Introduction: Why Disulfide Bond Mapping in Biosimilars Matters
Disulfide Bond Mapping in Biosimilars is a critical part of biosimilar characterization and regulatory approval. It is not just a routine laboratory step. This process helps scientists confirm that a biosimilar has the same structural arrangement as the reference biologic. Regulatory agencies such as the FDA and EMA closely review disulfide bond data because incorrect bond formation can affect protein stability, biological activity, safety, and immunogenicity.
Modern biologics are highly complex molecules. A standard IgG1 monoclonal antibody contains multiple intra-chain and inter-chain disulfide bonds that must be correctly identified and confirmed. IgG4 antibodies are even more challenging because of their flexible hinge-region disulfides. In addition, bispecific antibodies, Fc-fusion proteins, and antibody-drug conjugates (ADCs) include engineered cysteines and advanced conjugation systems that make structural analysis more difficult.
Explore our comprehensive Biosimilar Characterization Using Mass Spectrometry services.
Because of these complexities, Disulfide Bond Mapping in Biosimilars has become one of the most important analytical requirements during biosimilar development. Regulatory agencies expect detailed LC-MS data that clearly demonstrates correct disulfide connectivity and structural similarity between the biosimilar and the originator product.
Protein folding directly affects receptor binding, pharmacokinetics, and long-term product stability. Even small differences in disulfide pairing may impact clinical performance. For this reason, accurate disulfide characterization is considered a strong indicator of biosimilar structural integrity.
Learn how to Prove Biosimilarity Using LC-MS for your regulatory filings.
Share via:
▶ Article Summary
- Disulfide bond mapping is a key higher-order structure (HOS) assessment required by regulatory agencies such as the FDA, EMA, and ICH Q6B to confirm structural similarity in biosimilars.
- LC-MS peptide mapping under both non-reduced and reduced conditions is widely recognized as the primary analytical approach for identifying cysteine-containing peptides and verifying correct disulfide linkages.
- Artificially formed scrambled disulfide bonds during sample handling can lead to inaccurate results, making careful pH management and rapid alkylation essential during preparation.
- Combining partial reduction techniques with advanced ETD/ECD fragmentation enables detailed characterization of complex antibodies containing multiple disulfide connections.
- Measuring free thiol levels with reagents such as DTNB or NEM provides valuable orthogonal evidence to support comprehensive disulfide characterization.
- Reliable interpretation depends on direct comparison of biosimilar and reference product disulfide profiles using the same validated analytical workflow.
- ResolveMass Laboratories Inc. offers advanced, regulatory-focused disulfide mapping solutions for monoclonal antibodies, ADCs, bispecific antibodies, and Fc-fusion biologics.
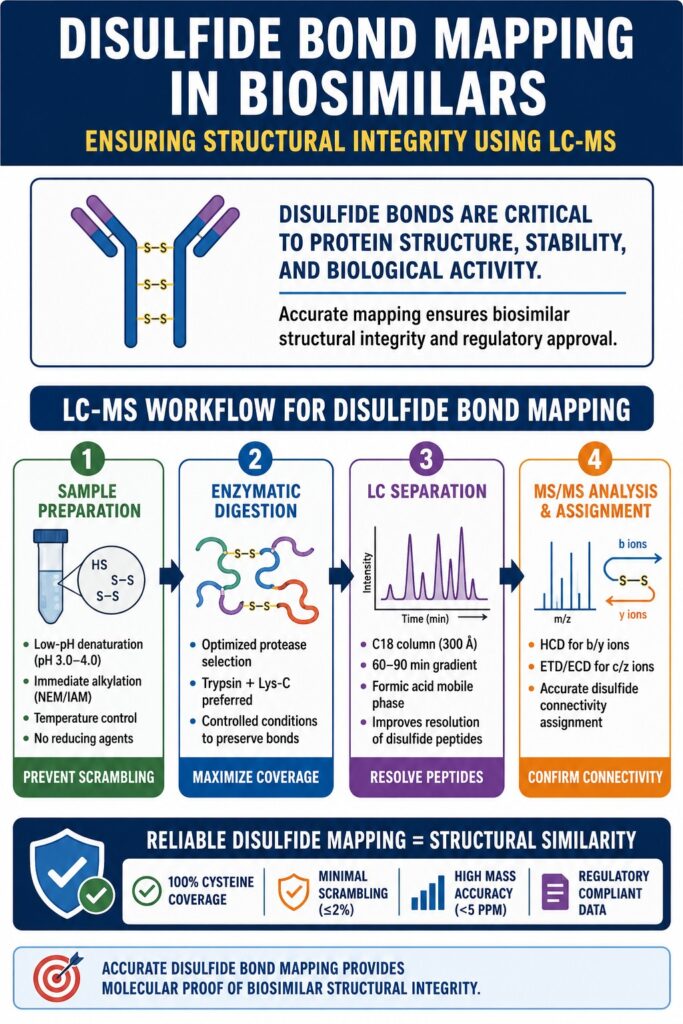
The LC-MS Workflow Architecture for Disulfide Bond Mapping in Biosimilars
A successful LC-MS workflow for disulfide mapping usually includes four major stages:
- Sample preparation and alkylation
- Enzymatic digestion
- Chromatographic separation
- MS/MS-based identification
Each stage must be carefully controlled to maintain native disulfide connectivity throughout the analysis.
Disulfide exchange reactions can occur quickly if pH, temperature, digestion conditions, or reagent quality are not properly managed. Poor control can create artificial disulfide bonds that interfere with accurate interpretation.
Today, regulatory submissions often require full documentation of all analytical procedures, including method parameters, system suitability testing, and scrambling controls. As a result, laboratories must prove that the method is reliable, reproducible, and scientifically justified.
Many organizations now combine peptide-level LC-MS with orthogonal methods such as native MS, HDX-MS, and ion mobility analysis. This multi-technique strategy improves confidence in biosimilar comparability assessments.
Read more about high-resolution Peptide Mapping in Biosimilars to ensure structural accuracy.
Phase 1: Sample Preparation — The Most Critical Step
Sample preparation is often the most sensitive stage in Disulfide Bond Mapping in Biosimilars. During denaturation and digestion, proteins can undergo unwanted thiol-disulfide exchange reactions that change the native bond structure.
Low-pH denaturation is commonly used to reduce scrambling. Guanidine-HCl at pH 3.0–4.0 helps protonate free thiols and lowers their reactivity. Maintaining acidic conditions early in the workflow is one of the best ways to preserve native disulfide structures.
Selective alkylation is another important step. Reagents such as N-ethylmaleimide (NEM) and iodoacetamide (IAM) are used to block free cysteine residues before digestion. This prevents artificial bond formation during analysis.
Temperature control also plays a major role. Digestion above 37°C can increase scrambling, especially when free cysteines are present. Many laboratories now use Lys-C digestion at room temperature under controlled pH conditions to reduce this risk.
Avoiding reducing agents is equally important. Even trace amounts of DTT or TCEP can reduce native disulfides and compromise the analysis.
Discover our specialized Intact Mass Analysis for Biosimilars to confirm molecular weight integrity.
Phase 2: Enzymatic Digestion Strategy
Protease selection directly affects the quality of disulfide mapping data. Different enzymes generate peptide fragments with different sizes and sequence coverage.
| Protease | Cleavage Specificity | Disulfide Mapping Utility | Known Limitation |
|---|---|---|---|
| Trypsin | Arg↓, Lys↓ | Gold standard for mAb peptide maps; generates manageable peptide lengths | Missed cleavages near Cys residues are common; Pro-adjacent sites resist cleavage |
| Lys-C | Lys↓ | Generates longer peptides; useful for hinge disulfides poorly resolved by trypsin | Large peptides may require MS² fragmentation beyond HCD capability |
| Asp-N | ↓Asp, ↓Cys | Generates Cys-terminal peptides; orthogonal coverage to trypsin | Non-specific cleavage; can produce many small fragments complicating assignment |
| Glu-C (V8) | Glu↓, Asp↓ (buffer-dependent) | Valuable orthogonal enzyme for regions missed by trypsin | Incomplete digestion and buffer dependency require optimization |
| Trypsin + Lys-C | Combined | Near-complete sequence coverage; preferred for regulatory submissions | Increased sample complexity; longer data processing time |
Phase 3: Reversed-Phase LC Separation
Disulfide-linked peptides are generally larger and more hydrophobic than reduced peptides. This makes chromatographic separation more difficult.
C18 columns with 300 Å pore sizes are commonly selected because they better support large peptide structures. Long acetonitrile gradients lasting 60–90 minutes improve peptide resolution and reproducibility.
Mobile phase composition is also important. Trifluoroacetic acid improves peak shape but can suppress ionization efficiency. Formic acid is often preferred because it supports better MS sensitivity while maintaining acceptable chromatography.
Many laboratories standardize LC separation temperatures around 60°C to improve reproducibility between runs.
Well-optimized chromatography helps separate native disulfides from scrambled artifacts and supports accurate peptide identification.
See how we handle Post-Translational Modifications (PTMs) in Biosimilars during LC separation.
Phase 4: MS/MS Fragmentation and Assignment
MS/MS fragmentation provides the final confirmation of cysteine connectivity.
Higher-energy Collisional Dissociation (HCD) is commonly used because it generates informative b and y ions from peptide fragments. HCD works well for many monoclonal antibody regions.
For more complex disulfide structures, Electron Transfer Dissociation (ETD) and Electron Capture Dissociation (ECD) are extremely valuable. These methods preserve disulfide bonds during fragmentation and generate c and z ions that improve structural interpretation.
IgG4 antibodies present unique challenges because their hinge disulfides may exist in multiple structural forms. Regulatory agencies increasingly expect quantitative analysis of these species.
Advanced software tools help automate spectral interpretation, but expert manual review remains essential for confirming difficult disulfide assignments.
Ensure complete structural profiles with Glycosylation Analysis of Biosimilars.
Scrambled Disulfides: A Major Risk in Biosimilar Characterization
Scrambled disulfides are artificial bond pairings generated during sample preparation. They do not represent the original protein structure and can create misleading analytical results.
These artifacts occur when free thiols attack native disulfide bonds during denaturation or digestion. Scrambling becomes more likely at higher pH values and elevated temperatures.
Regulatory agencies may interpret unexplained disulfide heterogeneity as evidence of manufacturing inconsistency or altered higher-order structure. Therefore, laboratories must demonstrate that detected disulfide patterns reflect the authentic protein architecture.
Preventing scrambling requires:
- Low-pH denaturation
- Immediate alkylation
- Tight temperature control
- Elimination of reducing contaminants
Without these controls, the reliability of the entire characterization package may be questioned.
Understand the risks of degradation with Forced Degradation of Biosimilars studies.
Identifying and Distinguishing Scrambled Disulfides
Retention time analysis is a useful method for identifying scrambled disulfides. Native peptides usually appear at consistent retention times, while scrambled species often show variable behavior.
Mass accuracy alone is not enough because scrambled peptides may have the same mass as native peptides. MS/MS fragmentation and chromatography are required for proper identification.
Reduced peptide mapping provides an important orthogonal approach for confirming cysteine involvement in native disulfides.
NEM-quench experiments are also highly effective. In these experiments, excess NEM is added immediately after denaturation to block free thiols before digestion. This helps scientists measure scrambling levels directly.
Combining multiple analytical strategies improves confidence in structural assignments and supports regulatory submissions.
Specific Disulfide Challenges in Biosimilar Comparability
Biosimilar developers must show that disulfide bond distribution and heterogeneity closely match the reference product.
Even small differences in variant abundance may raise regulatory concerns. Advanced biologics such as bispecific antibodies and ADCs create additional complexity because of engineered cysteines and linker systems.
As biosimilar products become more sophisticated, laboratories need highly sensitive and reproducible analytical workflows.
Explore our Regulatory Support for Generic Drug Development to navigate complex submission requirements.
Free Thiol Content and Regulatory Importance
Free thiol content is an important indicator of incomplete disulfide formation or unintended reduction.
For a standard IgG1 antibody, elevated free thiol levels may suggest instability or manufacturing problems.
Common orthogonal methods include:
- Ellman’s reagent (DTNB) analysis
- Isotope-dilution MS using NEM-d5
Regulators often review free thiol data alongside peptide mapping results to evaluate manufacturing consistency.
Inter-Chain vs. Intra-Chain Disulfide Heterogeneity
Reference biologics naturally contain low levels of disulfide heterogeneity. A biosimilar may still be acceptable if similar levels are observed.
However, detecting low-abundance variants requires highly sensitive analytical methods.
Many standard peptide mapping workflows only detect variants above 1–2% abundance. Lower-level variants may require enrichment strategies or advanced acquisition methods.
Understanding the natural heterogeneity profile of the reference product is essential for defining biosimilar acceptance criteria.
ADC Disulfide Mapping and Drug-Linker Complexity
Antibody-drug conjugates create additional analytical challenges because disulfides are often modified during conjugation.
In many ADC systems, inter-chain disulfides are partially reduced and re-bridged with linker molecules.
Before conjugation, scientists must confirm that the unconjugated antibody matches the reference product. After conjugation, the focus shifts to linker attachment sites and modified cysteine residues.
Hydrophobic interaction chromatography combined with LC-MS is often used to evaluate drug-antibody ratio distributions and conjugation profiles.
| Molecule Type | No. of Disulfide Bonds (typical) | Key Mapping Challenge | Recommended MS Strategy |
|---|---|---|---|
| IgG1 mAb | 16 intra + 4 inter = 20 | Hinge disulfide stoichiometry; CH1-CL assignment | Trypsin + Lys-C; HCD + ETD |
| IgG4 mAb | 16 intra + 2–4 inter = 18–20 | Half-antibody exchange; labile hinge Cys229 | HCD + native MS for intact half-Ab quantification |
| Bispecific (knob-into-hole) | Variable; engineered Cys at CH3 | Correct chain pairing; engineered Cys assignment | Asp-N + Trypsin combination; ECD fragmentation |
| Fc-Fusion Protein | Depends on fusion partner | Linker region disulfides; fusion partner-Fc interface | Multi-enzyme digest; middle-down LC-MS |
| Thiol-conjugated ADC | Reduced inter-chain Cys used for conjugation | Drug loading site confirmation; unconjugated Cys detection | HIC-MS + tryptic peptide map on DAR fractions |
Partial Reduction Strategies for Complex Disulfide Resolution
Partial reduction is a specialized technique used to simplify complex disulfide structures.
By carefully reducing selected disulfides, scientists can generate intermediate structures that help reveal bond hierarchy and connectivity.
This approach is especially useful when non-reduced peptide mapping cannot resolve overlapping peptide clusters.
Controlled TCEP or DTT concentrations are typically used for selective reduction. Reduced species are immediately quenched with alkylating agents to prevent re-oxidation or scrambling.
The resulting samples are analyzed by LC-MS to determine which disulfide bonds were reduced at each stage.
Similar reduction profiles between biosimilars and reference products provide additional evidence of structural comparability.
Data Interpretation and Regulatory Submission Requirements
A regulatory-ready disulfide bond map must fully characterize all cysteine residues in the protein.
Submission packages generally include:
- Annotated chromatograms
- MS/MS spectra
- Mass accuracy tables
- Sequence assignments
- Orthogonal confirmation data
Chromatograms should clearly identify disulfide-linked peptides with retention times, m/z values, and charge states.
High-resolution MS instruments are expected to deliver strong mass accuracy and reliable fragmentation data.
Overlay chromatograms comparing biosimilars with reference products are increasingly important for regulatory review.
Scrambling control studies and free thiol quantification should also be included to support analytical conclusions.
Acceptance Criteria Framework
| Attribute | Recommended Acceptance Criterion | Regulatory Basis |
|---|---|---|
| Disulfide bond connectivity | 100% match to reference product connectivity map; all Cys assigned | FDA Biosimilar Guidance (2015); ICH Q6B |
| Scrambled disulfide content | ≤2% relative peak area in non-reduced peptide map | Internal specification; justified by stability and bioactivity data |
| Free thiol content | ≤0.1 mol free Cys per mol antibody (IgG1) | Industry benchmark; batch-to-batch consistency criterion |
| MS mass accuracy | <5 ppm for Orbitrap; <10 ppm for Q-TOF instruments | Instrument performance specification |
| Sequence coverage (Cys-containing peptides) | ≥98% of total Cys residues assigned | ICH Q6B completeness requirement |
Access our specialized Analytical Services for Generic Drug Development for precise data generation.
Orthogonal Methods Supporting Disulfide Bond Mapping in Biosimilars
Although LC-MS peptide mapping remains the primary analytical tool, orthogonal methods strengthen structural characterization.
Common Orthogonal Techniques
| Method | Purpose |
|---|---|
| Native MS | Confirms intact molecular mass and overall disulfide integrity |
| DSC | Evaluates thermal stability and protein folding |
| HDX-MS | Measures backbone flexibility and solvent accessibility |
| Ion Mobility MS | Assesses conformational compactness and structural distribution |
These complementary methods provide additional evidence of higher-order structural similarity.
Regulatory Expectations for Disulfide Bond Mapping in Biosimilars
Regulatory expectations continue to evolve. Agencies now require highly detailed and quantitative disulfide characterization data.
Common regulatory concerns include:
- Incomplete sequence coverage
- Weak MS/MS confirmation
- Insufficient free thiol analysis
- Poor scrambling controls
Emerging techniques such as top-down and middle-down LC-MS are becoming more common in regulatory submissions.
For complex molecules like bispecific antibodies and Fc-fusion proteins, both peptide-level and subunit-level characterization are increasingly expected.
How ResolveMass Laboratories Inc. Performs Disulfide Bond Mapping in Biosimilars
ResolveMass Laboratories Inc. provides advanced LC-MS workflows specifically designed for biosimilar disulfide characterization.
The laboratory combines:
- Non-reduced peptide mapping
- Reduced peptide mapping
- Partial reduction studies
- Native mass spectrometry
Orbitrap-based LC-MS/MS systems with ETD and ECD fragmentation capabilities support detailed analysis of complex biologics, including:
- IgG1 antibodies
- IgG2 antibodies
- IgG4 antibodies
- Bispecific antibodies
- Fc-fusion proteins
- ADCs
Strict anti-scrambling controls are included in every workflow. These include low-pH denaturation, NEM-quench studies, and tightly controlled digestion conditions.
ResolveMass Laboratories also provides regulatory-ready reports designed to support IND, BLA, and MAA submissions.
Conclusion: Why Disulfide Bond Mapping in Biosimilars Is Essential
Disulfide Bond Mapping in Biosimilars remains one of the most important analytical tools for proving structural similarity between a biosimilar and its reference product.
A successful workflow requires:
- Anti-scrambling sample preparation
- Optimized digestion strategies
- High-resolution chromatography
- Advanced MS/MS fragmentation
- Complete cysteine coverage
Incomplete disulfide characterization can lead to regulatory delays, additional studies, or failure to demonstrate biosimilarity.
On the other hand, a strong disulfide mapping package significantly strengthens analytical similarity claims. Orthogonal methods such as native MS, HDX-MS, DSC, and ion mobility analysis further improve confidence in structural comparability.
As biosimilars become more complex, the importance of advanced LC-MS characterization will continue to increase. Companies developing biosimilars must maintain robust and regulatory-compliant analytical capabilities to meet growing industry expectations.
Ultimately, Disulfide Bond Mapping in Biosimilars provides direct molecular proof that a biosimilar reproduces the same structural foundation as the originator biologic.
Ready to scale? Learn about our Manufacturing Scale-Up for Generic Drugs to bring your product to market.
Frequently Asked Questions: Disulfide Bond Mapping in Biosimilars
For non-reduced peptide mapping, laboratories commonly use a 2.1 × 150 mm C18 column with a 300 Å pore size because it can efficiently separate large disulfide-linked peptides. A slow acetonitrile gradient over 75–90 minutes at around 60°C helps improve peptide resolution and reproducibility. Smaller pore-size columns designed for small molecules are generally not suitable for complex antibody peptide analysis.
Advanced fragmentation methods such as ETD and ECD allow peptide backbone fragmentation while keeping the disulfide bond intact. This helps identify exactly which peptide chains are connected through cysteine linkages. HCD can also support disulfide analysis, but ETD usually provides clearer and more reliable connectivity information for complex structures.
In most biosimilar studies, at least three reference product lots are analyzed to understand natural product variability. Testing multiple lots under the same analytical conditions improves confidence in comparability assessments. Regulatory agencies also expect developers to consider lot-to-lot variation when setting biosimilar acceptance criteria.
There is no single global regulatory limit for scrambled disulfides, but many companies aim to keep them below approximately 2% of the total peptide signal. Lower levels help demonstrate good process control and structural consistency. Any acceptable limit should be scientifically justified with data showing no impact on safety, efficacy, or stability.
Native mass spectrometry is useful for evaluating overall protein integrity and confirming intact molecular mass. However, it does not provide detailed peptide-level information about exact cysteine pairings. Regulatory agencies still require LC-MS/MS peptide mapping because it delivers residue-specific disulfide connectivity data.
A complete regulatory package should include annotated chromatograms, MS/MS spectra, peptide assignment tables, and free thiol analysis results. Comparative chromatograms between the biosimilar and reference product are also important. The final report should clearly confirm complete cysteine assignment and describe any detected disulfide-related variants.
High mass accuracy is essential because many disulfide-linked peptides have very similar masses. Instruments such as Orbitrap systems provide highly accurate measurements that improve confidence in peptide identification and structural assignment. Better resolution also helps detect low-abundance variants and complex disulfide species.
For originator biologics, disulfide mapping mainly confirms the molecule’s structural design during development. In biosimilars, the process is more demanding because developers must directly compare the biosimilar with the reference product under identical conditions. Biosimilar studies also require consistency testing across multiple manufacturing batches.
Reference:
- Segu, Z., Stone, T., Berdugo, C., Roberts, A., Doud, E., & Li, Y. (2020). A rapid method for relative quantification of N-glycans from a therapeutic monoclonal antibody during trastuzumab biosimilar development. MAbs, 12(1), 1750794. https://pmc.ncbi.nlm.nih.gov/articles/PMC7188402/
- Nupur, N., Joshi, S., Guillarme, D., & Rathore, A. S. (2022). Analytical similarity assessment of biosimilars: Global regulatory landscape, recent studies and major advancements in orthogonal platforms. Frontiers in Bioengineering and Biotechnology, 10, 832059. https://pmc.ncbi.nlm.nih.gov/articles/PMC8865741/
- D’Atri, V., Guillarme, D., & Beck, A. (2025). Biopharmaceutical analysis—current analytical challenges, limitations, and perspectives. Analytical and Bioanalytical Chemistry. https://pmc.ncbi.nlm.nih.gov/articles/PMC12783247/
- Berkowitz, S. A., Engen, J. R., Mazzeo, J. R., & Jones, G. B. (2012). Analytical tools for characterizing biopharmaceuticals and the implications for biosimilars. Nature Reviews Drug Discovery, 11(7), 527–540. https://pmc.ncbi.nlm.nih.gov/articles/PMC3714370/



