
Introduction:
De Novo GLP-1 Peptide Sequencing Accuracy is a critical factor in modern peptide drug development, especially for complex GLP-1 analogs used in metabolic disorder therapies. In simple terms, it defines how precisely we can determine a peptide’s amino acid sequence without relying on reference databases.
For pharmaceutical companies, CROs, and regulatory bodies, sequencing accuracy directly impacts drug safety, efficacy validation, and approval success.
At ResolveMass Laboratories Inc., advanced high-resolution mass spectrometry (HRMS) platforms are used to deliver high-confidence results, as detailed in their
GLP-1 peptide sequencing services and GLP-1 drug sequencing solutions.
Share via:
Summary:
- De Novo GLP-1 Peptide Sequencing Accuracy can reach 95–99% under optimized HRMS conditions
- Accuracy depends on instrument resolution, fragmentation quality, and peptide complexity
- GLP-1 analogs pose challenges due to modifications, isomers, and sequence length
- Combining LC-MS/MS + HRMS + bioinformatics tools significantly improves confidence
- Essential for regulatory submissions, biosimilars, and impurity characterization
For advanced analytical support, explore
GLP-1 peptide sequencing CRO services or
CRO solutions for GLP-1 peptide characterization.
1: What is De Novo Sequencing and How Accurate is It?
De novo sequencing determines the amino acid sequence directly from MS/MS spectra, with typical accuracy ranging from 85% to 99% depending on experimental conditions.
Unlike database-dependent approaches, de novo sequencing reconstructs peptide sequences purely from fragmentation patterns, making it indispensable for analyzing:
- Novel peptide drugs
- Unknown impurities
- Modified GLP-1 analogs
This capability is especially valuable in pharmaceutical research where reference sequences may not be available.
This approach is a core component of analytical characterization of GLP-1 peptide drugs.
Typical Accuracy Range
| Condition | Expected Accuracy |
|---|---|
| Standard LC-MS/MS | 85–92% |
| Optimized HRMS (Orbitrap/QTOF) | 92–97% |
| Advanced workflows (multi-fragmentation + software) | 95–99% |
Key Insight:
Accuracy improves significantly when combining HRMS, multi-fragmentation, and expert validation, as described in
GLP-1 peptide sequencing analytical techniques.
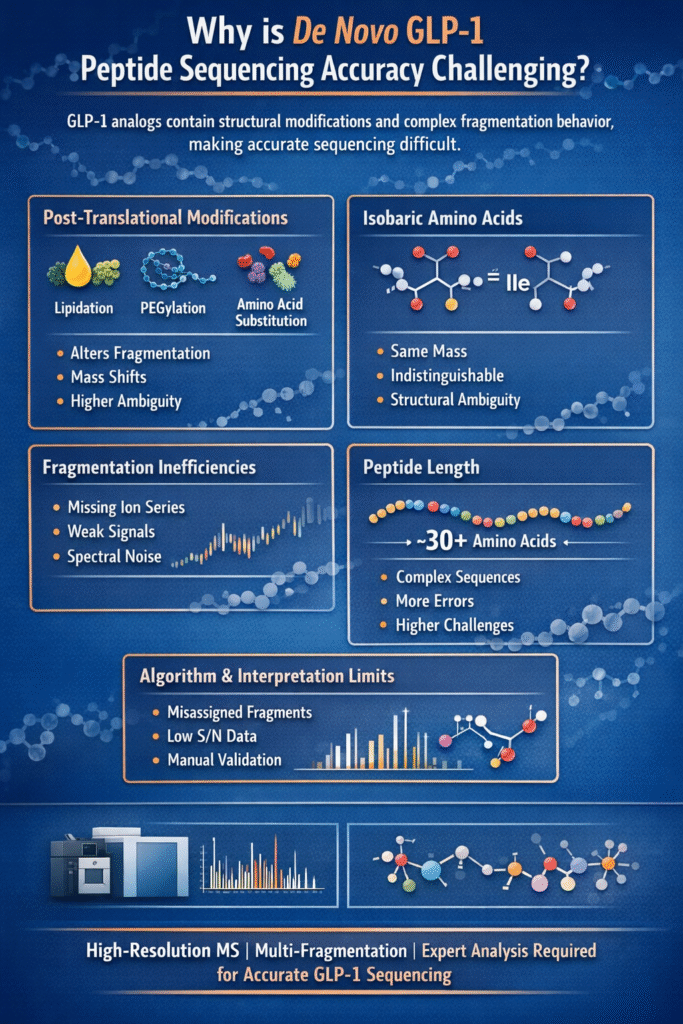
2: Why is De Novo GLP-1 Peptide Sequencing Accuracy Challenging?
De Novo GLP-1 Peptide Sequencing Accuracy is challenging because GLP-1 analogs contain structural modifications and complex fragmentation behavior, making precise sequence reconstruction more difficult than standard peptides.
These complexities require advanced instrumentation, optimized workflows, and expert interpretation to achieve high-confidence results.
Detailed challenges are discussed in GLP-1 peptide sequencing challenges.
Key Challenges
1. Post-Translational Modifications (PTMs)
GLP-1 analogs frequently contain chemical modifications that disrupt standard fragmentation patterns.
Common modifications include:
- Lipidation (e.g., fatty acid chains)
- PEGylation
- Amino acid substitutions
These changes alter ion fragmentation behavior, making it harder for algorithms to correctly assign sequences and reducing prediction clarity.
2. Isobaric Amino Acids
Certain amino acids have identical masses, making them indistinguishable in standard MS analysis.
- Leucine (Leu) vs Isoleucine (Ile)
- Same mass, different structures
Without advanced fragmentation techniques or orthogonal methods, distinguishing between these residues remains a major limitation.
3. Fragmentation Inefficiencies
Incomplete or weak fragmentation reduces sequence coverage and confidence.
Common issues:
- Missing b/y ion series
- Low-intensity fragment ions
- Spectral noise interference
This leads to gaps in sequence data, increasing ambiguity in de novo interpretation.
4. Peptide Length
GLP-1 analogs are relatively long peptides (~30+ amino acids), increasing sequencing complexity.
- Longer sequences produce more fragmentation possibilities
- Higher chance of incomplete coverage
- Increased cumulative error probability
As peptide length increases, maintaining high De Novo GLP-1 Peptide Sequencing Accuracy becomes significantly more challenging.
Key Insight
Achieving high accuracy in GLP-1 sequencing requires integrating high-resolution MS, multiple fragmentation strategies, and expert validation to overcome these inherent analytical challenges.
3: How to Improve De Novo GLP-1 Peptide Sequencing Accuracy
De Novo GLP-1 Peptide Sequencing Accuracy can be significantly improved by combining advanced instrumentation, optimized workflows, and expert data interpretation.
Improving accuracy requires a multi-layered analytical strategy supported by LC-MS characterization of GLP-1 peptides.
Achieving high-confidence results requires a multi-layered analytical approach, where technology and expertise work together to minimize ambiguity and maximize sequence coverage.
Best Practices Used by Experts
1. High-Resolution Mass Spectrometry (HRMS)
HRMS enhances accuracy by providing precise mass measurements.
- Enables exact mass determination
- Improves fragment ion identification
- Reduces ambiguity in sequence assignment
Impact: Higher confidence in identifying correct amino acid sequences
2. Multi-Fragmentation Techniques
Using multiple fragmentation methods improves sequence coverage and reliability.
Common techniques:
- CID (Collision-Induced Dissociation)
- HCD (Higher-energy C-trap Dissociation)
- ETD (Electron Transfer Dissociation)
Impact:
- Generates complementary ion series
- Improves detection of modified regions
- Enhances overall sequencing depth
3. Optimized Sample Preparation
Clean and well-prepared samples are critical for accurate sequencing.
- Efficient peptide purification
- Removal of contaminants and salts
- Reduction of matrix interference
Impact:
- Improves signal quality
- Enhances fragmentation efficiency
- Reduces spectral noise
4. Advanced Bioinformatics Tools
Modern software tools play a key role in improving sequencing accuracy.
- Automated de novo sequence prediction
- Confidence scoring algorithms
- PTM identification capabilities
Impact:
- Faster data processing
- Reduced human error
- Improved reproducibility
5. Manual Validation by Experts
Expert review remains essential for achieving regulatory-grade accuracy.
- Verification of ambiguous residues
- Interpretation of complex spectra
- Cross-checking algorithm outputs
Impact:
- Resolves uncertainties (e.g., Leu/Ile ambiguity)
- Ensures high-confidence final results
- Meets regulatory expectations

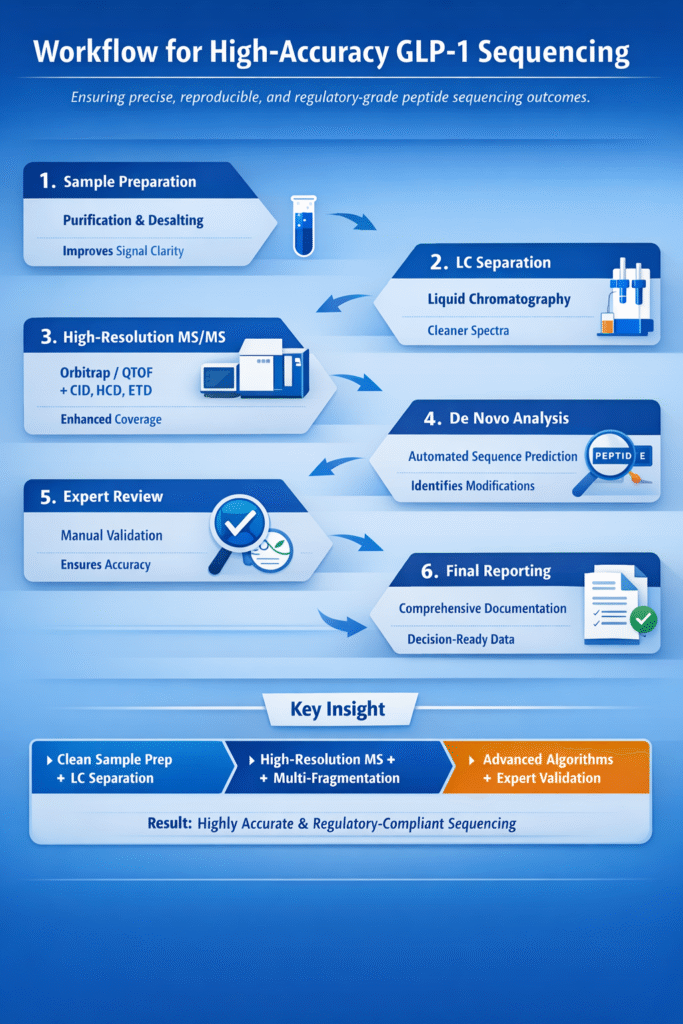
4: Workflow for High-Accuracy GLP-1 Sequencing
A robust and well-optimized workflow is essential to achieve high De Novo GLP-1 Peptide Sequencing Accuracy and ensure reproducible, regulatory-grade results.
Each step in the workflow plays a critical role in minimizing errors, improving data quality, and maximizing sequence confidence.
A structured workflow is essential, as outlined in GLP-1 analog peptide sequencing workflow.
Step-by-Step Process
1. Sample Preparation
High-quality sample preparation is the foundation of accurate sequencing.
- Peptide purification to remove impurities
- Desalting to eliminate ion suppression effects
Impact: Improves signal clarity and enhances MS performance
2. LC Separation (Liquid Chromatography)
LC separates peptide components before MS analysis.
- Reduces sample complexity
- Minimizes co-eluting compounds
Impact: Produces cleaner spectra and better fragmentation results
3. High-Resolution MS/MS Acquisition
Accurate mass measurement and fragmentation are critical for sequencing.
- Use of HRMS systems (Orbitrap/QTOF)
- Application of multiple fragmentation modes (CID, HCD, ETD)
Impact:
- Improves sequence coverage
- Enables detection of modifications
- Enhances confidence in fragment identification
4. De Novo Algorithm Processing
Advanced software reconstructs peptide sequences from MS/MS data.
- Automated sequence prediction
- Use of confidence scoring algorithms
Impact:
- Speeds up analysis
- Provides initial sequence candidates
- Identifies possible PTMs
5. Manual Expert Review
Expert validation is essential for high-confidence results.
- Verification of ambiguous residues
- Interpretation of complex fragmentation patterns
- Cross-checking algorithm outputs
Impact:
- Resolves uncertainties
- Ensures regulatory compliance
- Improves final accuracy
6. Final Reporting
Comprehensive reporting ensures clarity and usability of results.
- Confidence scoring
- Structural confirmation
- Documentation for regulatory submission
Impact:
- Provides reliable, decision-ready data
- Supports drug development and quality control
Key Insight
Maximum De Novo GLP-1 Peptide Sequencing Accuracy is achieved when:
- Clean sample + Effective LC separation
- High-resolution MS + Multi-fragmentation
- Advanced algorithms + Expert validation
Result: Highly accurate, reproducible, and regulatory-compliant peptide sequencing outcomes.
5: Accuracy Comparison: De Novo vs Database Sequencing
De novo sequencing is slightly less accurate than database matching but far more versatile for unknown peptides.
| Feature | De Novo Sequencing | Database Sequencing |
|---|---|---|
| Reference required | No | Yes |
| Accuracy | 85–99% | 95–100% |
| Novel peptide detection | Excellent | Limited |
| GLP-1 analog suitability | High | Moderate |
6: Applications Where Accuracy is Critical
High de novo GLP-1 peptide sequencing accuracy is essential across multiple regulatory, analytical, and research applications. Precise sequencing ensures data reliability, supports compliance, and minimizes risks in drug development and quality control.
Key Use Cases
1. Drug Development
Accurate sequencing is crucial for confirming the primary structure of GLP-1 peptides during early and late-stage development.
Importance:
- Ensures correct peptide identity
- Supports structure-function relationship studies
- Reduces risk of development failures
2. Biosimilar Comparison
In biosimilar development, even minor sequence variations can impact efficacy and safety.
Importance:
- Detects amino acid substitutions or modifications
- Confirms similarity with reference products
- Supports comparability studies
3. Impurity Profiling
High-accuracy sequencing enables identification of unknown impurities and degradation products.
Importance:
- Detects low-level impurities
- Characterizes structural variants
- Ensures product purity and safety
Supported by GLP-1 peptide impurity characterization.
4. Stability Studies
Monitoring peptide integrity over time is critical for determining shelf life and storage conditions.
Importance:
- Identifies structural changes and degradation pathways
- Supports formulation optimization
- Ensures long-term product stability
Learn more: GLP-1 peptide stability analytical methods
5. Regulatory Submissions
Regulatory agencies require highly accurate and well-documented sequencing data.
Importance:
- Meets stringent requirements of agencies like FDA and EMA
- Provides validated analytical evidence
- Ensures compliance with global guidelines
Aligned with regulatory requirements for GLP-1 peptide characterization.
Key Insight
Accurate GLP-1 sequencing is not just a technical requirement—it is a regulatory and scientific necessity that directly impacts:
- Product quality
- Patient safety
- Approval success
Result:
Reliable, compliant, and high-confidence peptide characterization across the product lifecycle.
7: Expert Insights from ResolveMass Laboratories Inc.
Expert-led workflows significantly enhance sequencing accuracy and reliability.
ResolveMass offers advanced expertise in GLP-1 peptide sequencing and characterization services.
At ResolveMass, the focus is on:
- State-of-the-art HRMS platforms
- Customized sequencing strategies for GLP-1 analogs
- Regulatory-compliant reporting standards
- Deep expertise in peptide characterization
What Sets Expert Labs Apart
- Hybrid fragmentation strategies
- Advanced spectral interpretation
- Experience with complex peptide modifications
- High reproducibility and validation standards
8: Limitations of De Novo Sequencing
Despite high accuracy, de novo sequencing is not flawless and requires careful interpretation.
Common Limitations
- Ambiguous residues (Leu/Ile)
- Incomplete fragmentation
- Noise interference in spectra
- Dependence on instrument quality
This is why manual verification and orthogonal techniques are often required.
9: Future Trends in Sequencing Accuracy
Emerging technologies are pushing De Novo GLP-1 Peptide Sequencing Accuracy closer to 100%.
Innovations to Watch
- AI-powered sequence prediction
- Improved fragmentation techniques
- Ultra-high-resolution mass spectrometry
- Integrated multi-omics approaches
Conclusion:
De Novo GLP-1 Peptide Sequencing Accuracy can reach up to 99% when advanced instrumentation, optimized workflows, and expert validation are combined.
While challenges remain—especially with modified GLP-1 analogs—modern analytical strategies have made de novo sequencing a powerful and reliable tool for pharmaceutical research and regulatory applications.
For organizations working with GLP-1 peptides, investing in high-quality sequencing workflows is not optional—it is essential for ensuring accuracy, compliance, and product integrity.
For pharmaceutical organizations, investing in high-quality sequencing solutions—such as
GLP-1 peptide sequencing CRO services—is essential for ensuring:
- Product quality
- Regulatory compliance
- Patient safety
Frequently Asked Questions:
De novo GLP-1 peptide sequencing is an analytical technique used to determine the amino acid sequence of GLP-1 peptides directly from MS/MS data without relying on reference databases. It is essential for analyzing novel peptides, modified analogs, and unknown impurities.
The accuracy typically ranges from 85% to 99%, depending on the analytical conditions. With optimized high-resolution mass spectrometry (HRMS) and advanced workflows, accuracy can reach up to 95–99%.
GLP-1 peptide sequencing is challenging due to:
-Post-translational modifications (PTMs)
-Isobaric amino acids (e.g., Leu/Ile)
-Complex fragmentation patterns
-Longer peptide chain length
These factors make sequence reconstruction more difficult compared to standard peptides.
Accuracy can be improved by:
-Using high-resolution MS systems (Orbitrap, QTOF)
-Applying multiple fragmentation techniques (CID, HCD, ETD)
-Optimizing sample preparation
-Utilizing advanced bioinformatics tools
-Performing expert manual validation
-De novo sequencing: Does not require a reference sequence and is ideal for unknown or modified peptides
-Database sequencing: Matches MS data with known sequences and generally offers slightly higher accuracy
De novo is more flexible, especially for GLP-1 analogs and impurities.
Reference
- Maheshwari D, Badgujar D, Kumar G, Sharma N. Peptide Mapping Using Multienzyme Digestion Strategies Integrated with LC‐HRMS Workflow: A Case Study. Journal of Peptide Science. 2026 Mar;32(3):e70089.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.70089
- Arbouche N, Ameline A, Gheddar L, Raul JS, Kintz P. Glucagon like peptide-1 analogues analysis in whole blood samples by the use of LC-ESI-HRMS method. Toxicologie Analytique et Clinique. 2025 Mar 1;37(1):S61.https://www.sciencedirect.com/science/article/pii/S2352007824002920
- Arbouche N, Blanchot A, Raul JS, Kintz P. Semaglutide and health risk: development and validation of a LC-HRMS method for testing semaglutide in whole blood and application to real cases. Journal of Chromatography B. 2024 Jul 15;1242:124187.https://www.sciencedirect.com/science/article/pii/S157002322400196X
- Badgujar D, Bawake S, Chawathe A, Sharma N. Size‐exclusion LC‐UV/HRMS based method for the analysis of aggregates in synthetic GLP‐1 analog liraglutide and evaluation of excipient impact on aggregation. Biomedical Chromatography. 2024 Oct;38(10):e5983.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bmc.5983
- Badgujar D, Bawake S, Sharma N. A comprehensive study on the identification and characterization of major degradation products of synthetic liraglutide using liquid chromatography‐high resolution mass spectrometry. Journal of Peptide Science. 2025 Jan;31(1):e3652.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.3652
- Kim SH, Kim SS, Kim HJ, Park EJ, Na DH. Peptide mapping analysis of synthetic semaglutide and liraglutide for generic development of drugs originating from recombinant DNA technology. Journal of Pharmaceutical and Biomedical Analysis. 2025 Apr 15;256:116682.https://www.sciencedirect.com/science/article/pii/S0731708525000238


