Introduction
The management of diabetes has traditionally relied on injectable insulin, which poses challenges related to patient compliance and quality of life. Oral insulin delivery presents a compelling alternative, potentially simplifying treatment regimens and improving patient adherence. However, the oral delivery of insulin is fraught with obstacles, including degradation in the gastrointestinal (GI) tract and poor absorption through the intestinal lining. Cyclodextrin-based dendrimers have emerged as a promising solution to these challenges, offering innovative approaches to enhance the stability, bioavailability, and efficacy of orally administered insulin. As a leading Contract Research Organization (CRO) specializing in custom synthesis and analytical services, Resolvemass Laboratories is at the forefront of advancing this technology. This blog explores the potential of cyclodextrin-based dendrimers for oral insulin delivery, examining their structure, benefits, challenges, and future prospects.
If you’re looking to collaborate with a trusted CRO on innovative drug delivery technologies, here’s why ResolveMass is the ideal partner.
Key Highlights
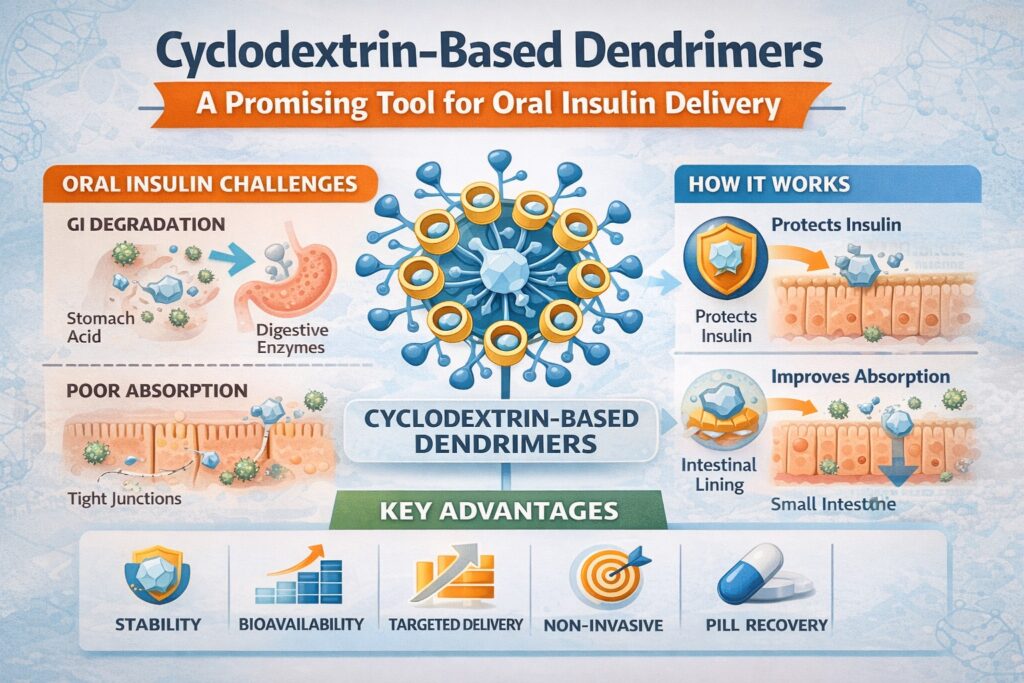
- Oral insulin offers a non-invasive alternative but faces major GI stability and absorption challenges.
- Cyclodextrin-based dendrimers protect insulin, enhance bioavailability, and improve intestinal uptake.
- Their tunable structure enables controlled release, targeted delivery, and enzyme protection.
- Challenges include formulation optimization, regulatory approval, scalability, and long-term safety.
- Advances in nanotechnology and personalized medicine continue to drive clinical potential.
Understanding Cyclodextrin-Based Dendrimers
1. Structure and Composition
Cyclodextrin-based dendrimers are sophisticated nanocarriers combining cyclodextrins and dendrimer architectures. Cyclodextrins are cyclic oligosaccharides with a hydrophobic central cavity capable of encapsulating various molecules, while dendrimers are highly branched macromolecules with a core, branching units, and surface functional groups.
- Cyclodextrin Units: These units are used to encapsulate insulin molecules, protecting them from enzymatic degradation and improving their stability.
- Dendrimer Core and Branches: The dendrimer core provides a scaffold for attaching cyclodextrin units, creating a highly branched, functionalized structure that enhances drug loading and release control.
Understand the difference between step-growth and chain-growth methods – A Comprehensive Guide to Polymerization Techniques
2. Mechanism of Action
Cyclodextrin-based dendrimers enhance oral insulin delivery by protecting insulin from the acidic environment of the stomach and digestive enzymes. The encapsulated insulin within the cyclodextrin cavities is shielded from degradation, allowing it to reach the intestinal tract intact. The dendrimer’s branched structure also facilitates improved permeability through the intestinal epithelium, potentially enhancing systemic absorption.
For deeper context on how materials like dendrimers are designed with purpose, explore monomer selection strategies.
3. Role of Surface Functionalization in Enhancing Oral Insulin Transport
Surface functionalization plays a critical role in determining how cyclodextrin-based dendrimers interact with biological barriers in the gastrointestinal tract. By modifying terminal groups with functional moieties such as amino acids, peptides, or polyethylene glycol (PEG), researchers can significantly influence mucoadhesion, cellular uptake, and transport efficiency. These modifications help dendrimers evade premature clearance while maintaining prolonged contact with intestinal epithelial cells, increasing the likelihood of insulin absorption.
Additionally, functionalized dendrimers can be engineered to respond to environmental triggers such as pH or enzymatic activity. For oral insulin delivery, pH-responsive surface groups allow dendrimers to remain stable in the stomach while releasing insulin in the more neutral intestinal environment. This tunable behavior improves delivery precision and reduces variability in absorption, a key challenge in oral peptide therapeutics.
4. Interaction with Intestinal Mucus and Epithelial Barriers
One of the most formidable barriers to oral insulin delivery is the intestinal mucus layer, which can trap or repel large biomolecules. Cyclodextrin-based dendrimers can be optimized to penetrate this mucus layer by adjusting surface charge and hydrophilicity. Neutral or slightly positive surface characteristics enable dendrimers to diffuse through mucus without becoming immobilized, improving transit toward epithelial cells.
Beyond mucus penetration, dendrimers can interact with tight junctions between epithelial cells. Certain formulations promote transient and reversible opening of tight junctions, enabling paracellular transport of insulin. Importantly, this process must be tightly controlled to avoid long-term disruption of intestinal integrity. Advances in dendrimer design have made it possible to enhance permeability while maintaining epithelial safety.
Advantages of Cyclodextrin-Based Dendrimers for Oral Insulin Delivery
1. Enhanced Stability
One of the primary challenges in oral insulin delivery is the degradation of insulin in the acidic environment of the stomach. Cyclodextrin-based dendrimers offer a protective barrier that prevents insulin degradation, ensuring that a significant portion of the insulin reaches the intestinal tract where absorption can occur. The cyclodextrin cavities effectively shield insulin from enzymatic breakdown, maintaining its biological activity.
2. Improved Bioavailability
The dendrimer structure allows for high loading capacities and controlled release of insulin. This controlled release can improve the bioavailability of insulin, ensuring a more consistent and effective therapeutic response. The cyclodextrin-based dendrimers can also be engineered to enhance the permeability of insulin across the intestinal barrier, further increasing its systemic absorption.
3. Targeted Delivery
Cyclodextrin-based dendrimers can be functionalized with specific targeting moieties that facilitate targeted delivery to certain regions of the gastrointestinal tract. This targeting capability can be particularly beneficial for optimizing insulin absorption in the intestine, maximizing therapeutic efficacy while minimizing potential side effects.
For more on how emerging technologies support this, read about Emerging Trends in Custom Polymer Synthesis for 2025 and Beyond
4. Non-Invasive Administration
The potential for oral administration of insulin via cyclodextrin-based dendrimers eliminates the need for injections, offering a non-invasive and more convenient alternative for diabetes management. This improvement can significantly enhance patient compliance and overall quality of life.
5. Protection Against Enzymatic Degradation in the Intestine
While gastric acidity poses a significant threat to insulin stability, proteolytic enzymes in the intestine present an equally serious challenge. Enzymes such as trypsin and chymotrypsin rapidly degrade free insulin molecules, drastically reducing bioavailability. Cyclodextrin-based dendrimers mitigate this risk by physically shielding insulin within their molecular architecture, limiting direct enzyme access.
This protective mechanism is further enhanced when dendrimers are designed with steric hindrance or enzyme-resistant surface coatings. Such features slow enzymatic interaction without inhibiting normal digestive processes. As a result, insulin retains its structural integrity long enough to be absorbed, improving consistency in therapeutic outcomes across patient populations.
Challenges and Considerations
1. Formulation and Stability
Developing an effective oral insulin formulation using cyclodextrin-based dendrimers requires careful optimization of the dendrimer composition, size, and surface functionalization. Achieving the right balance between stability, release kinetics, and bioavailability is crucial for the success of the formulation.
2. Regulatory Approval
The approval of new oral insulin formulations using cyclodextrin-based dendrimers involves rigorous testing and validation to ensure safety and efficacy. This process includes preclinical studies, clinical trials, and regulatory reviews, all of which must be addressed to bring the technology to market.
3. Cost and Scalability
The synthesis of cyclodextrin-based dendrimers can be complex and costly, potentially impacting the overall cost of the oral insulin formulation. Scaling up the production process while maintaining quality and cost-effectiveness is a key consideration for commercial viability.
To understand where the market is headed, read this – Global Market Insights: The Future of Custom Polymer Synthesis
4. Safety, Biocompatibility, and Long-Term Toxicity Considerations
Although cyclodextrin-based dendrimers offer promising delivery advantages, long-term safety remains a critical consideration. Repeated oral administration requires materials that are non-toxic, non-immunogenic, and capable of safe elimination from the body. Studies evaluating dendrimer size, surface charge, and biodegradability have shown that these parameters strongly influence biocompatibility.
Chronic exposure assessments are especially important for diabetes therapies, which involve lifelong treatment. Ongoing research focuses on developing biodegradable dendrimer frameworks that break down into non-harmful metabolites after insulin delivery. Such advancements are essential to ensuring regulatory acceptance and patient safety while maintaining therapeutic effectiveness.
5. Manufacturing Consistency and Quality Control Challenges
Achieving batch-to-batch consistency is a major hurdle in the commercialization of cyclodextrin-based dendrimers. Minor variations in synthesis conditions can affect dendrimer size distribution, surface functionality, and insulin loading efficiency. These inconsistencies can lead to variability in drug release profiles and therapeutic performance.
To address this, robust analytical characterization and quality control strategies are required throughout development and scale-up. Techniques such as high-resolution mass spectrometry, chromatographic profiling, and particle size analysis play a critical role in ensuring reproducibility. Strong quality frameworks not only support regulatory submissions but also build confidence in large-scale manufacturing feasibility.
Future Prospects
1. Advancements in Nanotechnology
Ongoing research in nanotechnology and material science continues to enhance the capabilities of cyclodextrin-based dendrimers. Innovations in dendrimer synthesis, functionalization, and formulation techniques are likely to further improve the effectiveness and feasibility of oral insulin delivery.
2. Integration with Other Technologies
Combining cyclodextrin-based dendrimers with other drug delivery technologies, such as nanoparticles or micelles, may offer additional benefits in terms of targeting, release control, and therapeutic efficacy. Exploring these combinations can lead to new and improved oral insulin formulations.
3. Personalized Medicine
As the field of personalized medicine advances, cyclodextrin-based dendrimers can be tailored to individual patient needs, optimizing insulin delivery based on specific metabolic profiles and treatment requirements. This approach has the potential to enhance therapeutic outcomes and patient satisfaction.
Read how custom polymer synthesis enables this level of flexibility.
4. Clinical Translation and Patient-Centric Formulation Design
Translating cyclodextrin-based dendrimer systems from laboratory research to clinical application requires a patient-centric approach to formulation design. Factors such as dosing frequency, pill size, and food interactions must be carefully evaluated to ensure usability in real-world settings. Oral insulin formulations must align with patient lifestyles to truly improve adherence.
Clinical translation also demands well-designed pharmacokinetic and pharmacodynamic studies to correlate dendrimer performance with glycemic control. These studies help establish optimal dosing regimens and identify patient subgroups that may benefit most. As clinical evidence grows, dendrimer-based oral insulin could become a transformative alternative to injectable therapies.
Conclusion
Cyclodextrin-based dendrimers represent a promising advancement in the quest for effective oral insulin delivery. Their ability to protect insulin from degradation, enhance bioavailability, and enable non-invasive administration addresses key challenges in diabetes management. At Resolvemass Laboratories, we are dedicated to advancing these innovative technologies through our expertise in custom synthesis and analytical services. By leveraging the potential of cyclodextrin-based dendrimers, we aim to contribute to the development of improved therapeutic solutions for diabetes, ultimately enhancing patient care and quality of life. The continued exploration and optimization of this technology hold great promise for the future of oral drug delivery.
Reference
- Arima, H., & Motoyama, K. (2012). Polyamidoamine dendrimer conjugates with cyclodextrins for drug and gene delivery. Journal of Controlled Release, 164(3), 183-192. https://doi.org/10.1016/j.jconrel.2012.02.023
- Wehl, L., Muggli, K., Möller, K., Engelke, H., & Bein, T. (2025). Cross-linked cyclodextrin-based nanoparticles as drug delivery vehicles: Synthesis strategy and degradation studies. ACS Omega, 10(10), 10352–10365. https://doi.org/10.1021/acsomega.4c10200

