
Introduction:
The Comparative Study of Cyclodextrin-Based Dendrimers and Polymer Micelles is essential for advancing modern drug delivery systems, especially for poorly soluble and complex therapeutic molecules. Both nanocarrier systems have gained significant attention in pharmaceutical R&D due to their ability to enhance solubility, stability, and bioavailability.
At ResolveMass Laboratories Inc., we leverage advanced analytical platforms and deep expertise in molecular characterization to evaluate and differentiate such nanocarriers, ensuring optimal selection for drug development and regulatory success.
Summary:
- Cyclodextrin-based dendrimers offer highly defined structures, strong host–guest interactions, and precise drug loading.
- Polymer micelles provide excellent solubilization of hydrophobic drugs and scalable formulation advantages.
- Dendrimers excel in targeted delivery and molecular precision, while micelles dominate in clinical translation and formulation simplicity.
- Selection depends on drug properties, delivery goals, and regulatory considerations.
- Advanced analytical tools such as LC-MS, HRMS, and NMR are critical for characterization and comparability studies.
1: What are Cyclodextrin-Based Dendrimers?
Cyclodextrin-based dendrimers are highly branched, tree-like macromolecules incorporating cyclodextrin units that enable host–guest inclusion complexes for drug delivery.
Key Characteristics
- Monodisperse structure with precise molecular weight
- Multiple functional groups for drug conjugation
- Hydrophobic cavity (cyclodextrin core) for inclusion complex formation
- High drug loading efficiency via encapsulation and surface attachment
Advantages
- Exceptional molecular precision and reproducibility
- Enhanced targeted drug delivery capabilities
- Reduced toxicity due to controlled architecture
Limitations
- Complex multi-step synthesis
- Higher production costs
- Limited large-scale industrial adoption
2: What are Polymer Micelles?
Polymer micelles are nanoscale, self-assembled structures formed from amphiphilic block copolymers, designed to encapsulate hydrophobic drugs within their core for improved solubility and delivery.
Key Characteristics
- Core-shell structure with hydrophobic core and hydrophilic shell
- Dynamic self-assembly in aqueous environments
- Nanoscale size (10–100 nm) suitable for passive targeting
Advantages
- Excellent solubilization of hydrophobic drugs
- Easier manufacturing and scalability
- Proven clinical applicability
Limitations
- Lower structural stability compared to dendrimers
- Potential premature drug release
- Less precise molecular architecture
3: Key Differences: Cyclodectrin-Based Dendrimers VS Polymer Micelles
Cyclodextrin-based dendrimers offer superior structural precision and strong host–guest interactions, whereas polymer micelles are simpler, scalable, and more clinically established.
| Feature | Cyclodextrin-Based Dendrimers | Polymer Micelles |
|---|---|---|
| Structure | Highly branched, monodisperse | Self-assembled, dynamic |
| Drug Loading | Inclusion + surface conjugation | Core encapsulation |
| Stability | High | Moderate |
| Synthesis | Complex | Simple (relatively easy) |
| Scalability | Limited | High |
| Targeting | Excellent (functionalizable) | Moderate |
| Clinical Use | Emerging | Widely studied and used |
Quick Insight
- Dendrimers are ideal when precision, targeting, and controlled delivery are critical.
- Micelles are preferred for ease of formulation, scalability, and practical pharmaceutical applications.
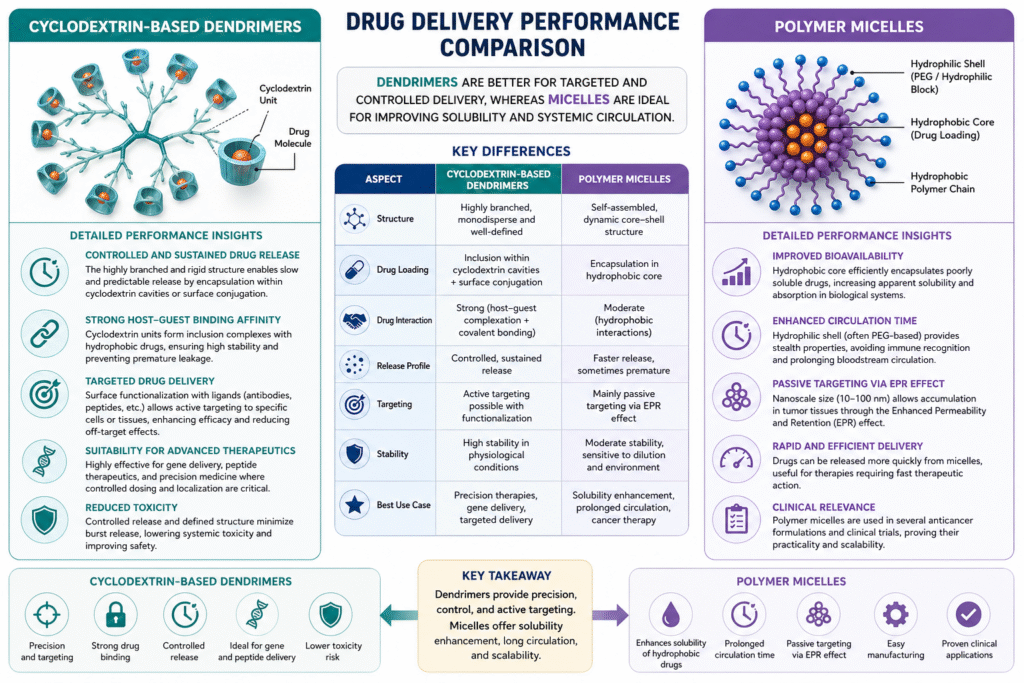
4: Drug Delivery Performance Comparison
Cyclodextrin-based dendrimers provide highly controlled, targeted, and stable drug delivery, whereas polymer micelles are optimized for enhancing solubility, circulation, and passive targeting of hydrophobic drugs.
Cyclodextrin-Based Dendrimers
Cyclodextrin-based dendrimers are engineered for precision drug delivery, combining dendritic architecture with cyclodextrin’s host–guest chemistry. This results in strong drug interactions and controlled therapeutic release.
Detailed Performance Insights
- Controlled and Sustained Drug Release
Their highly branched and rigid structure allows drugs to be either encapsulated within cyclodextrin cavities or conjugated on the surface. This dual mechanism enables slow, predictable, and sustained release profiles, reducing dosing frequency. - Strong Host–Guest Binding Affinity
Cyclodextrin units form inclusion complexes with hydrophobic drug molecules. This interaction is stronger and more specific compared to simple encapsulation, leading to improved drug stability and reduced premature leakage. - Targeted Drug Delivery
Surface functional groups can be modified with ligands (e.g., antibodies, peptides), enabling active targeting to specific cells or tissues such as tumors. This enhances therapeutic efficacy while minimizing off-target effects. - Suitability for Advanced Therapeutics
These systems are highly effective for gene delivery, peptide therapeutics, and precision medicine, where controlled dosing and localization are critical. - Reduced Toxicity
Due to their defined structure and controlled release, dendrimers help avoid burst release, thereby lowering systemic toxicity and improving safety profiles.
Polymer Micelles
Polymer micelles are widely used nanocarriers designed to improve solubility and pharmacokinetics, particularly for poorly water-soluble drugs.
Detailed Performance Insights
- Improved Bioavailability of Hydrophobic Drugs
The hydrophobic core of micelles efficiently encapsulates poorly soluble drugs, significantly enhancing their apparent solubility and absorption in biological systems. - Enhanced Circulation Time
The hydrophilic shell (often PEG-based) provides stealth properties, reducing recognition by the immune system. This leads to longer circulation time in the bloodstream. - Passive Targeting via EPR Effect
Due to their nanoscale size (10–100 nm), polymer micelles accumulate preferentially in tumor tissues through the Enhanced Permeability and Retention (EPR) effect, making them highly effective for cancer therapy. - Rapid and Efficient Drug Delivery
Compared to dendrimers, micelles may release drugs more quickly, which can be advantageous for therapies requiring fast therapeutic action. - Clinical Relevance
Polymer micelles are already used in several anticancer formulations and clinical trials, demonstrating their practicality and scalability.
Comparative Insight
| Aspect | Cyclodextrin-Based Dendrimers | Polymer Micelles |
|---|---|---|
| Release Profile | Controlled, sustained | Faster, sometimes premature |
| Drug Interaction | Strong (host–guest + conjugation) | Moderate (encapsulation) |
| Targeting | Active targeting possible | Mainly passive targeting |
| Stability | High | Moderate |
| Best Use Case | Precision therapies, gene delivery | Solubility enhancement, cancer therapy |
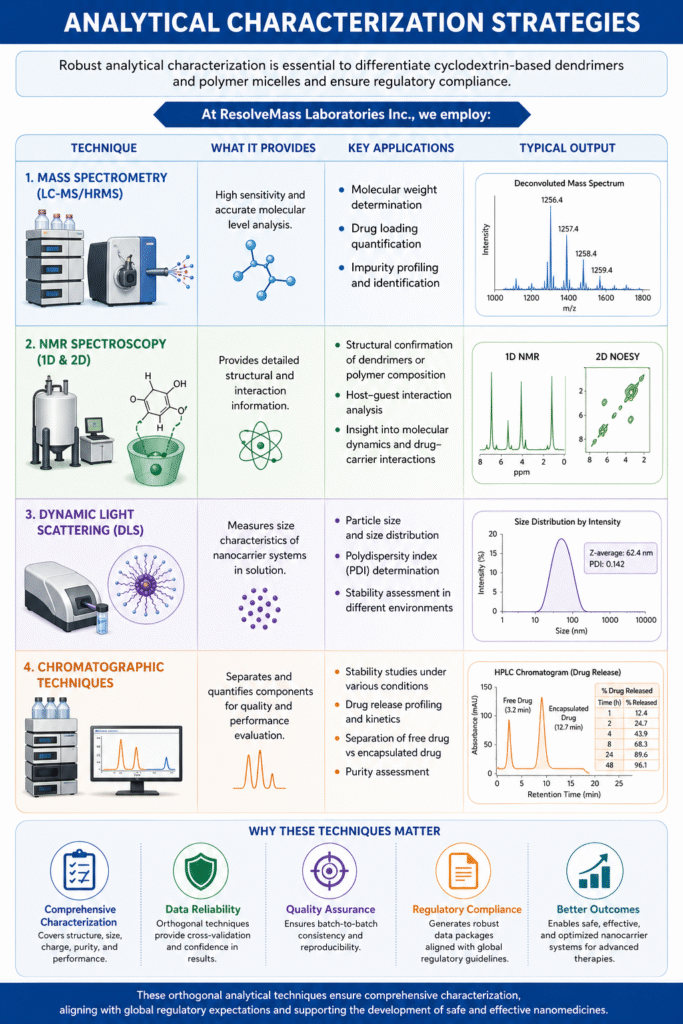
5: Analytical Characterization Strategies
Robust analytical characterization is critical to differentiate cyclodextrin-based dendrimers and polymer micelles, ensuring quality, performance, and regulatory compliance.
Mass Spectrometry (LC-MS/HRMS)
Mass spectrometry provides high sensitivity and accuracy for molecular-level analysis.
- Molecular weight determination for confirming structural integrity
- Drug loading quantification to assess encapsulation efficiency
- Impurity profiling for detecting degradation products and synthesis-related impurities
NMR Spectroscopy (1D & 2D)
NMR is essential for detailed structural and interaction analysis.
- Structural confirmation of dendrimer architecture or polymer composition
- Host–guest interaction analysis to verify inclusion complex formation in cyclodextrin systems
- Provides insight into molecular dynamics and drug–carrier interactions
Dynamic Light Scattering (DLS)
DLS is widely used for nanoparticle size characterization.
- Determines particle size and size distribution
- Evaluates polydispersity index (PDI) for uniformity
- Helps assess stability in different environments
Chromatographic Techniques
- Chromatography ensures separation and quantitative analysis of components.
- Separation of free drug vs encapsulated drug
- Stability studies under various conditions
- Drug release profiling to understand kinetics
6: Applications in Pharmaceutical Development
Cyclodextrin-based dendrimers and polymer micelles are both widely used in drug delivery, but they serve distinct therapeutic purposes based on their structural and functional properties.
Cyclodextrin-Based Dendrimers
Cyclodextrin-based dendrimers are primarily utilized where precision, targeting, and controlled delivery are essential.
- Targeted Cancer Therapy
Surface functionalization allows attachment of targeting ligands, enabling selective delivery to tumor cells, improving efficacy and reducing side effects. - Gene and Peptide Delivery
Their branched architecture and functional groups make them ideal for carrying nucleic acids, peptides, and biomolecules, ensuring protection and efficient cellular uptake. - Controlled Release Formulations
Strong host–guest interactions and structural stability support sustained and controlled drug release, enhancing therapeutic outcomes.
Polymer Micelles
Polymer micelles are widely applied where solubility enhancement and systemic delivery are the main goals.
- Chemotherapy Drug Delivery
Their ability to accumulate in tumors via the EPR effect makes them highly effective in anticancer drug formulations. - Poorly Soluble Drug Formulations
Hydrophobic core enables efficient encapsulation, significantly improving solubility and bioavailability of drugs. - Injectable Nanomedicines
Due to their nanoscale size and biocompatibility, they are suitable for intravenous administration, ensuring better circulation and therapeutic performance.
7: Regulatory and Development Considerations
Polymer micelles generally have a more established regulatory pathway, whereas cyclodextrin-based dendrimers require deeper characterization due to their structural complexity.
Key Considerations
- Reproducibility and Batch Consistency
Ensuring consistent manufacturing is critical for both systems. Variability in size, structure, or drug loading can impact safety and efficacy, making strict process control and validation essential. - Toxicological Evaluation
Comprehensive safety assessment is required to evaluate biocompatibility, biodistribution, and potential toxicity. Dendrimers, due to their synthetic complexity, often demand more extensive toxicological studies. - Impurity Profiling and Stability
Identification and control of process-related impurities, degradation products, and stability under different conditions are crucial for regulatory acceptance. - Comparability Studies (ANDA/NDA Submissions)
Demonstrating equivalence in quality, safety, and performance is essential, particularly for generic applications. This includes physicochemical characterization and in vitro/in vivo performance comparisons.
ResolveMass supports clients with regulatory-ready analytical data packages, ensuring smooth approval processes.
8: Which one should you Choose?
Choose cyclodextrin-based dendrimers for precision targeting and controlled delivery, while polymer micelles are better suited for scalability and clinically practical formulations.
Decision Factors
- Drug Solubility Profile
- Use polymer micelles for poorly water-soluble drugs due to their hydrophobic core.
- Use dendrimers when both encapsulation and specific molecular interactions are required.
- Required Targeting Efficiency
- Dendrimers are ideal for active targeting (ligand-based, site-specific delivery).
- Micelles mainly provide passive targeting (e.g., EPR effect in tumors).
- Manufacturing Scalability
- Polymer micelles are easier to manufacture and scale up.
- Dendrimers involve complex synthesis, limiting large-scale production.
- Regulatory Strategy
- Micelles have a more established regulatory pathway with existing clinical data.
- Dendrimers require more extensive characterization and regulatory justification.
Conclusion:
The Comparative Study of Cyclodextrin-Based Dendrimers and Polymer Micelles highlights that both nanocarrier systems play critical roles in modern drug delivery. While dendrimers offer unmatched precision and functional versatility, polymer micelles provide practical advantages in formulation and commercialization.
A strategic selection, supported by advanced analytical characterization, is essential to maximize therapeutic performance and regulatory success. ResolveMass Laboratories Inc. stands at the forefront of enabling such decisions through cutting-edge scientific expertise and reliable analytical solutions.
Frequently Asked Questions:
Cyclodextrin-based dendrimers are highly structured, branched systems with precise molecular architecture, while polymer micelles are self-assembled nanocarriers. Dendrimers offer better control and targeting, whereas micelles are easier to produce and scale. The choice depends on the required delivery performance and application.
Polymer micelles are generally better for improving the solubility of poorly water-soluble drugs. Their hydrophobic core efficiently encapsulates such drugs, enhancing bioavailability. Dendrimers can also improve solubility but are more focused on controlled and targeted delivery.
Yes, dendrimers are highly suitable for targeted drug delivery. Their surface can be functionalized with ligands, antibodies, or peptides, enabling precise delivery to specific tissues or cells. This makes them ideal for cancer therapy and advanced therapeutics.
Yes, polymer micelles can have lower structural stability compared to dendrimers. They may dissociate under certain physiological conditions, leading to premature drug release. However, formulation strategies can help improve their stability.
Cyclodextrin-based dendrimers are better suited for controlled and sustained drug release. Their defined structure and strong host–guest interactions allow precise control over how and when the drug is released.
They are characterized using advanced techniques such as LC-MS/HRMS for molecular analysis, NMR for structural confirmation, DLS for particle size, and chromatography for stability and release profiling. These methods ensure quality and regulatory compliance.
Reference
- Topuz F, Uyar T. Recent advances in cyclodextrin‐based nanoscale drug delivery systems. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2024 Nov;16(6):e1995.https://wires.onlinelibrary.wiley.com/doi/abs/10.1002/wnan.1995
- Li X, Liu J, Qiu N. Cyclodextrin-based polymeric drug delivery systems for cancer therapy. Polymers. 2023 Mar 11;15(6):1400.https://www.mdpi.com/2073-4360/15/6/1400
- Nafee N, Hirosue M, Loretz B, Wenz G, Lehr CM. Cyclodextrin-based star polymers as a versatile platform for nanochemotherapeutics: enhanced entrapment and uptake of idarubicin. Colloids and Surfaces B: Biointerfaces. 2015 May 1;129:30-8.https://www.sciencedirect.com/science/article/pii/S0927776515001484
- Soh WW, Li J. Cyclodextrin‐Based Pseudocopolymers and Their Biomedical Applications for Drug and Gene Delivery. Small. 2025 Sep;21(36):e01304.https://onlinelibrary.wiley.com/doi/abs/10.1002/smll.202501304
- Santos AC, Costa D, Ferreira L, Guerra C, Pereira-Silva M, Pereira I, Peixoto D, Ferreira NR, Veiga F. Cyclodextrin-based delivery systems for in vivo-tested anticancer therapies. Drug Delivery and Translational Research. 2021 Feb;11(1):49-71.https://link.springer.com/article/10.1007/s13346-020-00778-5


