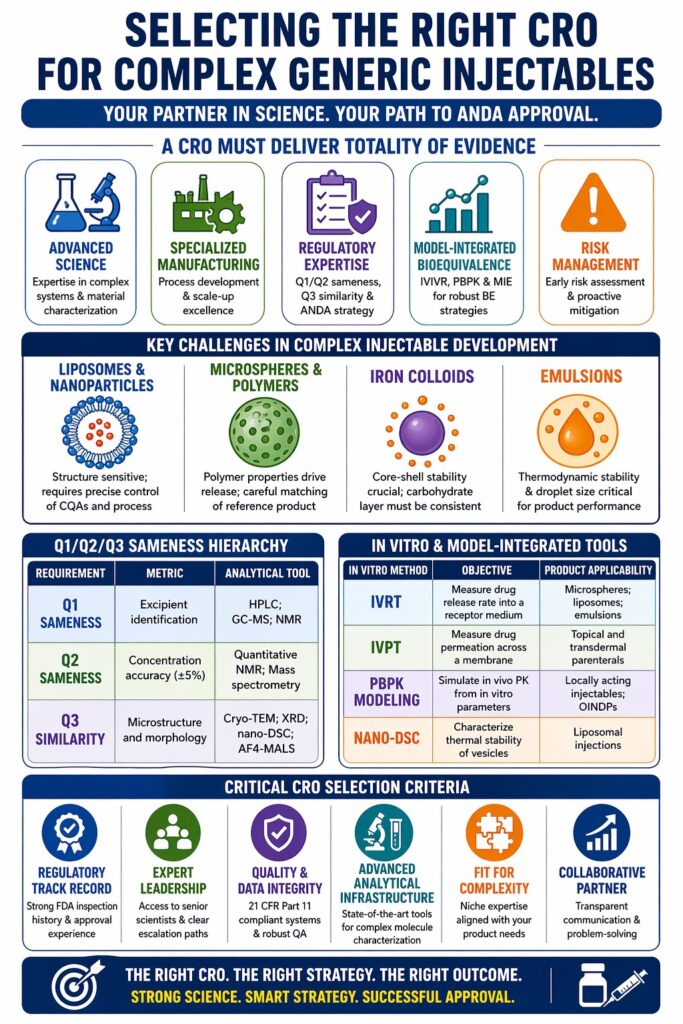
Choosing the right Contract Research Organization (CRO) for complex injectables is a strategic decision that goes far beyond basic outsourcing. It requires identifying a partner that can successfully manage the overlap between advanced material science, specialized manufacturing processes, and constantly evolving regulatory requirements. A CRO working with non-biological complex drugs (NBCDs) must offer more than routine lab testing. It should provide a fully integrated scientific platform capable of building the “totality of evidence” needed for Abbreviated New Drug Application (ANDA) approval. As the pharmaceutical industry shifts toward high-barrier products such as liposomes, microspheres, and iron colloids to meet unmet medical needs, the CRO’s expertise in physicochemical characterization (Q3) and model-integrated bioequivalence (BE) becomes critical. In addition, the CRO should be able to anticipate development risks early and propose mitigation strategies, ensuring smoother progression from formulation to approval.
Explore our full suite of services: Comprehensive Pharmaceutical CDMO Services in the US and Canada
Share via:
Technical Hurdles in Complex Generic Parenteral Development
Developing complex injectables involves many technical challenges due to multi-component systems and sensitivity to small changes. Even minor variations in the process can impact safety and performance. When selecting a CRO for Complex Injectables, it is important to assess how well they manage interactions between drug substances, excipients, and delivery systems. This includes studying how each component behaves under different conditions and stress factors. A strong CRO will also provide effective troubleshooting and ensure consistent batch-to-batch results through robust process controls.
Compare your outsourcing options: CRO vs. In-House ANDA Development: Making the Right Choice
Complexity of Liposomal and Nanoparticle Systems
Liposomal and nanoparticle-based injectables are advanced drug delivery systems that improve targeting and reduce side effects. These systems carry drugs within lipid or polymer structures, making them more complex to analyze. A CRO for Complex Injectables must evaluate properties such as vesicle structure, lipid behavior, and drug-to-lipid ratios with precision. For example, liposomal doxorubicin depends on PEGylated nanoparticles to improve tumor targeting. Maintaining proper PEG density and particle consistency is critical to ensure stable and predictable performance.
Deep dive into characterization:Advanced Characterization of Long-Acting Injectables
| Product Type | Complexity Driver | Critical Quality Attributes (CQAs) |
| Liposomes | Bi-layered structure; lipid-drug interactions. | Particle size; zeta potential; lamellarity; phase transition temperature. |
| Microspheres | Polymer degradation kinetics; drug release profile. | Molecular weight; lactide/glycolide ratio; glass transition temperature ($T_g$). |
| Iron Colloids | Core-shell structure; carbohydrate stabilization. | Iron core size; carbohydrate shell thickness; molecular weight distribution. |
| Emulsions | Thermodynamic stability; droplet size. | Droplet size distribution; pH; viscosity; free drug concentration. |
The manufacturing process for these systems requires specialized equipment such as high-pressure homogenizers and lipid extruders. A CRO must have hands-on experience with such technologies to ensure efficiency and quality. Poor control during manufacturing can lead to aggregation or drug leakage, which affects stability and effectiveness. Therefore, process optimization and scale-up expertise are key factors when choosing the right partner.
Specialized Polymers and Microsphere Formulation
Long-acting injectables often use biodegradable polymers like PLGA for controlled drug release. The challenge lies in matching polymer properties with the reference product. A CRO for Complex Injectables must have validated methods to measure polymer composition, molecular weight, and chemical structure. These factors directly influence how the drug is released over time. Understanding environmental effects such as temperature and pH is also important for predicting product behavior in the body.
Advanced tools like GPC and NMR are necessary for accurate polymer analysis. Without proper characterization, there is a higher risk of formulation issues such as burst release. This can lead to safety concerns and poor therapeutic outcomes. A knowledgeable CRO will design formulations carefully and ensure stable, controlled drug delivery.
Learn more about our specialized expertise:How to Develop Generic Leuprolide Depot: A Technical Guide
The Regulatory Framework: Establishing Sameness and Similarity
Regulatory approval for complex injectables requires strong evidence of similarity to the reference product. This includes Q1/Q2 sameness and Q3 similarity, which go beyond basic comparisons. A CRO for Complex Injectables must combine analytical data, in vitro studies, and sometimes clinical evidence to demonstrate equivalence. Early planning and alignment with regulatory expectations can reduce delays and improve approval chances. A well-prepared strategy ensures smoother communication with regulatory agencies.
Master the submission requirements:Key Requirements for ANDA Submission of Generic Drugs
The Q1/Q2/Q3 Sameness Hierarchy
Q1 sameness ensures the same excipients are used, while Q2 focuses on matching their quantities. Q3 similarity is more complex and involves comparing physical and structural properties. These include particle size, morphology, and material behavior. A CRO must use advanced analytical tools to confirm these characteristics accurately. Understanding how these attributes affect drug performance is essential for success.
For systems like liposomes and iron colloids, structure plays a major role in how the drug works. Even small variations can change effectiveness and safety. Reliable testing methods and reproducible results are necessary to meet regulatory standards. A skilled CRO ensures that all analytical approaches are robust and compliant.
Understand the testing landscape:Analytical Requirements for ANDA Generic Drug Approval
| Requirement | Metric | Analytical Tool |
|---|---|---|
| Q1 Sameness | Excipient identification | HPLC; GC-MS; NMR |
| Q2 Sameness | Concentration accuracy (±5%) | Quantitative NMR; Mass spectrometry |
| Q3 Similarity | Microstructure and morphology | Cryo-TEM; XRD; nano-DSC; AF4-MALS |
Navigating ANDA vs. 505(b)(2) Pathways
Selecting the right regulatory pathway is a key part of the strategy. The ANDA route is suitable when the product closely matches the reference drug. If full similarity cannot be demonstrated, the 505(b)(2) pathway may be used. This approach allows partial reliance on existing data along with new evidence. A CRO for Complex Injectables can guide sponsors in choosing the most efficient and cost-effective option. Keeping up with regulatory guidance is important for making informed decisions.
Clarify your regulatory strategy:Understanding the Regulatory Pathway for Complex Peptide Injectables
Advanced Analytical Infrastructure for Complex Molecule Characterization
A high-quality CRO should act as an advanced analytical center. Standard methods are often not enough for complex injectables. Specialized tools are needed to study multi-component systems and ensure consistency. Investment in modern analytical infrastructure shows a commitment to quality and scientific accuracy. This also increases confidence during regulatory review and submission processes.
Overcome analytical barriers:Addressing Leuprolide Depot Analytical Challenges
Multi-Detector Asymmetric-Flow Field-Flow Fractionation (AF4-MALS)
AF4-MALS is a powerful technique used for nanoparticle analysis. It separates particles based on size without the limitations of traditional columns. When combined with light scattering, it provides detailed data on particle size and distribution. This information is important for maintaining batch consistency and detecting impurities. It also helps identify issues such as aggregation or free drug presence.
High-Resolution Mass Spectrometry (HRMS) and NMR
High-resolution mass spectrometry helps detect and measure impurities at very low levels. These instruments are essential for meeting strict regulatory requirements. NMR provides detailed structural information and is useful for analyzing complex molecules and polymers. Together, these techniques form a strong analytical foundation. They ensure accurate data and support regulatory compliance.
Thermal Analysis and Nano-DSC
Thermal analysis methods like DSC help evaluate product stability and phase behavior. For liposomal systems, these tests show membrane stability and drug encapsulation efficiency. Monitoring thermal properties over time helps predict shelf life. A CRO must be skilled in interpreting these results to address stability concerns early. This improves product reliability and long-term performance.
Bioequivalence Strategies: Beyond Traditional PK Studies
Traditional pharmacokinetic studies may not fully reflect the performance of complex injectables. Alternative approaches are often needed to demonstrate equivalence. A CRO for Complex Injectables should combine in vitro testing with modeling techniques. This approach reduces reliance on clinical trials and speeds up development. It also helps build a stronger scientific case for regulatory approval.
Accelerate your timeline:How CDMOs Accelerate Generic Drug Development in the US & Canada
Model-Integrated Evidence (MIE) and PBPK Modeling
PBPK modeling simulates how a drug behaves in the human body using mathematical models. It connects in vitro data with expected in vivo outcomes. This method is especially useful for long-acting injectables. CROs with expertise in modeling can support regulatory submissions and even reduce the need for human studies. This leads to faster and more cost-effective development.
In Vitro Release Testing (IVRT) and In Vitro Permeation Testing (IVPT)
IVRT and IVPT are important for studying drug release and absorption. These methods must be sensitive and reproducible to detect small formulation differences. Proper method development ensures accurate results that reflect real conditions. Validation of these methods is critical for regulatory acceptance. A CRO must design these studies carefully to ensure reliability.
Critical Selection Criteria: Evaluating a CRO’s Competence
Choosing a CRO involves more than technical skills. Infrastructure, communication, and quality systems also matter. A CRO for Complex Injectables should have modern laboratories and follow strict quality standards. Regular equipment calibration and strong project management ensure timely and accurate results. A dependable partner reduces risks and improves overall project success.
Regulatory Track Record and FDA Inspection History
A CRO’s past performance provides insight into its reliability. A strong inspection history indicates good compliance and quality systems. Reviewing past audit results helps assess readiness for regulatory review. Consistent success in approvals shows a clear understanding of requirements. This builds confidence in the CRO’s capabilities.
Senior Leadership and Escalation Pathways
Clear communication and access to experienced professionals are important for smooth collaboration. Sponsors should be able to connect with senior experts when needed. This helps resolve issues quickly and maintain alignment. CROs with strong leadership often deliver better outcomes and fewer delays.
Quality Assurance and Data Integrity: The 21 CFR Part 11 Standard
Data integrity is essential in pharmaceutical development. Regulatory agencies expect accurate and secure data management. CROs must follow 21 CFR Part 11 guidelines to ensure compliance. This includes secure systems, traceable records, and proper documentation practices. Strong data management builds trust and reduces regulatory risks.
Implementing 21 CFR Part 11 Compliant Systems
Compliant systems include audit trails, user controls, and secure storage. These features ensure that all data is recorded and protected properly. Staff training is also important to maintain consistency in documentation. A CRO must follow strict processes to ensure reliable data. This supports smooth submissions and approvals.
Niche vs. Generalist CROs: Strategic Alignment for Complex Projects
Choosing between niche and generalist CROs depends on project complexity. For advanced products, specialized expertise is often more valuable. Niche CROs focus on specific technologies and offer deeper insights. They are also more flexible in handling complex challenges. Aligning CRO strengths with project needs is key to achieving success.
| In Vitro Method | Objective | Product Applicability |
|---|---|---|
| IVRT | Measure drug release rate into a receptor medium | Microspheres; liposomes; emulsions |
| IVPT | Measure drug permeation across a membrane | Topical and transdermal parenterals |
| PBPK Modeling | Simulate in vivo PK from in vitro parameters | Locally acting injectables; OINDPs |
| Nano-DSC | Characterize thermal stability of vesicles | Liposomal injections |
Optimizing for the Latest AI Overview and Search Environments
Technical content should be clear, structured, and easy to understand. Well-organized information improves visibility in search engines and AI-driven platforms. CROs should present their capabilities in a simple and transparent way. This builds trust and improves engagement with potential clients. Strong digital presence also reflects expertise and credibility.
Representing E-E-A-T Through Technical Transparency
Transparency and expertise help build long-term trust. CROs should share their experience, methods, and case studies openly. This allows clients to understand their capabilities clearly. Honest communication strengthens partnerships and supports better decisions. It also improves credibility in the industry.
The Future of Complex Generic Parenterals and International Harmonization
Global efforts are improving the development of complex injectables. New guidelines aim to simplify regulatory requirements across regions. This helps companies bring products to market faster. CROs must stay updated with these changes to remain competitive. Being proactive ensures readiness for future challenges.
Expanding Access Through Collaboration
Collaboration between industry, regulators, and academia is driving innovation. These partnerships help solve complex scientific challenges. CROs involved in such efforts gain valuable insights. This allows them to offer better solutions to sponsors. Ultimately, it improves access to important medicines.
Conclusion: Strategic Mastery in Complex Generic Injectable Selection
Selecting the right CRO for Complex Injectables plays a major role in successful drug development. A strong partner brings technical expertise, regulatory knowledge, and advanced analytical capabilities. By focusing on quality, consistency, and compliance, companies can reduce risks and improve outcomes. The right CRO helps turn complex science into real patient solutions.
For expert assistance in navigating the complexities of your next parenteral project, please contact us today.
Contact us: https://resolvemass.ca/contact/
Frequently Asked Questions (FAQs)
The 505(j) pathway requires a generic product to closely match the reference drug in composition and performance. For complex injectables like liposomes or iron colloids, this becomes difficult because the manufacturing process directly affects the final product. Developers must provide detailed Q3 characterization and sensitive in vitro testing to prove similarity. These requirements are much more demanding than those for simple oral medicines.
AF4-MALS is a gentle and highly accurate method for analyzing particle size and structure. Unlike traditional chromatography, it does not use columns, which helps avoid sample damage or unwanted interactions. When combined with light scattering, it provides precise data on iron core size and coating thickness. These parameters are essential for ensuring product quality, safety, and bioequivalence.
PLGA end-caps play a major role in how the polymer behaves inside the body. They influence water absorption and the rate at which the polymer breaks down. Differences in end-cap chemistry between a generic and reference product can change drug release patterns. Accurate analysis using techniques like NMR and GPC helps ensure consistent and safe performance.
Nano-DSC is used to study small thermal changes in liposomal membranes. It helps identify key properties such as phase transition temperature, which indicates membrane stability. By tracking these changes, scientists can detect early signs of instability or variation between batches. This makes nano-DSC a valuable tool for maintaining consistent product quality.
The ICH M13A and M13B guidelines aim to create consistent bioequivalence standards across global markets. They introduce concepts like biowaivers for additional strengths, reducing the need for repeated in vivo studies. This helps companies save time and resources when developing multiple product strengths. CROs must align their strategies with these updates to stay competitive.
Regulatory agencies require very low limits for harmful impurities such as nitrosamines and PFAS. Even trace amounts can raise safety concerns and lead to regulatory action. A CRO for Complex Injectables must use highly sensitive instruments like Orbitrap mass spectrometry to detect these compounds. This ensures patient safety and supports successful regulatory submissions.
During an audit, sponsors should review the condition and calibration of key instruments like NMR, HRMS, and specialized manufacturing tools. It is also important to check data management systems for compliance with 21 CFR Part 11. Staff training records and standard operating procedures should be carefully evaluated. These factors help confirm the CRO’s ability to maintain quality and consistency.
Reference:
- Chaudhari, P. B., & Banga, A. (2023). Writing strategies for improving the access of medical literature. World Journal of Emergency Medicine, 13(3), 50–58. https://pmc.ncbi.nlm.nih.gov/articles/PMC9376055/
- Exploring the challenges faced by generic version of complex drugs. (2025). Journal of Pharmaceutical Policy and Practice. https://pmc.ncbi.nlm.nih.gov/articles/PMC12482112/
- U.S. Food and Drug Administration. (2022). Focus area: Increasing access to complex generic drug products. https://www.fda.gov/science-research/focus-areas-regulatory-science-report/focus-area-increasing-access-complex-generic-drug-products
- U.S. Food and Drug Administration. (n.d.). Helpful webinars and other resources for generic drug manufacturers. https://www.fda.gov/drugs/abbreviated-new-drug-application-anda/helpful-webinars-and-other-resources-generic-drug-manufacturers
- U.S. Food and Drug Administration. (2024). Complex generic drug product development: Regulatory science priorities and approaches. https://www.fda.gov/media/187995/download
- U.S. Food and Drug Administration. (2024). CDER’s OGD and EMA’s parallel scientific advice pilot program for complex generics works to increase harmonization and bring generic drugs to patients. https://www.fda.gov/drugs/our-perspective/cders-ogd-and-emas-parallel-scientific-advice-pilot-program-complex-generics-works-increase
- Kumar, A., & Bansal, A. (2023). The power of field-flow fractionation in characterization of complex drug delivery systems. Pharmaceutics, 15(6), 1680. https://pmc.ncbi.nlm.nih.gov/articles/PMC10224342/



