Introduction
In the dynamic landscape of the pharmaceutical industry, the development of effective and innovative products is paramount. Companies are continuously seeking advanced formulation strategies to enhance the efficacy, safety, and patient adherence of their therapeutic products. Custom product formulation development services play a crucial role in this process, offering tailored solutions that address specific therapeutic needs and market demands. Resolvemass Laboratories, a leading Contract Research Organization (CRO) in custom synthesis and analytical services, excels in providing bespoke formulation development services that drive pharmaceutical innovation and success.
Share via:
Article Summary
- Custom product formulation development helps create safe, effective, and patient-friendly pharmaceutical products tailored to specific therapeutic needs.
- Advanced technologies and drug delivery systems improve drug stability, bioavailability, and treatment outcomes.
- The formulation process includes key stages such as preformulation studies, formulation design, optimization, analytical testing, and stability evaluation.
- Quality by Design (QbD), proper excipient selection, and suitable packaging ensure consistent product quality and regulatory compliance.
- Real-world formulation strategies help overcome challenges like poor solubility, stability issues, and taste masking in specialized formulations.
Emerging Trends in Pharmaceutical Formulation Development
The pharmaceutical industry is continuously evolving with the integration of new technologies and scientific approaches in formulation development. Modern formulation strategies increasingly incorporate advanced delivery systems, nanotechnology, and personalized medicine concepts to improve drug performance and therapeutic outcomes. These innovations allow scientists to address long-standing challenges such as poor solubility, rapid drug degradation, and limited bioavailability of certain active pharmaceutical ingredients.
Additionally, digital tools and data-driven research methods are transforming the way formulation development is conducted. Computational modeling, artificial intelligence, and predictive analytics help scientists evaluate formulation behavior before laboratory testing begins. This significantly reduces development time while improving accuracy in selecting excipients, delivery mechanisms, and dosage forms, ultimately accelerating the path from concept to commercialization.
Ensure your product meets all regulatory and manufacturing standards. Explore our Chemistry, Manufacturing, and Controls (CMC) Services today.
The Significance of Custom Product Formulation Development
Custom product formulation development is the process of designing and creating pharmaceutical products with specific characteristics tailored to meet unique therapeutic requirements and patient needs. This process is essential for several reasons:
- Enhancing Drug Efficacy and Safety: Custom formulations can improve the bioavailability and stability of active pharmaceutical ingredients (APIs), ensuring that they are delivered effectively to the target site within the body. This can result in better therapeutic outcomes and reduced side effects.
- Improving Patient Compliance: Formulations can be designed to enhance patient adherence to treatment regimens. For example, extended-release formulations can reduce the frequency of dosing, making it easier for patients to follow their prescribed treatments.
- Addressing Specific Therapeutic Needs: Custom formulations allow for the development of specialized products for niche markets or specific patient populations, such as pediatric or geriatric patients, who may require different dosage forms or strengths.
- Expanding Product Portfolios: Pharmaceutical companies can diversify their product offerings by developing new formulations of existing drugs, creating combination products, or exploring new delivery routes.
Role of Advanced Drug Delivery Systems
Advanced drug delivery systems have become a critical component of modern formulation development. These systems are designed to control how and where a drug is released within the body, improving therapeutic efficiency while minimizing unwanted side effects. Technologies such as liposomal carriers, polymeric nanoparticles, and transdermal patches allow drugs to be delivered more precisely, ensuring optimal absorption and targeted action.
By integrating advanced delivery technologies into custom formulations, pharmaceutical developers can enhance drug stability and improve patient outcomes. Controlled-release mechanisms, for example, enable medications to maintain therapeutic levels in the bloodstream over extended periods, reducing dosing frequency and improving patient convenience. Such innovations are particularly beneficial for chronic disease treatments where consistent drug exposure is essential for effective management.
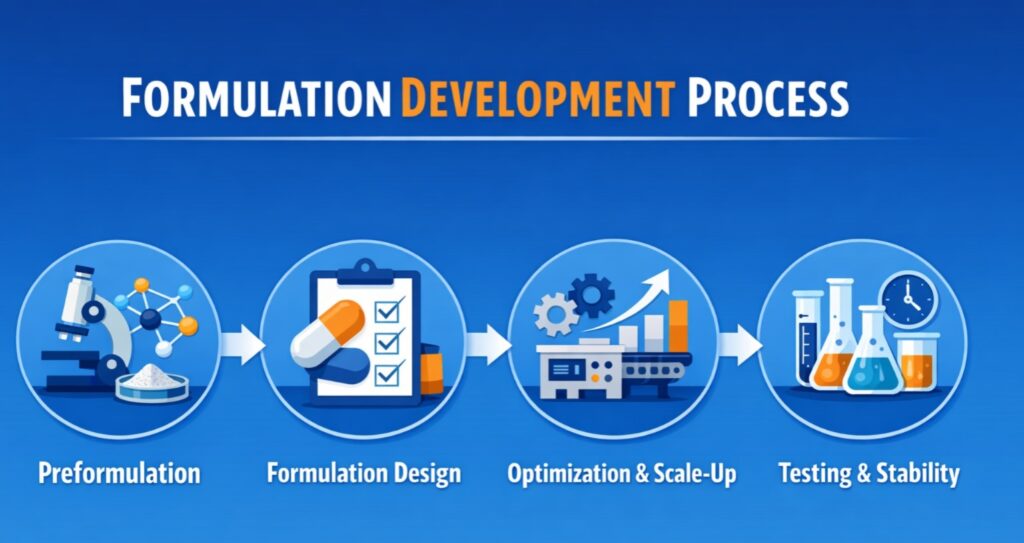
The Custom Formulation Development Process
Custom formulation development involves several key stages, each critical to ensuring the final product meets the desired specifications and regulatory requirements. At Resolvemass Laboratories, we follow a comprehensive and systematic approach to formulation development:
1. Preformulation Studies
The formulation development process begins with preformulation studies, which involve the characterization of the API and other excipients. These studies provide essential information on the physicochemical properties of the API, such as solubility, stability, and compatibility with excipients. Understanding these properties is crucial for designing an effective and stable formulation.
2. Formulation Design and Development
Based on the preformulation data, our formulation scientists design prototype formulations. This involves selecting appropriate excipients, determining the optimal drug-to-excipient ratio, and choosing the suitable dosage form (e.g., tablets, capsules, liquids, or topical formulations). Various formulation strategies, such as solid dispersions, liposomes, or nanoparticles, may be explored to enhance the bioavailability and stability of the API.
3. Optimization and Scale-Up
Once the prototype formulations are developed, they undergo optimization to fine-tune their characteristics. This may involve adjusting the formulation composition, processing parameters, or manufacturing techniques to achieve the desired properties. Scale-up studies are then conducted to ensure the formulation can be manufactured consistently and efficiently on a larger scale.
Importance of Quality by Design (QbD) in Formulation Development
Quality by Design (QbD) has emerged as a fundamental principle in pharmaceutical development, emphasizing a systematic approach to designing formulations that meet predefined quality objectives. Rather than relying solely on end-product testing, QbD focuses on understanding the relationship between formulation components, manufacturing processes, and product performance. This approach ensures that quality is built into the product from the earliest stages of development.
Implementing QbD involves identifying critical quality attributes (CQAs), critical material attributes (CMAs), and critical process parameters (CPPs). By thoroughly studying these variables, scientists can establish robust formulations that consistently deliver the desired therapeutic effect. This proactive methodology not only improves product reliability but also facilitates smoother regulatory approvals by demonstrating a clear understanding of the formulation process.
4. Analytical Method Development and Validation
Accurate and reliable analytical methods are essential for evaluating the quality and performance of the formulation. At Resolvemass Laboratories, we develop and validate robust analytical methods to assess critical quality attributes such as assay, dissolution, content uniformity, and stability. These methods ensure that the formulation meets the required specifications and regulatory standards.
Need precise testing for your new formulation? Learn more about our Analytical Method Development and Validation Service.
Importance of Excipients in Custom Formulation
Excipients play a vital role in pharmaceutical formulations by supporting the stability, effectiveness, and usability of the final product. Although they do not possess therapeutic activity themselves, excipients contribute significantly to the drug’s performance by influencing factors such as solubility, absorption, and shelf life. The careful selection of excipients is therefore a critical aspect of custom formulation development.
In addition to improving stability and bioavailability, excipients can enhance patient experience through taste masking, improved texture, or easier administration. For instance, suspending agents help maintain uniform distribution of active ingredients in liquid formulations, while disintegrants enable tablets to break down quickly once ingested. The strategic use of excipients ensures that pharmaceutical products remain safe, effective, and patient-friendly.
5. Stability Studies
Stability studies are conducted to evaluate the shelf life and storage conditions of the formulation. These studies involve subjecting the formulation to various environmental conditions, such as temperature, humidity, and light, to assess its stability over time. Stability data is crucial for determining the appropriate packaging and storage requirements for the final product.
Detect and quantify trace contaminants with our specialized Impurity Profiling Using LCMS.
Packaging Considerations in Formulation Development
Packaging plays an essential role in maintaining the stability and integrity of pharmaceutical formulations throughout their shelf life. The choice of packaging materials must protect the product from environmental factors such as moisture, light, oxygen, and temperature fluctuations. In many cases, specialized packaging solutions are required to preserve the chemical and physical stability of sensitive formulations.
Moreover, modern pharmaceutical packaging must also address usability and safety concerns. Child-resistant closures, tamper-evident seals, and unit-dose packaging are commonly implemented to ensure patient safety and regulatory compliance. Proper packaging not only safeguards the product but also enhances convenience and accessibility for patients and healthcare providers.
6. Regulatory Support
Navigating the regulatory landscape is a critical aspect of formulation development. Our regulatory experts provide comprehensive support throughout the development process, ensuring that the formulation complies with relevant regulatory guidelines and requirements. This includes preparing and submitting the necessary documentation for regulatory approvals and providing guidance on Good Manufacturing Practices (GMP).
Case Study: Development of a Pediatric Oral Suspension
One of our recent projects involved the development of a pediatric oral suspension for an existing API with poor solubility and taste masking challenges. The objective was to create a palatable and stable formulation that could be easily administered to children.
Challenges
- Poor Solubility: The API exhibited poor water solubility, leading to low bioavailability.
- Unpleasant Taste: The bitter taste of the API made it difficult for children to tolerate.
- Stability Issues: The API was prone to degradation in aqueous solutions, requiring careful formulation to ensure stability.
Formulation Strategy
- Solubility Enhancement: To address the solubility issue, we employed a combination of solubilization techniques, including the use of solubilizing agents and the development of a solid dispersion system. This approach significantly improved the solubility and bioavailability of the API.
- Taste Masking: To mask the bitter taste, we incorporated taste-masking agents and flavoring agents into the formulation. Additionally, the use of microencapsulation technology helped to further reduce the perception of bitterness.
- Stabilization: To enhance the stability of the API in the aqueous suspension, we optimized the pH of the formulation and added appropriate stabilizers. These adjustments ensured the formulation remained stable throughout its shelf life.
Outcome
The final pediatric oral suspension exhibited excellent solubility, a pleasant taste, and robust stability. In vitro and in vivo studies demonstrated improved bioavailability and patient acceptability, making it a successful formulation development project.
Get unparalleled clarity on your molecular structures with our High-Resolution Mass Spectrometry (HRMS) Analysis.
Future Outlook for Custom Formulation Development
As pharmaceutical research continues to advance, the demand for customized formulation solutions is expected to grow significantly. Emerging therapeutic areas such as biologics, gene therapies, and personalized medicine require highly specialized formulation strategies that go beyond traditional approaches. These complex therapies often require innovative delivery technologies and precise formulation techniques to ensure stability and effectiveness.
Furthermore, collaboration between pharmaceutical companies, research institutions, and contract research organizations will play an increasingly important role in accelerating innovation. By combining scientific expertise, advanced technologies, and regulatory knowledge, organizations can develop next-generation pharmaceutical products more efficiently. This collaborative environment will continue to drive progress in formulation science and improve patient care worldwide.
Conclusion
Custom product formulation development is a critical component of modern pharmaceutical research and innovation. By carefully designing formulations that address specific therapeutic requirements, pharmaceutical companies can enhance drug effectiveness, improve patient adherence, and expand treatment possibilities across diverse patient populations. A well-structured formulation development process ensures that every stage—from preformulation studies to stability testing—is guided by scientific precision and regulatory compliance.
At Resolvemass Laboratories, our expertise in custom synthesis, analytical testing, and formulation science enables us to deliver tailored solutions that support successful pharmaceutical product development. Through advanced technologies, strategic research approaches, and a commitment to quality, we help pharmaceutical partners transform promising drug candidates into safe, effective, and market-ready therapeutic products. Our goal is to contribute to the advancement of healthcare by supporting innovative pharmaceutical solutions that improve patient outcomes worldwide.
Contact Us For more information about our custom product formulation development services and how we can assist in your pharmaceutical projects, please visit Resolvemass laboratories & contact us
FAQs on Custom Product Formulation Development Services
Formulation development is essential because it determines how a drug performs in the body. A well-developed formulation improves drug absorption, stability, and therapeutic efficiency. It also ensures that the medication is convenient for patients to use, which ultimately contributes to better treatment outcomes and product success.
Scientists consider multiple factors such as the physicochemical properties of the active ingredient, compatibility with excipients, dosage form requirements, and patient needs. Stability, bioavailability, and manufacturing feasibility are also evaluated. These considerations ensure the final product remains safe, effective, and consistent throughout its shelf life.
Contract research organizations (CROs) provide specialized expertise, advanced laboratories, and technical resources to support pharmaceutical development. They assist with formulation design, analytical testing, stability studies, and regulatory documentation. This allows pharmaceutical companies to accelerate product development while maintaining high quality standards
Excipients are inactive ingredients that support the performance of the active pharmaceutical ingredient. They help improve drug stability, enhance solubility, control drug release, and make the product easier for patients to use. Proper selection of excipients is crucial for maintaining formulation quality and effectiveness.
Common challenges include poor solubility of active ingredients, stability issues, taste masking difficulties, and achieving consistent drug release. Scientists must also ensure compatibility between formulation components and meet strict regulatory requirements. Overcoming these challenges often requires innovative formulation strategies and extensive testing.
Reference:
- Chang, R. K., Raw, A., Lionberger, R., & Yu, L. (2013). Generic development of topical dermatologic products: Formulation development, process development, and testing of topical dermatologic products. The AAPS Journal, 15(1), 41–52. https://doi.org/10.1208/s12248-012-9411-0
- Latha, P., & Sailaja, B. (2014). Bioanalytical method development and validation by HPLC: A review. Journal of Medical and Pharmaceutical Innovation, 1(6S), 1–9. View article PDF
- Tihhonova, M., Sjöholm, E., Mathiyalagan, R., Lindfors, L., Wang, Q., Wang, X., & Sandler, N. (2024). The formulation and evaluation of customized prednisolone gel tablets prepared by automated extrusion-based material deposition for veterinary applications. Pharmaceutics, 16, Article 678. https://pmc.ncbi.nlm.nih.gov/articles/PMC11677990/



