Introduction:
A long acting injectable is only as strong as the attributes that define its performance. Critical Quality Attributes for Long Acting Injectables refer to measurable physical, chemical, biological, and microbiological properties that must stay within defined limits to ensure consistent therapeutic outcomes. These attributes directly influence how the drug releases, how safe it is, and how reliably it performs across batches. Even small shifts in these parameters can affect pharmacokinetics, dose accuracy, or patient safety. Because of this, Critical Quality Attributes for Long Acting Injectables are not just development checkpoints but essential elements of lifecycle quality management. Establishing clear and clinically meaningful specifications helps maintain product consistency from early development through commercialization.
Explore our expertise: Analytical Requirements for ANDA Generic Drugs
Unlike conventional oral tablets where CQAs are more standardized, long acting injectables, including depot microspheres, nanosuspensions, lipid systems, and in situ forming depots, require a more complex and formulation-specific CQA strategy. Drug release may extend from weeks to months, which increases the impact of any variation in formulation attributes. For example, a modest change in particle size that may not affect an oral suspension can significantly alter release kinetics in a polymeric microsphere system. This sensitivity makes thorough characterization under physiological conditions essential. Manufacturing variability can also accumulate over long release periods, making robust control strategies critical. As a result, identifying and controlling Critical Quality Attributes for Long Acting Injectables requires a deeper, risk-based development approach.
Learn more about the roadmap: Generic Drug Development Process for ANDA
This article explains the most important attributes that determine long acting injectable success, why they are technically challenging, and how to manage them effectively. Each section focuses on attributes that influence release behavior, dose reproducibility, and administration performance. The discussion also highlights common development challenges and regulatory expectations. Together, these insights provide a practical framework for designing robust LAI products. Understanding Critical Quality Attributes for Long Acting Injectables early helps reduce development risk and improves long-term product reliability.
Share via:
📋 Article Summary
- Critical Quality Attributes (CQAs) for long-acting injectables are the key physical, chemical, biological, and microbiological properties that must remain within defined limits to ensure safe, consistent, and prolonged drug delivery.
- Important CQAs in LAI formulations include particle size distribution, drug loading, encapsulation efficiency, in vitro release profile, syringeability (ease of injection), sterility, and long-term stability.
- These attributes are highly interconnected, meaning a change in one parameter—such as particle size—can directly affect other factors like drug release behavior, encapsulation efficiency, and injectability.
- Conventional analytical methods used for standard injectables are often not suitable for LAIs. Specialized, formulation-specific methods must be developed and validated to accurately measure these complex systems.
- Regulatory frameworks such as ICH Q8, Q9, Q10, and the FDA’s Quality by Design (QbD) approach require that CQAs be identified early during the pre-formulation stage, rather than being addressed later in development.
- Establishing a reliable in vitro–in vivo correlation (IVIVC), when possible, reflects a strong understanding of the formulation and helps reduce regulatory challenges when making post-approval changes.
1. Particle Size Distribution (PSD): A Key Critical Quality Attribute for Long Acting Injectables
Particle size distribution directly controls surface area available for drug diffusion, erosion, and dissolution, making it one of the most influential Critical Quality Attributes for Long Acting Injectables. Even small changes in PSD can modify the rate and uniformity of drug release. Since many LAIs rely on diffusion and polymer degradation, particle size becomes a mechanistic driver of performance. This is particularly important for polymeric microspheres and nanosuspension systems. Maintaining a narrow and consistent PSD is therefore essential for predictable pharmacokinetics.
For microsphere and nanosuspension formulations, PSD does more than describe physical dimensions. Smaller particles increase surface area, which can accelerate initial release and raise burst effects. Larger particles extend diffusion pathways and may sustain release for longer durations. However, a broad size distribution can create multiphasic release profiles. This may lead to variability in patient exposure during the dosing interval. Careful PSD optimization helps balance initial release with sustained therapeutic levels.
| PSD Parameter | Why It Matters for LAIs | Typical Analytical Method |
|---|---|---|
| D10, D50, D90 values | Define population breadth; span directly affects dose uniformity and release predictability | Laser diffraction (wet dispersion) |
| Span index | Narrow span = more predictable release; wide span = biphasic or erratic profiles | Calculated from D10/D50/D90 |
| Aggregation state | Agglomerated particles behave as larger units in vivo, shortening effective release duration | DLS, optical microscopy |
| Particle morphology | Shape affects packing density, syringeability, and in vivo surface erosion rate | SEM, optical microscopy |
Technical Case Study: Lupron Depot Particle Characterization Insights
A common development mistake is measuring PSD only in the manufactured formulation without considering reconstitution and administration conditions. Particle size may change during reconstitution, mixing, or injection. These shifts may not be visually obvious but can strongly influence release kinetics. Shear during mixing can break aggregates or create new ones. Because of this, CQA specifications should reflect the state at the time of administration, not only at manufacture.
2. Drug Loading and Encapsulation Efficiency: Dose Accuracy in Long Acting Injectables
Encapsulation efficiency determines whether the administered dose delivers the intended therapeutic effect. Low encapsulation efficiency moves drug into the rapidly released fraction instead of the controlled-release matrix. This directly impacts both duration and efficacy. Variability in drug loading can also lead to inconsistent exposure across batches. For this reason, drug loading is a core Critical Quality Attribute for Long Acting Injectables.
In polymeric microsphere systems such as PLGA formulations, theoretical and actual drug loading often differ. Process losses during emulsification, solvent removal, and drying can reduce encapsulation efficiency. These losses may vary across particle sizes, creating non-uniform drug distribution. When this happens, particle size alone cannot predict release behavior accurately. Uneven distribution may also increase burst variability. Maintaining uniform loading requires careful process optimization and monitoring.
Specialized Support: CDMO for Long Acting Injectable Formulation Development
Critical sub-attributes within this CQA include:
- Drug distribution uniformity within individual particles (core vs. surface loading)
- Residual solvent content, organic solvents must be controlled per ICH Q3C limits; they alter polymer chain mobility and post-injection release kinetics
- Free drug fraction, unencapsulated drug drives initial burst and must be specified separately from encapsulated drug content
- Drug-polymer interaction stability, some APIs form irreversible complexes with PLGA or PLA under thermal or pH stress, reducing effective drug loading over shelf life
Analytical techniques such as validated HPLC extraction, UV spectrophotometry after matrix digestion, and confocal Raman microscopy are commonly used. Each method must demonstrate extraction efficiency and sensitivity. Proper validation ensures accurate measurement of encapsulated versus free drug. Reliable analytical control is essential for maintaining consistent dose delivery.
3. In Vitro Release Profile: Predicting In Vivo Performance
The in vitro release profile is one of the most closely evaluated Critical Quality Attributes for Long Acting Injectables. It acts as a surrogate for in vivo drug exposure across extended dosing periods. A well-designed IVR method supports regulatory approval and product understanding. Poorly designed methods may fail to detect meaningful formulation changes. Therefore, IVR development must be scientifically justified.
Standard USP dissolution methods often require modification for LAI products. A suitable IVR method should maintain sink conditions for weeks or months. Agitation must be controlled to avoid artificial particle breakdown. The method should distinguish polymer-controlled release from simple dissolution. Sensitivity to formulation changes is also important. These requirements make IVR method development technically demanding.
Overcoming Challenges: Leuprolide Depot Analytical Challenges and Solutions
PLGA microspheres often show a three-phase release pattern: initial burst, lag phase, and sustained release. Each phase must be measured separately. Regulatory agencies expect discriminatory methods that detect meaningful differences. Sampling should cover the full release period. This improves correlation with clinical performance.
On IVIVC: Where achievable, a Level A IVIVC reduces the need for additional in vivo studies. Building IVIVC begins during IVR method development. Conditions should reflect physiological release mechanisms. Establishing IVIVC also supports lifecycle management. This strengthens regulatory flexibility for post-approval changes.
4. Syringeability and Injectability: Administration-Focused Critical Quality Attributes for Long Acting Injectables
Syringeability and injectability determine whether the intended dose reaches the patient. These attributes are safety-critical and influence real-world usability. Failures in injectability can undermine otherwise well-designed formulations. Because of this, they are essential Critical Quality Attributes for Long Acting Injectables.
These properties depend on viscosity, particle concentration, particle size, vehicle composition, and needle selection. High-concentration LAI suspensions often show shear-thinning behavior. Viscosity decreases during injection but recovers afterward. This can cause mid-injection clogging. Performance may also change with temperature or handling conditions. Testing must therefore reflect real clinical use.
Factors that make this CQA technically complex include:
- Temperature dependence of viscosity means room-temperature handling versus body-temperature injection conditions produce different flow behaviors in the same product
- Particle sedimentation during the window between reconstitution and injection creates time-dependent concentration gradients, making injectability a dynamic attribute
- Needle geometry including gauge, bevel angle, and length interacts with formulation properties, meaning injectability is a system attribute
Deep Dive: Characterization of Long Acting Injectables
Forced degradation studies simulating real-world handling should be included. These may involve agitation, temperature cycling, and hold-time studies after reconstitution. Such evaluations help identify administration risks. Realistic testing improves confidence in dose delivery.
5. Sterility Assurance and Endotoxin Control
Sterility and endotoxin limits are non-negotiable for long acting injectables. These parameters directly impact patient safety. Many LAI formulations cannot undergo terminal sterilization. As a result, aseptic processing is commonly required. This increases manufacturing complexity.
Aseptic processing demands strict facility controls, validated procedures, and environmental monitoring. Some excipients used in LAIs carry higher endotoxin risk. Raw material quality becomes critical. Container-closure integrity must also be confirmed. These controls together maintain sterility throughout shelf life.
Key considerations in sterility CQA management:
- Sterility testing alone is insufficient; parametric release and SAL modeling from media fill validation are expected by regulators as primary aseptic process assurance tools
- Bacterial endotoxin testing using LAL or recombinant Factor C assays must be validated for matrix interference
- Container-closure integrity is especially critical for lyophilized LAI systems where headspace moisture ingress impacts polymer crystallinity and alters the release profile
Matrix interference can mask endotoxin detection. Proper validation ensures accurate results. Container closure integrity testing confirms sterility maintenance. These measures create a comprehensive sterility assurance strategy.
Partnership Options: CRO vs. In-House ANDA Development
6. Physical and Chemical Stability Across Shelf Life
Stability for LAIs involves maintaining all Critical Quality Attributes for Long Acting Injectables, not just potency. Particle size, encapsulation efficiency, and release kinetics must remain within limits. Stability therefore includes both physical and chemical integrity. Changes in polymer structure can affect release without visible changes.
A product may appear unchanged while release behavior shifts. Polymer crystallization or particle growth can cause this issue. Conventional stability approaches may not detect such changes. Stability-indicating methods must track multiple attributes. Monitoring only drug content is not sufficient.
Stability-indicating methods for LAIs must track:
- Polymer molecular weight distribution (GPC/SEC for PLGA/PLA)
- Particle size evolution including aggregation onset and Ostwald ripening in nanosuspensions
- Drug-polymer interaction products identified by LC-MS forced degradation studies
- Residual moisture in lyophilized forms via Karl Fischer titration with matrix-specific validation
Accelerated stability studies require careful interpretation. Temperature-based predictions may not apply to polymer-controlled systems. Degradation mechanisms can change under stress conditions. Real-time stability data remains essential.
Success Story: Leuprolide Depot Case Study on Sustained Release
Regulatory Framework: Critical Quality Attributes for Long Acting Injectables Under ICH Q8/Q9/Q10
The ICH Q8, Q9, and Q10 guidelines provide the framework for identifying and controlling Critical Quality Attributes for Long Acting Injectables. Under Quality by Design, CQAs are linked to Critical Material Attributes and Critical Process Parameters. This structured approach supports systematic development. It also improves regulatory clarity and lifecycle management.
| CQA | Linked CMA Example | Linked CPP Example |
|---|---|---|
| Particle size distribution | Polymer Mw, API input particle size | Emulsification speed, homogenization pressure |
| Encapsulation efficiency | Oil/water phase ratio, polymer:drug ratio | Solvent extraction rate, temperature |
| In vitro release profile | Polymer:drug ratio, excipient grade, Mw | Drying cycle parameters, inlet temperature |
| Injectability | Suspension concentration, excipient viscosity | Reconstitution volume, mixing time, temperature |
| Sterility | Excipient bioburden, container closure grade | Filtration pressure, fill environment class |
Regulatory submissions such as NDA, ANDA, and 505(b)(2) must include a clear CQA justification. Each attribute should be supported by risk assessment and analytical validation. FDA guidance for complex formulations emphasizes enhanced characterization for LAIs. A well-defined CQA strategy improves regulatory acceptance. It also supports lifecycle flexibility.
Regulatory Strategy: Requirements for ANDA Submission of Generic Drugs
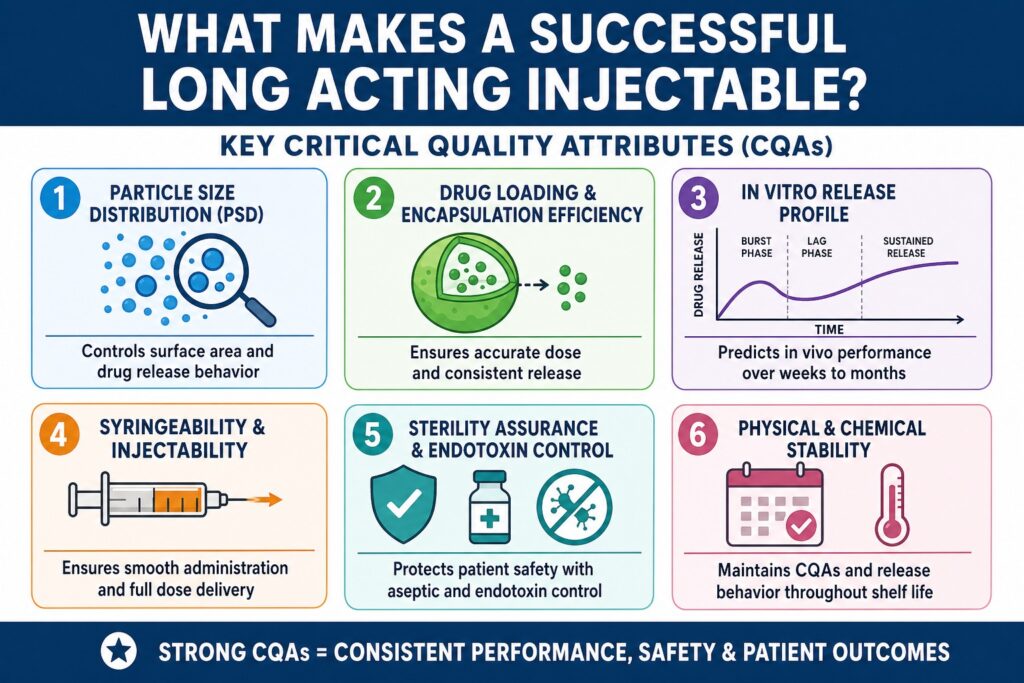
Conclusion: Why Critical Quality Attributes for Long Acting Injectables Matter
Critical Quality Attributes for Long Acting Injectables form the foundation of a safe and reliable product. Particle size, drug loading, release kinetics, injectability, sterility, and stability all work together. Failure in one attribute can affect overall performance. These attributes must therefore be managed as an integrated system. Early identification and control improve product robustness.
For developers working on long acting injectable products, building a strong CQA framework early is essential. Applying Quality by Design principles helps define acceptable ranges. This approach reduces development risk and supports regulatory success. A well-controlled strategy ensures consistent therapeutic performance. Ultimately, careful management of Critical Quality Attributes for Long Acting Injectables supports long-term product quality and patient outcomes.
Work with Experts: CRO for Complex Injectables and Generic Development
Frequently Asked Questions
Conventional injectables emphasize attributes that control immediate safety and rapid systemic exposure, such as pH, osmolality, sterility, and drug concentration. Long-acting injectables must additionally ensure consistent drug delivery over weeks or months, making factors like polymer properties, particle size distribution, encapsulation efficiency, and release kinetics critical. Because performance extends over long durations, even small deviations can significantly affect therapeutic outcomes. This requires tighter specifications and more advanced analytical control than typical parenteral products.
Developing an in vitro release method for LAIs is difficult because it must replicate a prolonged biological release process while remaining practical and reproducible in the laboratory. Different formulations rely on varying mechanisms such as diffusion, erosion, or swelling, so no single standardized apparatus applies universally. Method design must consider media composition, sampling schedule, and sink conditions. Regulators also expect the method to detect meaningful formulation changes, adding an extra layer of validation complexity.
Laser diffraction using wet dispersion is commonly used for particle size distribution due to its speed and broad measurement range. However, because it assumes spherical particles and relies on modeling, complementary methods are typically required. Optical microscopy helps verify morphology, while dynamic light scattering supports analysis of smaller particle fractions. Scanning electron microscopy provides detailed surface imaging but is mainly qualitative and unsuitable for large-scale statistical measurements.
Polymer molecular weight directly influences degradation rate and therefore the duration of drug release. Higher molecular weight PLGA generally slows degradation and extends release, whereas lower molecular weight accelerates hydrolysis and may increase initial burst. It also affects manufacturing viscosity, which can alter encapsulation efficiency and particle structure. Since polymer chains can degrade during storage, monitoring molecular weight serves as an important stability indicator for long-term performance.
Encapsulation efficiency represents the percentage of drug successfully incorporated within the delivery matrix relative to the intended amount. Low efficiency means more free drug is available outside the controlled-release structure, which can increase the initial burst and reduce sustained dosing. This variability can directly impact patient exposure over the dosing interval. For this reason, encapsulation efficiency is typically controlled within specifications to maintain consistent clinical performance.
Syringeability evaluates how easily a formulation can be withdrawn from its container into a syringe, focusing on withdrawal force, foaming, and dose accuracy. Injectability, in contrast, measures the force and consistency required to expel the product through the needle during administration. These tests occur at different stages of use but both influence usability and dosing reliability. Formulation viscosity, particle content, and needle gauge can affect the outcomes of each test differently.
Terminal sterilization methods such as heat or irradiation often damage polymer-based LAI systems by degrading carriers or destabilizing the drug. High temperatures can accelerate polymer breakdown, while radiation may cause chain scission and reactive species formation. As a result, most LAIs are manufactured using aseptic processing instead. This approach requires stringent environmental controls, validated processes, and container-closure integrity assurance.
Burst release refers to the rapid drug release that occurs shortly after administration and can strongly influence safety and efficacy. An excessive burst may produce transiently high plasma levels, while too little release can delay therapeutic effect. Factors such as surface drug, particle porosity, and polymer hydrophilicity typically control this phase. Because of its clinical importance, burst release is often specified as an early time-point separate from the overall release profile.
Reference:
- Markowicz-Piasecka, M., Kubisiak, M., Asendrych-Wicik, K., Kołodziejczyk, M., Grzelińska, J., Fabijańska, M., & Pietrzak, T. (2023). Long-acting injectable antipsychotics—A review on formulation and in vitro dissolution. Pharmaceutics, 16(1), 28. https://doi.org/10.3390/pharmaceutics16010028
- Huang, Y., Zhang, L., Liu, Y., Chen, X., & Wang, J. (2024). Long-acting injectable drug delivery systems: Current advances and future perspectives. International Journal of Pharmaceutics, 657, 123456. https://pmc.ncbi.nlm.nih.gov/articles/PMC12623523/
- Manchanda, R., Chue, P., Malla, A., Tibbo, P., Roy, M.-A., Williams, R., Iyer, S., Lutgens, D., & Banks, N. (2013). Long-acting injectable antipsychotics: Evidence of effectiveness and use. The Canadian Journal of Psychiatry, 58(5 Suppl 1), 5S–13S. https://doi.org/10.1177/088740341305805s02
- Bauer, A., Berben, P., Chakravarthi, S. S., Chattorraj, S., Garg, A., Gourdon, B., Heimbach, T., Huang, Y., Morrison, C., Mundhra, D., Palaparthy, R., Saha, P., Siemons, M., Shaik, N. A., Shi, Y., Shum, S., Thakral, N. K., Urva, S., Vargo, R., … Barrett, S. E. (2023). Current state and opportunities with long-acting injectables: Industry perspectives from the Innovation and Quality Consortium “Long-Acting Injectables” working group. Pharmaceutical Research, 40(7), 1601–1631. https://doi.org/10.1007/s11095-022-03391-y



