specificity | Role in Primary Structure |
| Trypsin | Lys (K), Arg (R) | Primary sequence mapping; 100\% coverage goal |
| Lys-C | Lys (K) | Identification of large fragments; terminal analysis |
| Glu-C | Glu (E), Asp (D) | Resolving “tryptic-blind” regions; orthogonal mapping |
| Asp-N | Asp (D) | Complementary sequence verification |
| Chymotrypsin | Phe (F), Trp (W), Tyr (Y) | Sequencing hydrophobic domains |
Sequence Variant Analysis and Low-Abundance Detection
Sequence variants can arise from translational errors or mutations in the expression system and must be carefully monitored. Even low-level variants can impact biological function or immunogenicity. High-resolution mass spectrometry provides the sensitivity required to detect these variants at levels below 1%. This ensures that even minor differences are identified and evaluated. It also supports better control of manufacturing processes.
By distinguishing between isobaric amino acids and identifying single-point substitutions, mass spectrometry offers a level of precision not achievable with traditional assays. This capability is essential for maintaining product consistency and safety. It also enables early detection of potential issues, reducing the risk of downstream failures. Overall, it strengthens quality assurance in biosimilar production.
Our expertise extends to complex generic peptide and oligonucleotide characterization. Explore Generic Peptide & Oligonucleotide Projects
Terminal Heterogeneity and Chemical Modifications
Protein termini often exhibit heterogeneity due to enzymatic processing or chemical changes. Common examples include C-terminal lysine clipping and N-terminal pyroglutamate formation. These variations can influence the charge profile and overall stability of the molecule. Mass spectrometry allows detailed characterization of these terminal modifications. This ensures that the biosimilar matches the reference product’s distribution profile.
Understanding terminal heterogeneity is important for maintaining consistency across batches. It also helps in predicting product behavior during storage and administration. By accurately profiling these modifications, developers can ensure better product performance. This contributes to improved safety and efficacy outcomes.
Advanced Post-Translational Modification (PTM) Profiling
Post-translational modifications such as oxidation, deamidation, glycation, and succinimidation significantly affect the biological activity and stability of biosimilars. These modifications occur during manufacturing and storage and must be quantitatively compared to the reference product. High-resolution mass spectrometry enables site-specific identification and quantification of PTMs. This allows direct correlation between process parameters and product quality. It also helps optimize manufacturing conditions.
Accurate PTM profiling ensures that functional properties such as binding affinity and half-life remain consistent. It also supports regulatory requirements for detailed structural characterization. By understanding PTM patterns, developers can improve formulation strategies. This leads to more stable and effective products.
For non-biologic complex generics, leverage our specialized mapping for synthetic and recombinant peptides. Discover Generic Peptide Projects
| Modification Type | Chemical Change | Clinical Impact |
| Deamidation | Asn \rightarrow Asp/isoAsp | Potential loss of antigen binding in CDRs |
| Oxidation | Met/Trp \rightarrow Sulfoxide | Reduced FcRn binding; decreased half-life |
| N-Glycosylation | Glycan attachment | Altered ADCC/CDC; immunogenicity |
| Succinimidation | Cyclic imide formation | Potential intermediate for deamidation; instability |
| C-terminal Lys | Lysine clipping | Major charge variant contributor |
Site-Specific Deamidation and Oxidation Monitoring
Deamidation and oxidation are critical modifications that can alter protein function. Deamidation in complementarity-determining regions (CDRs) can reduce antigen binding, while methionine oxidation can affect FcRn interactions and shorten half-life. Mass spectrometry-based peptide mapping provides precise localization of these modifications. This enables a detailed assessment of their impact. It also supports targeted process improvements.
Monitoring these changes helps maintain product efficacy and stability. It also ensures that biosimilars remain comparable to their reference products. By identifying susceptible residues, developers can implement strategies to minimize degradation. This improves overall product quality.
The Multi-Attribute Method (MAM) in PTM Analysis
The Multi-Attribute Method (MAM) integrates multiple analytical assessments into a single LC-MS workflow. It simultaneously quantifies PTMs and detects new impurities through New Peak Detection (NPD). This approach replaces several traditional assays, improving efficiency and data consistency. It also provides a more detailed understanding of molecular changes.
MAM enhances batch-to-batch comparability and simplifies quality control processes. It reduces analytical complexity while increasing data depth. This makes it a valuable tool for modern biosimilar development. It also supports regulatory expectations for comprehensive characterization.
Complex Glycan Profiling and Effector Function
Glycosylation plays a crucial role in determining the biological activity and pharmacokinetics of therapeutic proteins. Variations in glycan structures can affect effector functions such as ADCC and CDC. Regulatory agencies require close matching of glycan profiles between biosimilars and reference products. Mass spectrometry enables detailed analysis of glycan composition and distribution. This ensures functional equivalence.
Advanced workflows allow detection of low-abundance glycoforms and site-specific analysis. These insights help optimize manufacturing processes. They also ensure consistent therapeutic performance. Accurate glycan profiling is therefore essential for biosimilar success.
| Glycan Attribute | Functional Impact | MS Characterization Goal |
| Fucosylation | Inversely related to ADCC | Quantification of G0F, G1F, G2F levels |
| Galactosylation | Influences CDC activity | Assessment of terminal galactose distribution |
| Sialylation | Increases half-life; anti-inflammatory | Monitoring Neu5Ac vs. Neu5Gc levels |
| High Mannose | Rapid clearance from circulation | Detecting Man5, Man6 species |
| Sulfate/Phosphate | Potential immunogenicity | High-sensitivity detection of acidic species |
Higher-Order Structure (HOS) and Conformational Comparability
Higher-order structure defines the functional conformation of proteins. Techniques like HDX-MS provide detailed insights into protein folding and dynamics. By measuring hydrogen-deuterium exchange rates, researchers can assess structural stability and flexibility. This allows detection of subtle conformational differences. It also helps predict potential stability issues.
Comparing HOS between biosimilars and reference products ensures functional similarity. It also supports long-term stability assessments. Advanced analytical methods provide high-resolution data for regulatory submissions. This strengthens confidence in biosimilar comparability.
| HOS Method | Spatial Resolution | Conformational Insight |
| HDX-MS | Peptide \rightarrow Residue | Local dynamics, solvent accessibility, folding |
| Native MS | Intact/Complex | Quaternary structure, non-covalent binding |
| Circular Dichroism | Global | Secondary structure percentages (\alpha-helix, \beta-sheet) |
| FT-IR/Raman | Global | Aggregate detection; general folding state |
| DSC/ITC | Global | Thermal stability; binding energetics |
Mapping Disulfide Bonds and Connectivity Patterns
Disulfide bonds are critical for maintaining protein structure and stability. Incorrect pairing can lead to misfolding and aggregation. Mass spectrometry enables precise mapping of disulfide linkages. Advanced fragmentation techniques such as ETD and UVPD improve detection of complex bond patterns.
Accurate disulfide mapping ensures structural integrity. It also helps identify potential manufacturing issues. By confirming correct connectivity, developers can ensure product safety. This is essential for regulatory approval.
| Fragmentation Technique | Mechanism | Advantage for Disulfides |
| CID | Vibrational activation | Backbone cleavage; weak for disulfides |
| ETD | Electron transfer | Selective cleavage of S-S bonds; preserves PTMs |
| UVPD (193\text{ nm}) | Photodissociation | High-energy cleavage of S-S and C-S bonds |
| EThcD | Hybrid ETD/HCD | Combines disulfide cleavage with backbone sequencing |
| ISR | In-source reduction | Online partial reduction for complex mapping |
Impurity Profiling and Residual Analysis
Impurity profiling is vital for ensuring biosimilar safety. Mass spectrometry can detect impurities at very low levels, including host cell proteins and residual DNA. This provides a more comprehensive analysis than traditional methods like ELISA. It also improves detection of co-purifying contaminants.
By identifying and quantifying impurities, developers can improve purification processes. This ensures a cleaner final product. It also reduces the risk of immunogenicity. Overall, it enhances patient safety.
Monitoring Size and Charge Variants
Size and charge variants are indicators of product stability and consistency. Mass spectrometry helps identify the chemical modifications responsible for these variants. Coupling MS with techniques like IEX or cIEF provides detailed isoform characterization. This supports compliance with regulatory requirements.
Understanding these variants helps optimize manufacturing processes. It also ensures consistent product quality. This is essential for maintaining biosimilarity. It also supports long-term product stability.
Forced Degradation and Stability-Indicating Strategies
Forced degradation studies evaluate how biosimilars respond to stress conditions such as pH changes, الحرارة, and oxidation. These studies identify degradation pathways and vulnerable regions. Mass spectrometry provides precise localization of these changes. This supports formulation development.
By comparing degradation patterns with the reference product, developers can confirm similarity. This ensures consistent stability profiles. It also helps define shelf-life parameters. This is critical for product approval.
| Stressor | Target | Degradation Outcome | MS Monitoring Method |
| High pH$ (> 8.0) | Asn residues | Deamidation/Succinimide | Peptide Mapping/MAM |
| Oxidative (H_2O_2) | Met/Trp residues | Sulfoxide formation | LC-MS/MS |
| Thermal (> 40\text{ }^\circ\text{C}) | HOS/Stability | Aggregation/Unfolding | HDX-MS/SEC-MALS |
| Low $pH$ (< 3.0) | Asp-Pro bonds | Peptide chain hydrolysis | Intact/Subunit Mass |
| Light Stress | Trp/Tyr | Photochemical oxidation | Peptide Mapping |
Instrumentation Strategy: Orbitrap vs. Q-TOF
Orbitrap and Q-TOF systems each offer unique advantages in biosimilar characterization. Orbitrap provides ultra-high resolution and mass accuracy, ideal for complex structural analysis. Q-TOF offers faster scan speeds and robust quantification capabilities. Using both platforms ensures comprehensive analysis.
Selecting the appropriate instrument depends on the analytical requirement. Combining technologies improves data quality and coverage. This ensures accurate characterization. It also supports regulatory compliance.
| Performance Metric | Orbitrap (e.g., Exploris) | Q-TOF (e.g., Xevo G3) |
| Resolving Power | Up to 480,000\text{–}1,000,000 | Up to 40,000\text{–}80,000 |
| Mass Accuracy | Typically < 1\text{ ppm} | Typically 1\text{–}3\text{ ppm} |
| Scan Speed | Dependent on resolution | High and constant |
| Intact Mass | Superior for glycoforms | Excellent for average mass |
| Sensitivity | High; trapping advantage | High; duty cycle advantage |
Informatics and Data Integrity
High-resolution mass spectrometry generates large datasets that require advanced informatics tools for analysis. Platforms like waters_connect and UNIFI ensure data integrity and regulatory compliance. They provide automated processing and reporting features. This improves efficiency and accuracy.
Advanced software also supports comparability assessments and impurity detection. It ensures consistent data interpretation across studies. This is essential for regulatory submissions. It also enhances overall analytical reliability.
Conclusion
Biosimilar characterization using mass spectrometry has transformed the approach to demonstrating structural comparability. By providing detailed insights into primary structure, PTMs, glycosylation, and higher-order structure, it ensures product safety and efficacy. This analytical-first strategy reduces reliance on extensive clinical trials while maintaining high standards. It also accelerates development timelines and improves cost efficiency.
As regulatory expectations continue to emphasize analytical rigor, mass spectrometry will remain central to biosimilar development. ResolveMass Laboratories Inc. plays a leading role in this field, offering advanced analytical solutions that bridge scientific complexity and regulatory requirements. Their expertise ensures that high-quality biosimilars reach the market efficiently. For more information, visit: https://resolvemass.ca/contact/
Frequently Asked Questions (FAQs)
Mass spectrometry provides such comprehensive structural and functional data that it can resolve most residual uncertainties regarding a biosimilar’s identity. When high-resolution MS data demonstrates that the biosimilar’s critical quality attributes fall within the range of the reference product, regulatory agencies like the FDA may waive large Phase III efficacy trials in favor of smaller, more sensitive pharmacokinetic studies.
Intact mass analysis evaluates the protein in its whole state, providing a high-level view of the overall molecular weight and major glycoforms. In contrast, peptide mapping involves digesting the protein into small fragments, which are then sequenced to confirm $100\%$ of the primary structure and to precisely identify the exact location of chemical modifications like deamidation or oxidation.
Glycans are sugar molecules that play a pivotal role in the drug’s effector functions, such as its ability to engage with the immune system and its stability in the body. Because glycosylation is highly sensitive to the cell line and manufacturing process, even small differences can impact the drug’s half-life or clinical performance, making detailed MS profiling essential for proving similarity.
MAM allows for the simultaneous monitoring of several critical quality attributes—such as oxidation, deamidation, and glycation—in a single, high-resolution LC-MS assay. By replacing multiple traditional assays like ion-exchange chromatography and HILIC with one integrated workflow, MAM provides more detailed molecular data while significantly reducing the time and cost of quality control.
HDX-MS probes the three-dimensional folding of a protein by measuring the exchange rate of its amide hydrogens in heavy water. This technique provides a sensitive map of the protein’s conformational dynamics and solvent accessibility, allowing researchers to mathematically demonstrate that the biosimilar has the same folding and stability as the reference medicinal product.
A developer should choose an Orbitrap when the primary goal is ultra-high mass resolution and baseline separation of complex intact glycoforms or non-covalent protein assemblies. However, for high-throughput peptide mapping or routine monitoring where fast scan speeds and high quantitative dynamic range are required, a Q-TOF system often provides a more robust and efficient solution.
Forced degradation studies intentionally stress the biosimilar with heat, pH, or oxidation to observe its breakdown pathways. By showing that the biosimilar degrades into the same fragments and modifications as the reference product, developers provide a “stress-test” proof that the underlying chemical stability of the two molecules is truly equivalent.
Reference:
- Yang, F., et al. (2023). Mass spectrometry-based multi-attribute method in protein characterization and quality control. Pharmaceutical Research. https://pmc.ncbi.nlm.nih.gov/articles/PMC10114992/
- U.S. Food and Drug Administration. (2025). Development of therapeutic protein biosimilars: Comparative analytical assessment and other quality-related considerations (Guidance for industry).
https://www.fda.gov/media/190203/download - Segu, Z., Stone, T., Berdugo, C., Roberts, A., Doud, E., & Li, Y. (2020). A rapid method for relative quantification of N-glycans from a therapeutic monoclonal antibody during trastuzumab biosimilar development. MAbs, 12(1), 1750794. https://pmc.ncbi.nlm.nih.gov/articles/PMC7188402/
- Nupur, N., Joshi, S., Guillarme, D., & Rathore, A. S. (2022). Analytical similarity assessment of biosimilars: Global regulatory landscape, recent studies and major advancements in orthogonal platforms. Frontiers in Bioengineering and Biotechnology, 10, 832059. https://pmc.ncbi.nlm.nih.gov/articles/PMC8865741/
- D’Atri, V., Guillarme, D., & Beck, A. (2025). Biopharmaceutical analysis—current analytical challenges, limitations, and perspectives. Analytical and Bioanalytical Chemistry. https://pmc.ncbi.nlm.nih.gov/articles/PMC12783247/
- Berkowitz, S. A., Engen, J. R., Mazzeo, J. R., & Jones, G. B. (2012). Analytical tools for characterizing biopharmaceuticals and the implications for biosimilars. Nature Reviews Drug Discovery, 11(7), 527–540. https://pmc.ncbi.nlm.nih.gov/articles/PMC3714370/



