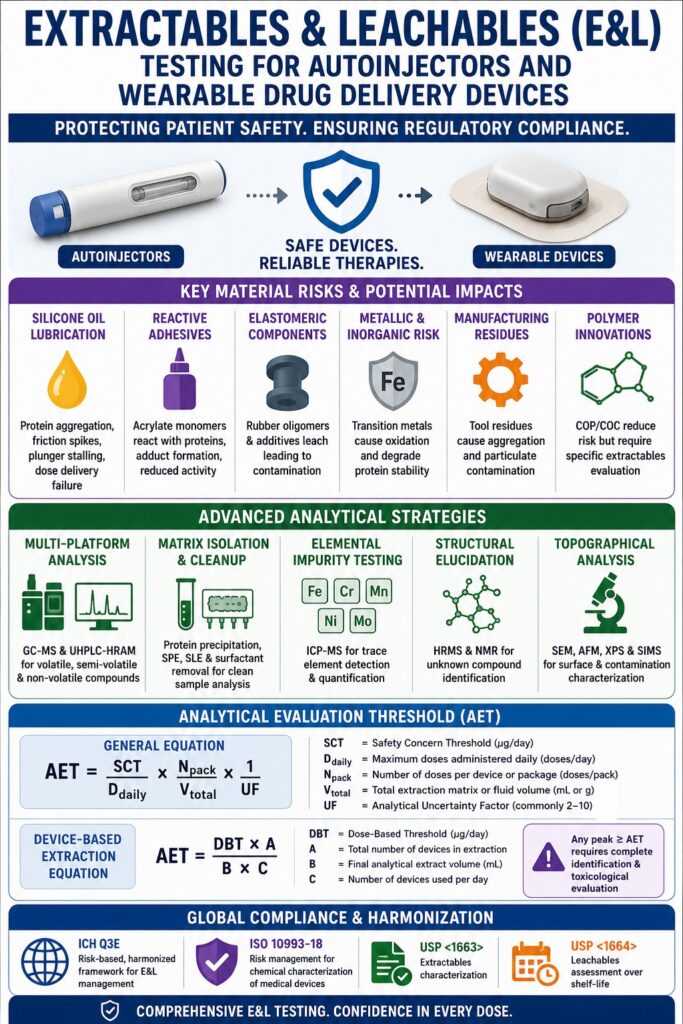
Primary Regulatory and Technical Challenges in Combination Device Development
Chemical migration assessment for drug-device combination systems plays a central role in connecting medical device biocompatibility requirements with pharmaceutical impurity evaluations to protect patient safety and support regulatory acceptance. Conducting comprehensive Extractables and Leachables Testing for Autoinjectors establishes a detailed safety framework for the finished product and strengthens quality assurance across large-scale commercial manufacturing operations. In contrast to conventional vial-and-stopper systems, automated delivery platforms introduce far more complex primary packaging interactions, requiring manufacturers to monitor chemical species released from specialized lubricants, reactive adhesives, elastomeric laminates, and other internal device materials.
The growing use of highly sensitive biologics, advanced therapies, and high-viscosity formulations significantly increases these risks because even trace-level contaminants can destabilize delicate protein structures. As a result, a robust extractables and leachables strategy has become an essential component of regulatory compliance, product quality management, and long-term patient protection.
Learn more about the fundamentals of chemical migration in pharmaceuticals: Explore Extractables and Leachables in Pharmaceutical Products
Share via:
Summary:
- Extractables and Leachables (E&L) testing is essential for autoinjectors and wearable drug delivery devices to ensure patient safety, product stability, and regulatory compliance.
- Complex device materials such as silicone lubricants, elastomers, adhesives, glass, and metals can release chemical migrants that may cause protein aggregation, contamination, oxidation, or device malfunction.
- Biologics and advanced therapies are highly sensitive to trace-level leachables, which can alter protein structure, reduce therapeutic efficacy, and increase immunogenicity risks.
- Modern wearable injectors increasingly use COP and COC polymers to reduce extraction risks, but these systems still require extensive testing for residual monomers, additives, adhesives, and electronic component emissions.
- Advanced analytical techniques including GC-MS, UHPLC-HRAM MS, ICP-MS, CAD, and NMR are used to detect volatile, semi-volatile, non-volatile, and elemental impurities at trace levels.
- Regulatory compliance requires alignment with ISO 10993-18, USP <1663>/<1664>, and the evolving ICH Q3E framework, which promotes a science-based and risk-managed E&L strategy.
- The Analytical Evaluation Threshold (AET) is a critical toxicological benchmark used to determine which detected compounds require identification and safety qualification during E&L studies.
Material Complexities and Chemical Migration Risk Profiling
Device components produced from advanced plastics, elastomers, and metallic materials can release a broad spectrum of organic and inorganic contaminants under operational stress conditions. These material systems create complex interactions that require detailed risk characterization to maintain product stability and compatibility throughout the product lifecycle.
Tribological Integrity and Silicone Oil Lubrication Risks
Polydimethylsiloxane oil is commonly applied to syringe barrels to reduce glide force during injection. However, silicone oil can migrate into the drug formulation, resulting in protein aggregation and disruption of the mechanical delivery system. During manufacturing, silicone oil is often distributed unevenly along the inner barrel surface, leading to lubrication inconsistencies over time. In autoinjector systems operated through automated spring-based mechanisms, this non-uniform coating can generate localized friction increases. Elevated resistance may interrupt plunger movement during administration, causing incomplete dose delivery or altered injection kinetics that negatively affect therapeutic performance.
In addition, free silicone oil droplets may function as nucleation centers for therapeutic proteins, accelerating the generation of sub-visible particulate matter and altering critical quality attributes of the drug product.
Understand the specific risks associated with biologic formulations: Read about Extractables and Leachables in Biopharma
Reactive Adhesives and Acrylate Adduct Formation Mechanisms
UV-cured acrylate adhesives used for needle bonding can release unreacted monomers and photoinitiators capable of chemically modifying therapeutic proteins. Residual oligomers, photoinitiator fragments, and unreacted acrylate species frequently migrate from adhesive junctions directly into the fluid pathway. Because acrylates possess strong electrophilic behavior, they readily react with nucleophilic amino acid residues located on biologic molecules.
Quantitative investigations have demonstrated that prolonged exposure under elevated temperature conditions can result in irreversible adduct formation at lysine, histidine, and N-terminus sites. These reactions modify the protein surface chemistry and may reduce receptor binding capability or overall therapeutic activity.
Elastomeric Formulations and Rubber Oligomer Leachables
Elastomeric closures, plungers, and protective caps may release rubber oligomers, antioxidants, sulfur derivatives, and vulcanization accelerators when exposed to liquid drug products. Uncoated rubber systems are especially vulnerable to leaching because compounding additives can migrate directly into the formulation matrix.
To reduce this risk, many modern autoinjectors utilize fluoropolymer-laminated plungers. Nevertheless, the integrity of the lamination layer must remain intact during manufacturing, assembly, transportation, and long-term storage. Micro-fissures or localized delamination can expose the underlying rubber surface to the formulation, reactivating the rubber-drug interface and causing unpredictable chemical migration that may contaminate production batches.
Evaluate the risks of nitrosamine and packaging-related contaminants: See Packaging Leachables and Nitrosamine E&L Insights
Metallic and Inorganic Degradation Pathways
Stainless steel needles and borosilicate glass syringe barrels can release transition metals and inorganic oxides that initiate degradation pathways within pharmaceutical formulations. Borosilicate glass may experience ion exchange, weathering, surface pitting, or erosion corrosion, resulting in the release of boron, silicon, sodium, and aluminum species into the formulation. These materials may appear as glass flakes or sub-visible particulate contaminants.
At the same time, hypodermic needles may leach trace quantities of iron, chromium, manganese, nickel, and molybdenum. Although these elemental impurities are typically present below direct toxicological concern levels, certain transition metals, particularly manganese, can strongly catalyze site-specific oxidation reactions in nearby proteins, ultimately reducing formulation stability.
Syringe Manufacturing Tool Residues
Manufacturing equipment used during primary container fabrication can introduce trace residues that persist even after automated cleaning processes. These residues may subsequently interact with the final drug formulation.
For instance, tungsten pins used during glass syringe barrel formation generate tungsten oxide vapors that deposit onto internal glass surfaces. In acidic formulations with a pH below 5.0, these residues can polymerize into tungstate polyanions that interact electrostatically with proteins, triggering severe macromolecular aggregation.
Similarly, nylon transport pins subjected to continuous mechanical wear may release microscopic polymer fragments that adhere to syringe interiors. These particles can later detach and enter the drug product as particulate contaminants.
| Component | Common Construction Materials | Key Potential Migrants | Clinical or Mechanical Impact |
|---|---|---|---|
| Glass Barrel | Type I Borosilicate Glass | Boron, silicon, aluminum, sodium oxides | Glass delamination, surface pitting, sub-visible particulate formation |
| Plunger Stopper | Elastomeric Rubber (Bromobutyl/Chlorobutyl) | Rubber oligomers, zinc accelerators, antioxidants | Cellular toxicity, formulation contamination, API absorption |
| Hypodermic Needle | Stainless Steel | Iron, chromium, manganese, nickel, molybdenum | Heavy metal migration, catalyzed protein oxidation |
| Needle Adhesive | UV-Activated Acrylate Resins | Unreacted acrylate monomers, photoinitiators | Covalent protein adduct formation at lysine and histidine residues |
| Syringe Lubricant | Polydimethylsiloxane (Silicone Oil) | Free silicone polymer chains, cyclic siloxanes | Plunger stalling, friction spikes, protein aggregation |
| Manufacturing Pins | Tungsten and Nylon Alloys | Tungstate polyanions, nylon micro-particulates | Electrostatic protein aggregation ($pH < 5$), particulate contamination |
Examine real-world regulatory findings regarding syringe and device failures: Review FDA Extractables and Leachables Case Studies
Advanced Polymer Innovations in Wearable Drug Delivery Devices
Modern wearable injectors increasingly utilize Cyclic Olefin Polymers (COP) and Cyclic Olefin Co-polymers (COC) to reduce extraction risks associated with conventional glass packaging systems. These advanced polymers provide exceptional optical clarity, improved break resistance, and highly consistent low-energy surface properties that minimize the requirement for extensive silicone oil lubrication.
From an extractables standpoint, COP and COC materials generally exhibit significantly cleaner baseline chemical profiles, reducing the migration of inorganic ions and packaging additives. However, evaluation programs for these polymer systems must still focus on polymer-specific migrants, including residual monomers, low-molecular-weight oligomers, antioxidants, and specialized slip agents introduced during molding operations.
The structural design of wearable injectors also introduces additional risks associated with skin-contacting adhesives, embedded electronics, and multi-lumen fluid transfer pathways. Because these systems remain attached to patients for extended periods, adhesive layers require extensive extraction screening in parallel with internal fluid-contacting materials.
Chemical compounds originating from adhesive backings may permeate through external polymer housings and eventually reach the drug reservoir. Regulatory agencies therefore require wearable injector testing programs to evaluate adhesion durability, thermal stability, structural performance under realistic environmental conditions, and long-term volatile organic compound migration from internal electronic assemblies.
Discover how E&L requirements differ for emerging therapies: Read about E&L in Emerging Biologics and ATMPs
Macromolecular Vulnerabilities of Biologics and Advanced Therapies
Biopharmaceutical products and advanced therapies are highly vulnerable to trace-level chemical migrants because of their delicate three-dimensional conformations and sensitive surface chemistry. Leachables can disrupt the electrostatic, hydrophobic, and van der Waals interactions responsible for maintaining tertiary and quaternary protein structures.
Organic contaminants, including residual solvents, plasticizers, and polymer-derived surfactants, may penetrate hydrophobic protein cores, causing denaturation and structural unfolding. Once proteins unfold, buried hydrophobic residues become exposed, promoting self-association and the formation of visible or sub-visible particles. This aggregation represents a major regulatory concern because particulate matter may alter pharmacokinetics and contribute to vascular obstruction or localized tissue irritation during administration.
Direct interactions between chemical migrants and active pharmaceutical ingredients can also induce irreversible molecular modifications that reduce therapeutic potency. Organic peroxides and transition metals can oxidize methionine and cysteine residues, while aldehydes and residual monomers may trigger deamidation reactions involving asparagine or glutamine residues.
These structural alterations modify the charge distribution and spatial conformation of biologic molecules, impairing receptor binding and accelerating systemic clearance. Chemical modification of protein surfaces may additionally generate highly immunogenic neo-epitopes. When a leachable covalently binds to a therapeutic protein, the immune system may no longer recognize the molecule as biologically native, potentially initiating anti-drug antibody (ADA) production.
Advanced Analytical Strategies for Extractables and Leachables Testing for Autoinjectors
Characterizing and quantifying trace-level migrants within highly complex drug matrices requires an orthogonal analytical strategy integrating advanced chromatography and mass spectrometry technologies. Dedicated Extractables and Leachables Testing for Autoinjectors enables comprehensive profiling across all volatility ranges, minimizing the possibility that critical contaminants remain undetected.
Consult with industry leaders for comprehensive device testing: Learn about ResolveMass Extractables and Leachables Testing Services
Multi-Platform Chromatographic and Mass Spectrometry Instrumentation
Combining gas chromatography and liquid chromatography with high-resolution mass spectrometry enables simultaneous monitoring of volatile, semi-volatile, and non-volatile compounds. Volatile organic compounds (VOCs) and semi-volatile organic compounds (SVOCs), including residual solvents, monomers, antioxidants, and plasticizers, are typically analyzed using headspace and direct-injection GC-MS methodologies.
Controlled sample introduction procedures and carefully regulated injector temperatures are essential for preventing thermal degradation while maintaining reproducible response factors.
Non-volatile and polar compounds with high molecular weights are generally characterized using Ultra-High Performance Liquid Chromatography (UHPLC) coupled with High-Resolution Accurate Mass (HRAM) instruments such as Orbitrap or Quadrupole Time-of-Flight (Q-TOF) mass spectrometers.
Traditional UV and electrospray ionization mass spectrometry methods often fail to detect non-chromophoric or poorly ionizing unknown compounds. Incorporating Charged Aerosol Detection (CAD) provides a near-universal response mechanism independent of compound structure, improving quantification reliability for unknown non-volatile analytes without requiring dedicated reference standards.
Matrix Isolation and Advanced Sample Preparation Protocols
Highly concentrated protein formulations and non-ionic surfactants create substantial analytical challenges due to severe ionization suppression during electrospray ionization mass spectrometry analysis. Therapeutic proteins and surfactants such as Polysorbate 20 or Polysorbate 80 compete for ionization efficiency, reducing analyte sensitivity and increasing quantification limits.
To address these matrix effects, specialized sample preparation workflows must be implemented. Targeted protein precipitation methods, Solid-Phase Extraction (SPE), and Solid-Supported Liquid Extraction (SLE) are routinely used to isolate target leachables while removing interfering macromolecular components.
In addition, advanced surfactant depletion technologies help prevent chromatographic column fouling, eliminate inconsistent compound elution behavior, and improve instrument uptime while maintaining high recovery rates for trace-level migrants.
| Analytical Parameter | Target Compounds | Preferred Analytical Instrument Platform | Detection Optimization Tool |
|---|---|---|---|
| Volatility Screening (VOCs) | Residual solvents, low-weight monomers | Headspace Gas Chromatography-Mass Spectrometry (HS-GC/MS) | Thermal desorption direct sampling |
| Semi-Volatility (SVOCs) | Plasticizers, antioxidants, slip agents | Direct Injection GC-MS/MS (Triple Quadrupole) | Hot split/splitless injection ($280^\circ\text{C}$) |
| Non-Volatile Organics | Surfactants, polymer degradation products | UHPLC coupled to Orbitrap HRAM MS | Charged Aerosol Detection (CAD) response factors |
| Elemental Impurities | Transition metals (Fe, Cr, Mn, Ni, Mo) | Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) | High-transmission collision cells |
| Structural Elucidation | Unknown organic degradants | High-Resolution Orbitrap MS and NMR Spectroscopy | Internal databases and commercial spectral libraries |
Global Harmonization and Compliance Frameworks Under ICH Q3E
Regulating combination products requires the integration of pharmaceutical packaging requirements with global medical device biocompatibility standards. Device components within autoinjector systems, including external housings, activation sleeves, and skin-contacting adhesive materials, are governed by ISO 10993-18:2020, which defines a risk-management framework for chemical characterization.
This standard requires both simulated-use and exaggerated extraction studies to establish worst-case patient exposure scenarios. Simultaneously, fluid-contacting components and container-closure systems must comply with pharmaceutical guidance, particularly USP \langle1663\rangle for extractables characterization and USP \langle1664\rangle for leachables assessment throughout shelf-life stability programs.
The development of the ICH Q3E draft guideline represents a major advancement in global harmonization by establishing a unified scientific and risk-based framework for extractables and leachables management across the entire product lifecycle. ICH Q3E creates a consistent methodology for pharmaceutical combination products, reducing historical differences among regulatory authorities such as the FDA, EMA, MHRA, Health Canada, and PMDA.
This framework emphasizes science-based decision-making and encourages manufacturers to perform structured material risk assessments, understand upstream material compositions, and establish toxicological safety thresholds during early-stage development.
To support analytical screening activities, laboratories also apply physical and topographical characterization techniques to evaluate surface contamination, delamination behavior, and mechanical damage following stress testing.
| Topographical Method | Analytical Core Mechanism | Evaluation Application in Device Testing |
|---|---|---|
| TGA / DSC | Thermoanalytical profiling | Polymer degradation and thermal stability verification |
| X-Ray Fluorescence (XRF) | Elemental surface mapping | Screening for metallic catalysts and inorganic contamination |
| SEM / REM | Scanning Electron Microscopy | Visualization of micro-fractures, delamination, and sub-visible particulates |
| Atomic Force Microscopy (AFM) | Nanoscale surface topography | Evaluation of surface roughness changes from lubricant depletion |
| XPS / AES | Photoelectron and Auger Spectroscopy | Identification of ultra-trace chemical contamination at interfaces |
| SIMS-TOF | Time-of-Flight Secondary Ion Mass Spectrometry | Surface depth profiling of migrated lubricants and plasticizers |
Stay updated on the latest global harmonization guidelines: Read the ICH Q3E Guideline for Extractables and Leachables
Mathematical Derivation of the Analytical Evaluation Threshold
The Analytical Evaluation Threshold (AET) establishes a concentration-based benchmark used to determine which unknown compounds require formal identification and toxicological qualification. It is derived from the Safety Concern Threshold (SCT) and clinical dosing parameters and functions as a critical decision point during non-targeted screening studies.
For combination injection systems, the absolute Safety Concern Threshold (\text{SCT}) represents the dose-based exposure limit below which a leachable presents negligible carcinogenic or non-carcinogenic risk. For parenteral systems, the Product Quality Research Institute (PQRI) and multiple global regulatory agencies generally recommend an SCT of 1.5,\mu\text{g/day}.
The general equation used to convert the dose-based safety threshold into a concentration-based reporting threshold is:
AET=SCTDdaily×NpackVtotal×1UF\text{AET} = \frac{\text{SCT}}{D_{\text{daily}}} \times \frac{N_{\text{pack}}}{V_{\text{total}}} \times \frac{1}{\text{UF}}AET=DdailySCT×VtotalNpack×UF1
Where:
- SCT = Safety Concern Threshold expressed in micrograms per day (\mu\text{g/day})
- D{\text{daily}} = Maximum number of doses administered daily (\text{doses/day})
- N{\text{pack}} = Number of doses contained within a single device or package configuration (\text{doses/pack})
- V{\text{total}} = Total extraction matrix volume or fluid volume within the system (\text{mL} or \text{g})
- \text{UF} = Analytical Uncertainty Factor accounting for response factor variability during non-targeted screening
When extraction studies involve pooled devices within a defined extraction solvent volume, the following device-specific equation from ISO/TS 21726 and standard combination workflows is commonly applied:
AET=DBT×AB×C\text{AET} = \frac{\text{DBT} \times A}{B \times C}AET=B×CDBT×A
Where:
- \text{DBT} = Dose-Based Threshold corresponding to the toxicological exposure limit (\mu\text{g/day})
- A = Total number of devices included in the extraction protocol
- B = Final analytical extract volume after preparation (\text{mL})
- C = Number of devices clinically used by the patient per day
The Analytical Uncertainty Factor (\text{UF}) is incorporated to minimize the possibility of under-reporting highly toxic unknown compounds with poor relative response factors during mass spectrometry analysis. Standard screening workflows generally apply uncertainty factor values ranging from 2 to 10.
If an uncertainty factor is not applied (\text{UF} = 1), manufacturers must provide a detailed toxicological justification supported by an extensive response factor database. Every detected peak at or above the final calculated AET must undergo complete structural identification and formal toxicological review by qualified experts.
Deep dive into risk assessment methodologies: Understand ICH Q3E Extractables and Leachables Risk Assessment
Conclusion
Managing chemical migration within automated drug delivery systems requires a highly specialized, risk-based strategy that combines advanced analytical characterization with rigorous toxicological evaluation. Implementing validated Extractables and Leachables Testing for Autoinjectors is critical for minimizing both clinical and mechanical risks throughout the entire product lifecycle and for eliminating potential failure mechanisms before commercial release.
As automated delivery technologies continue progressing toward higher-volume reservoirs and extended on-body use durations, understanding molecular migration behavior across complex material interfaces becomes increasingly important. Maintaining compliance with evolving standards such as ISO 10993-18, USP \langle1663\rangle/\langle1664\rangle, and the harmonized ICH Q3E framework enables manufacturers to reduce the likelihood of late-stage failures, accelerate regulatory approvals, and maintain consistent product quality throughout commercialization.
Ensure your product meets U.S. regulatory standards: Explore E&L Requirements for U.S. Market Authorization
To discuss specialized study strategies or request a technical consultation for an autoinjector or wearable drug delivery development program, contact the scientific team directly at Resolve Mass Technologies Contact Page
Frequently Asked Questions
Extractables are chemical substances that can be released from device materials when exposed to aggressive laboratory conditions such as elevated temperatures, extended extraction periods, or strong solvents. These studies are designed to simulate worst-case scenarios and identify all potential chemical migrants. Leachables, in contrast, are the compounds that actually migrate into the drug product during normal storage, transportation, and clinical use, making them directly relevant to patient exposure and product safety.
Autoinjectors depend on precisely controlled mechanical force to deliver accurate drug doses. When silicone oil is distributed inconsistently inside the syringe barrel, certain regions develop higher friction during plunger movement. These friction increases can interrupt the delivery mechanism, causing incomplete injections, irregular injection speeds, or total device failure during administration.
UV-cured adhesives commonly used for needle bonding may release residual acrylate monomers and oligomers into the formulation pathway. These reactive compounds can chemically interact with amino acid residues on therapeutic proteins and form irreversible covalent bonds. Such modifications may alter protein structure, reduce therapeutic activity, and increase the likelihood of unwanted immune responses.
Tungsten residues may remain on syringe surfaces after the glass-forming process because tungsten pins are used during barrel manufacturing. In acidic drug formulations, these residues can transform into tungstate polyanions that interact strongly with therapeutic proteins. This interaction destabilizes protein structures and promotes large-scale aggregation, which can compromise formulation stability and product quality.
The Analytical Evaluation Threshold establishes the concentration level at which detected chemical compounds must undergo toxicological review and structural identification. It serves as a scientifically derived screening limit that helps laboratories focus on contaminants with potential patient safety implications. By applying the AET, manufacturers can efficiently distinguish significant chemical impurities from low-risk background signals.
The ICH Q3E guideline introduces a harmonized and risk-based framework for extractables and leachables evaluations across major international regulatory agencies. It aligns expectations for safety thresholds, analytical testing strategies, and lifecycle risk management. This global standardization reduces duplicate regional testing requirements and helps manufacturers streamline regulatory submissions for combination products.
Polysorbates are widely used surfactants in biologic formulations because they help stabilize proteins and minimize aggregation. However, their high concentration can interfere with LC-MS analysis by suppressing ionization signals within the electrospray source. This interference can mask low-level leachables, making specialized sample preparation and surfactant removal techniques necessary for accurate detection.
Cyclic Olefin Polymers and Cyclic Olefin Co-polymers provide strong mechanical durability, excellent optical clarity, and highly stable surface characteristics. These materials require less silicone oil lubrication compared to traditional glass systems, reducing the risk of silicone-related contamination. In addition, COP and COC materials demonstrate cleaner extractable profiles with lower inorganic ion migration, making them highly suitable for wearable drug delivery technologies.
Reference:
- U.S. Pharmacopeia. (n.d.). Extractables and leachables. USP. https://www.usp.org/impurities/extractables-and-leachables
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. (2025). ICH harmonised guideline: Guideline for extractables and leachables Q3E (Draft version, Step 2). ICH Database PDF
- U.S. Food and Drug Administration. (2025). Q3E guideline for extractables and leachables. U.S. Food and Drug Administration. FDA Q3E Guidance Page
- European Medicines Agency. (2025). Draft ICH Q3E guideline for extractables and leachables (EMA/CHMP/ICH/236669/2025). European Medicines Agency. EMA Draft ICH Q3E Guideline PDF



